| |
Switch to Doravirine 2 Studies, DRIVE-SHIFT
Real-Life Safety of Doravirine in Treatment-Experienced, Virologically Suppressed PLWHIV
|
| |
| |
Download the PDF here
Download the PDF here
"Interestingly, we observed significant reduction in serum creatinine after 24 weeks; this finding has not been reported in the results of the DRIVE-SHIFT trial. Only a minimal percentage of DOR is eliminated through the renal pathways;5 hence, the drug should have a very light effect on renal biomarkers."
Letters to the Editor
JAIDS Sept 1 2021 - Arturo Ciccullo, MDaAnna D'Angelillo, MDbValentina Iannone, MDbDamiano Farinacci, MDbFrancesca Lombardi, PhDbElena Visconti, MDcEnrica Tamburrini, MDb,cSimona Di Giambenedetto, MDb,caInfectious Diseases Unit, San SalvatoreHospital, L'Aquila, ItalybDepartment of Safety and Bioethics,Catholic University of the Sacred Heart,Rome, ItalycInfectious Diseases Unit, FondazionePoliclinico Universitario Agostino GemelliIRCCS, Rome, Italy
To the Editors:
We read with interest the recently published results from the DRIVE-SHIFT trial,1 whose results confirm the efficacy and safety of doravirine (DOR), in association with a 2-nucleoside reverse transcriptase inhibitor (NRTI) backbone, in virologically suppressed people living with HIV (PLWHIV). However, as we have learned in the past, results from clinical trials do not always translate directly to the "real-life" setting.
In our clinical center, we aimed to assess the safety and tolerability of DOR in clinical practice by analyzing all treatment-experienced, virologically suppressed PLWHIV starting a DOR-based treatment in our center. All patients started a 3-drug regimen with DOR and a tenofovir-based (TAF or TDF) NRTI backbone. The primary study objective was to evaluate time to treatment discontinuation (TD, defined as the interruption of DOR at any point during follow-up). Survival analysis was used to determine time to TD, whereas predictors were analyzed by Cox regression. We also collected results from previous genotypic tests, previous virological failures (VFs, defined as a single HIV-RNA determination ≥1000 copies/mL or 2 consecutive determinations ≥50 copies/mL), and immunological and metabolic parameters and compared them using parametric and nonparametric tests, as appropriate.
We analyzed data from 33 patients; 22 (66.7%) were men, with a median age of 52.9 years [interquartile range (IQR) 46.5-59.5]. The median time of HIV infection was 11.3 years (IQR 4.9-21.8), median time of antiretroviral exposure was 11.1 years (IQR 4.8-18.4), and 8 (24.2%) patients had experienced at least one previous VF. The characteristics of patients are summarized in Table 1.
During the 167.1 patient-month of follow-up, we registered 3 TD, a rate of 1.8 per 100 patient-month of follow-up. Reasons for discontinuation were as follows: 1 case of VF and 2 cases of gastrointestinal toxicity. The patient with VF had no known nonnucleoside reverse transcriptase inhibitor (NNRTI) mutation at baseline and showed no acquired mutations in a subsequent genotypic test. Failure was observed after 6 months from DOR initiation, with the patient reporting poor adherence to antiretroviral therapy as the probable reason for VF. In our survival analysis, estimated probability of maintaining DOR was 93.8% (SD ± 0.6) after 6 months and 80.4% (SD ± 1.3) after 12 months.
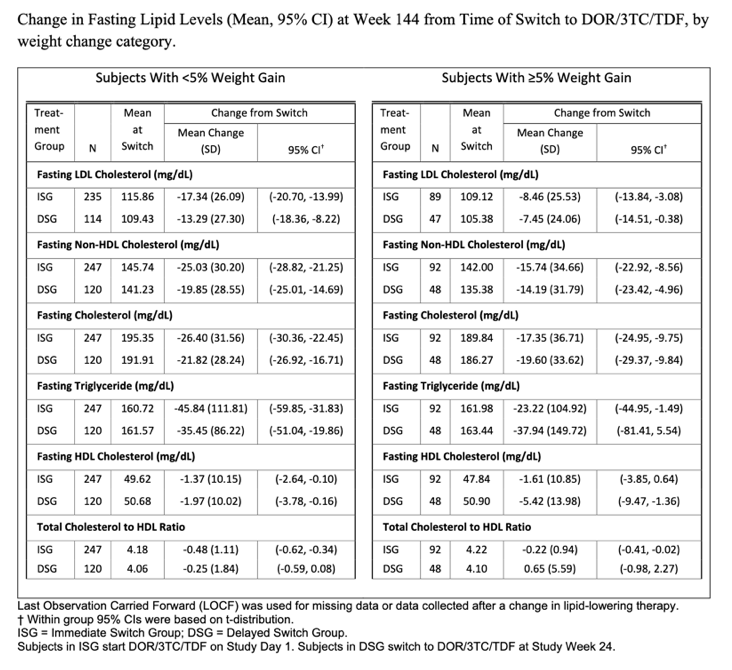
Regarding metabolic parameters, we registered a significant decrease in serum creatinine after 3 months of observation (median change 0.07 mg/dL, P = 0.028), with no correlation with previous antiretroviral exposure or to baseline creatinine. As to fasting lipids, we did register a decreasing trend for triglycerides after 6 months although not significant (median change -59 mg/dL, P = 0.063).
