| |
Motor function declines over time in human immunodeficiency virus and is associated with cerebrovascular disease, while HIV-associated neurocognitive disorder remains stable
|
| |
| |
2018 Journal of NeuroVirology - Isabel M. Elicer1,2 & Desiree Byrd1 & Uraina S. Clark1 & Susan Morgello1,3 & Jessica Robinson-Papp1
Download the PDF here
Abstract
HIV-associated neurocognitive disorders (HAND) remain prevalent in the combined antiretroviral therapy (CART) era, especially the milder forms. Despite these milder phenotypes, we have shown that motor abnormalities persist and have quantified them with the HIV Dementia Motor Scale (HDMS). Our objectives were to replicate, in an independent sample, our prior findings that the HDMS is associated with cognitive impairment in HIV, while adding consideration of age-associated comorbidities such as cerebrovascular disease, and to examine the longitudinal trajectories of cognitive and motor dysfunction. We included all participants enrolled in the Manhattan HIV Brain Bank (MHBB) from January 2007 to May 2017 who had complete baseline data (N = 164).
MHBB participants undergo standardized longitudinal assessments including documentation of comorbidities and medications, blood work, the HDMS, and neurocognitive testing. We found that motor dysfunction, cognitive impairment, and cerebrovascular disease were significantly associated with each other at baseline. Cerebrovascular disease independently predicted cognitive impairment in a multivariable model. Longitudinal analysis in a subset of 78 participants with ≥ 4 years of follow-up showed a stable cognition but declining motor function. We conclude that the HDMS is a valid measurement of motor dysfunction in HIV-infected patients and is associated with cognitive impairment and the presence of cerebrovascular disease. Cognitive impairment is mild and stable in CART-treated HIV; however, motor function declines over time, which may be related to the accrual of comorbidities such as cerebrovascular disease. Further research should examine the mechanisms underlying motor dysfunction in HIV and its clinical impact.
This study demonstrates that the HDMS is capable of measuring subtle motor abnormalities in a CART-era HIV population and that these abnormalities correlate with cognitive impairment. However, it also raises the question of whether motor and cognitive dysfunction in patients with long-standing HIV infection might be due in part to the effects of aging and vascular comorbidities.
Our participants had a high prevalence of cardiovascular comorbidities, with around 40% having hypertension, hyperlipidemia, and being smokers. Other groups have reported similar findings. A Chicago-based cohort of 87 HIV-infected patients older than 60 years reported a prevalence of 61% for hypertension and 26% for diabetes (Adeyemi et al. 2003). A study including 13 cohorts from North America and Europe that analyzed the causes of death of HIV patients from 1996 through 2006 showed that 6.5% of the deaths were attributable to cardiovascular disease (Sackoff et al. 2006). Modeling based on data from the CHORUS cohort estimated that 41% of HIV-infected individuals will die of non-HIV-related causes, and of these 35% will be due to cardiovascular causes (Braithwaite et al. 2005).
Cerebrovascular disease in HIV is also increasingly recognized; autopsy studies have demonstrated cerebral ischemic lesions in 4-34% of patients (Sharer and Kapila 1985; Connor et al. 2000; Berger et al. 1990; Mizusawa et al. 1988; Moskowitz et al. 1984), and clinical series have shown a prevalence of stroke between 0.6 and 5% (Arentzen et al. 2015; Benjamin et al. 2012; Evers et al. 2003). The etiology of cerebrovascular disease in HIV is likely related to a combination of traditional vascular risk factors and HIV-specific factors. HIV infection has been associated with greater adventitial inflammation of large brain arteries and dolichoectasia, compared to HIV-negative controls (Gutierrez et al. 2016), and in a recent review (Benjamin et al. 2016), several types of HIV-associated vasculopathies were defined. Given this, it is possible that an increasing burden of cerebrovascular disease in the aging HIV population is one explanation why motor abnormalities still accompany cognitive dysfunction, even when the most severe form (i.e., HAD) is in decline.
In summary, this study reinforces the continued relevance of motor dysfunction, as measured by the HDMS in HIV-infected patients. In response to current phenotypic changes in HAND, we proposed to rename it HIV Motor Scale (HMS). The advantages of this scale include that it is a bedside objective motor assessment, resistant to practice effects, and not influenced by a low premorbid function. We also confirmed the relationship with cognitive dysfunction, which in this aging population can be a result of a HIV-associated cognitive impairment or cerebrovascular disease. It is as yet unknown whether the accrual of the disorders of aging like cerebrovascular disease will henceforth change the phenotype of HAND, reversing the decline in the more severe forms, or whether the greater potency of today’s antiretrovirals and guidelines directing their earlier use (Panel on Antiretroviral Guidelines for Adults and Adolescents n.d.) will be sufficiently protective to preserve the milder HAND phenotype and perhaps prevent new cases from developing.
Introduction
Cognitive impairment has long been recognized as an important complication of human immunodeficiency virus (HIV) infection (McArthur et al. 1993; Navia et al. 1986). Based on clinical observations, early descriptions included cognitive as well as non-cognitive features, such as motor and behavioral abnormalities (Navia et al. 1986; Janssen et al. 1991). However, with the widespread use of combined antiretroviral therapy (CART), the clinical features of cognitive syndromes in HIV have changed over the last decades including a decline in the prevalence of severe forms, i.e., HIV-associated dementia (HAD), and an increasing prevalence of milder phenotypes. In response to these changes, in 2007, the Frascati criteria were developed, to include milder and asymptomatic forms of cognitive impairment under the umbrella of HIV-associated neurocognitive disorder (HAND) (Antinori et al. 2007).
