| |
Sofosbuvir, Glecaprevir, Pibrentasvir, and Ribavirin as a Rescue Therapy in Difficult-to-Treat HCV Patients
|
| |
| |
Download the PDF here
Hepatology May 2021
Pangenotypic direct-acting antiviral (DAA) drugs have an HCV cure rate of >95% in almost all treated patients.(1, 2) When DAA treatment fails, retreatment must be guided by virus resistance profiles, and phase 3 trials have reported sustained virological responses (SVR) of 96%-98% after a 12-week course of sofosbuvir (SOF), velpatasvir (VEL), and voxilaprevir (VOX).(3) However, the management is more uncertain after SOF/VEL/VOX failure, and there is still insufficient evidence to support a particular retreatment. For instance, Dietz et al.(4) reported 77% SVR at 12 weeks (SVR12) in patients with different HCV profiles retreated with glecaprevir (GLE)/pibrentasvir (PIB), SOF, and ribavirin (RBV) for 12-24 weeks after SOF/VEL/VOX failure.
Clinical Observation
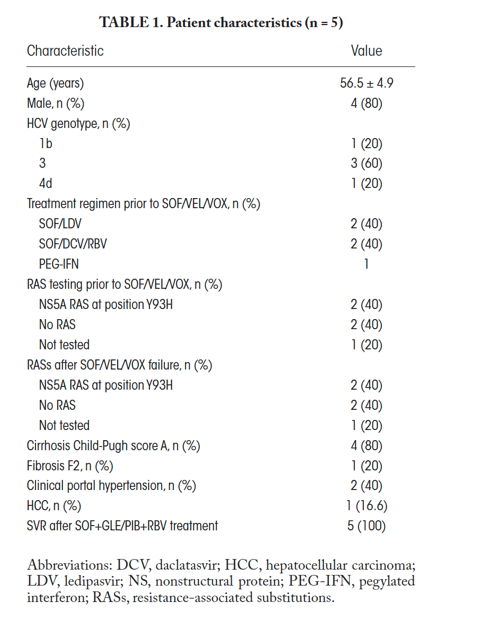
The characteristics of 5 patients treated at three tertiary care hospitals who failed a 12-week course of SOF/VEL/VOX rescue therapy for chronic HCV are shown in Table 1. Four out of 5 (80%) patients were male and had Child-Turcotte-Pugh (CTP) compensated cirrhosis, 2 of 5 (40%) had clinically significant portal hypertension (CSPH) as defined by the presence of esophageal varices on gastroduodenal endoscopy, and 1 had hepatocellular carcinoma (HCC). Three out of 5 (60%) had HCV genotype 3, 1 (20%) had genotype 1b, and 1 (20%) had genotype 4d. Eighty percent of patients had previously been treated with DAAs: 2 had received SOF and daclatasvir for 24 weeks and 2 had received SOF and ledipasvir for 12 weeks prior to SOF/VEL/VOX treatment.
Baseline resistance-associated substitution (RAS) results prior to SOF/VEL/VOX retreatment were available in 4 patients and showed nonstructural protein 5A (NS5A) RAS at position Y93H in 2 (50%) patients with genotype 3 HCV and no RASs in 1 patient; NS3A or NS5B RASs were not detected. RAS testing was also conducted after SOF/VEL/VOX treatment, and there were no changes in RAS status in any patient. All 5 patients were retreated with combined SOF and GLE/PIB with RBV for 16 weeks, and all (100%) achieved an SVR. Tolerance and adherence were excellent.
The 2 patients with CSPH and the patient with HCC had compensated CTP A cirrhosis with a Model for End-Stage Liver Disease (MELD) score <10. They were monitored for adverse events monthly by clinical examination and standard blood test. The patient with HCC also underwent a CT scan 3 months after retreatment. CTP and MELD scores did not worsen during treatment. Two patients died after achieving SVR, 1 from metastasis from the HCC diagnosed prior to salvage treatment and 1 from an extrahepatic cause.
Discussion
There are currently no clinical guidelines to support decision-making when retreating patients failing SOF/VEL/VOX rescue therapy. Genotype 3 HCV, consistent with 3/5 of our patients, is more likely to be difficult to treat. While Bourlière et al.(3) reported that 95% of genotype 3 patients treated with SOF/VEL/VOX achieved an SVR, the study only included a few genotype 3 infections in patients with cirrhosis, making it difficult to generalize results in this subgroup.
Interestingly, although treatment-emergent RASs can occur, we did not detect any RAS changes after SOF/VEL/VOX therapy. The only detected RAS was a substitution at NS5A amino acid position 93, which is known to confer a high level of resistance to NS5A inhibitors like velpatasvir. However, this substitution is still susceptible to PIB,(5) motivating our choice of retreatment. In contrast to Dietz et al.,(4) our patients received 16 weeks of treatment as standard (versus 12-24 weeks), resulting in an SVR12 of 100%. Furthermore, we only included patients with compensated liver disease as protease inhibitors are contraindicated in decompensated cirrhosis.
Our observations confirm that SOF/GLE/PIB/RBV represents a promising alternative rescue treatment when SOF/VEL/VOX fails, and further studies are necessary to validate this therapeutic option.
| |
| |
| |
|
|
|