Despite the decline of HAD and the rise of the milder phenotype of asymptomatic neurocognitive impairment (ANI), subtle motor abnormalities have persisted. Accordingly, we developed and validated the HIV Dementia Motor Scale (HDMS) to capture and quantify these findings. The HDMS is derived from standard elements of a neurologic examination which were selected based on the motor abnormalities included in the original descriptions of HAD (Janssen et al. 1991; Navia et al. 1986; Robinson-Papp et al. 2008). Our motivation for the HDMS was the rationale that motor abnormalities were still clinically observable in the era of CART despite the relatively milder phenotypes of HAND and that these motor abnormalities might contribute to diagnostic accuracy, by being relatively specific to HIV, and by being unaffected by confounders of neuropsychological testing such as low premorbid function. We demonstrated that the HDMS was associated with cognitive impairment in a CART-era cohort and also preliminary longitudinal analyses suggested that the change in HDMS over time paralleled that of cognitive function.
Ten years have passed since the development of the Frascati classification, CART has become even more potent, and HIV research has continued to focus on understanding HIV as a chronic disease which interacts with aging and comorbidities (Mateen and Mills 2012). Similarly, investigations of HAND must now consider the increasingly important effects of non-infectious comorbidities, particularly, cerebrovascular disease. HIV itself has been shown to be an independent risk factor for cerebrovascular disease (Gutierrez et al. 2017) and is estimated to be one of the most important causes of death in the CART-era HIV population (Braithwaite et al. 2005).
Thus, we undertook the present study to re-explore the issue of motor dysfunction as a feature of HAND. Specifically, our goals were as follows: (1) to compare the performance of the HDMS to two other measures of motor function: the motor domain of a neuropsychological testing battery (based on the Grooved Pegboard test performance) and the motor section of the Unified Parkinson Disease Rating Scale (UPDRS); (2) to replicate in an independent sample our prior findings that the HDMS was associated with neurocognitive dysfunction in HIV; (3) to determine whether relationships between motor dysfunction and cognitive impairment in HIV are affected by the presence of cerebrovascular disease, a common disorder of aging and HIV; and (4) to explore the relative longitudinal trajectories of change in cognitive and motor dysfunction in a subset of participants with at least 4 years of follow-up.
Study participants
The Manhattan HIV Brain Bank (MHBB, U24MH100931) is an ongoing prospective cohort and autopsy study founded in 1998, located in New York City, which serves as a research resource of nervous system tissues obtained at the time of death from highly characterized donors.
Results
Cohort characteristics
A total of 164 patients were included in the study. As shown in Table 1, the sample was diverse with regard to race, ethnicity, gender, and age, with participants ranging from 29 to 80 years old. The majority were CART treated, but medically complex, with a high prevalence of cardiovascular and metabolic comorbidities. Comparisons between gender, showed that on average, men were older by 5 years (P < 0.01) and they had 1.5 years of education more than women (P = 0.002). Among comorbidities, women had higher BMI values (mean for females = 29.9, mean for males = 25.0, P < 0.01) and therefore a higher prevalence of obesity status than men. No other characteristic was significantly different according to gender.
HDMS: correlations with other motor measures and clinical features
Our first goal was to explore the performance of the HDMS and compare it with other motor measures. Sixty-one participants (37.2%) scored zero on the HDMS, while the remainder (n = 103, 62.9%) had some degree of motor dysfunction. As shown in Table 2, among the five subscales of the HDMS, the reflexes and gait items were the most frequently abnormal.
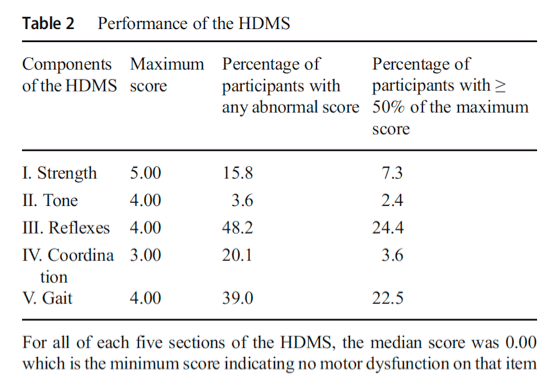
In terms of demographic variables, the HDMS (rs = 0.300, P < 0.01) was moderately positively correlated with age, but not with ethnicity or race. Participants who reported a clinical history of cerebrovascular disease had higher HDMS scores (median of 3.25 vs. 1.0, P = 0.002). The other comorbidities and HIV-related factors (CD4+ nadir, absolute CD4+ count at entry, HIV-1 plasma viral load, current use of CART) were not associated with differences in the distribution of HDMS scores.
As shown in Fig. 1, median HDMS scores were low for those participants with normal cognitive function or ANI (0.5 and 0.0, respectively), moderate in participants with MND (2.00) and NPI-O (2.00), and highest in participants with HAD (4.5). Overall, 66% of participants with HAND (including ANI, MND, HAD, and NPI-O) had some degree of motor dysfunction, compared to 56% of those with normal cognition.
|
|
| |
| |
|
|
|