Five (15.2%) patients presented a resistance mutation to NNRTIs at baseline: 3 had the E138A mutation, one had the K103N mutation, and, most notably, one patient presented the Y188L mutation in a previous genotypic analysis. None of these patients incurred VF or viral blips during follow-up.
DOR is a novel NNRTI recommended for the treatment of both treatment-naïve and virologically suppressed PLWHIV.2 In our real-life scenario, DOR-based regimens demonstrated optimal tolerability and safety in the short-term. Data from patients with previous resistance mutations to NNRTIs are particularly reassuring because none of them experienced any viral blip. These preliminary data seem to confirm the genetic barrier of DOR, especially when compared with previous NNRTIs.3,4
In line with results from the DRIVE-SHIFT study,1 a trend toward reduction in fasting lipids was observed at 24 weeks from the switch. Interestingly, we observed significant reduction in serum creatinine after 24 weeks; this finding has not been reported in the results of the DRIVE-SHIFT trial. Only a minimal percentage of DOR is eliminated through the renal pathways;5 hence, the drug should have a very light effect on renal biomarkers. In conclusion, although other studies with ample sample size and an adequate follow-up time are needed, our preliminary results show that DOR-based 3-drug regimens are a safe and tolerable choice for the treatment of virologically suppressed PLWHIV.
-----------------------------
Brief Report: Switching to DOR/3TC/TDF Maintains HIV-1 Virologic Suppression Through Week 144 in the DRIVE-SHIFT Trial
JAIDS June 1 2021
Abstract
Background:
In the primary analysis of the DRIVE-SHIFT trial, switching to doravirine/lamivudine/tenofovir disoproxil fumarate (DOR/3TC/TDF) maintained suppression of HIV-1 through week 48. Here, we present long-term efficacy and safety outcomes through week 144 of the DRIVE-SHIFT trial.
Methods:
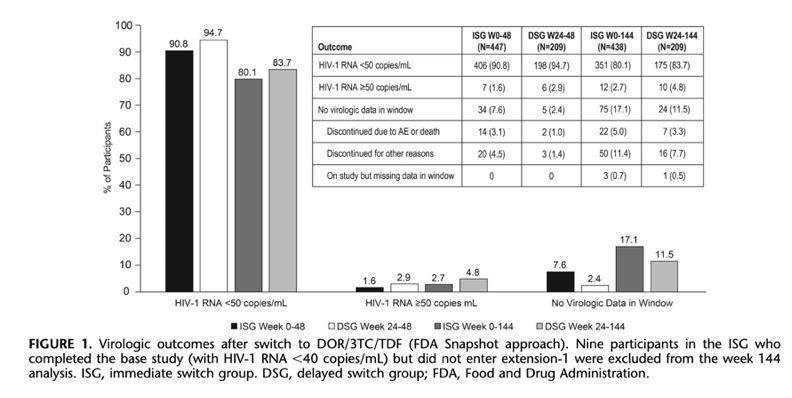
This phase 3, randomized, open-label trial evaluated switching from a stable antiretroviral regimen to once-daily DOR/3TC/TDF in adults with HIV-1 suppressed for ≥6 months and no previous virologic failure. Participants switched at day 1 [immediate switch group (ISG); n = 447] or week 24 [delayed switch group (DSG); n = 209]. Nine ISG participants who completed week 48 but did not enter extension-1 were excluded from week 144 efficacy analyses.
Results:
At week 144, HIV-1 RNA <50 copies/mL was maintained in 80.1% of the ISG (351/438) and 83.7% of the DSG (175/209), while 2.7% (12/438) and 4.8% (10/209), respectively, had HIV-1 RNA ≥50 copies/mL (Food and Drug Administration Snapshot approach). Protocol-defined virologic failure after switch occurred in 2.1% of ISG (9/438) and 3.3% of DSG (7/209); no viral resistance to doravirine was detected in 4 participants with samples available.
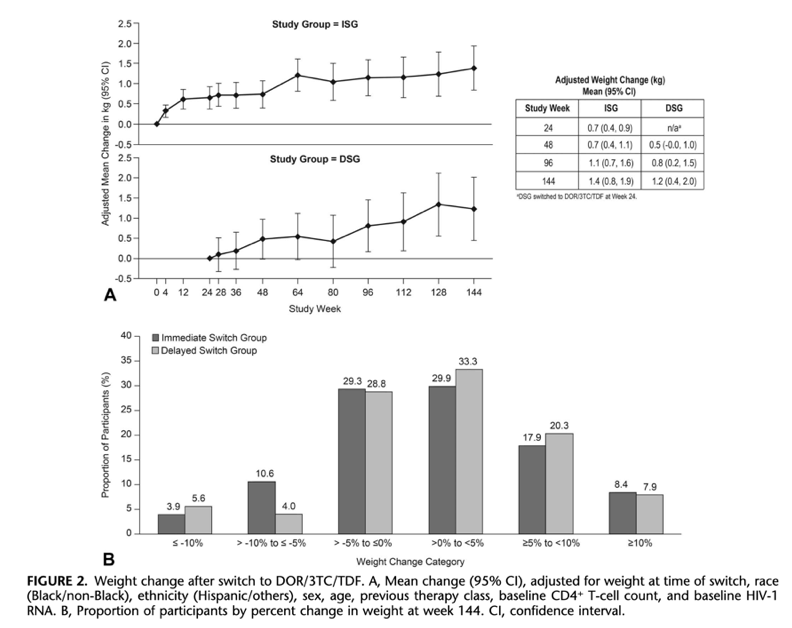
Reductions in fasting lipids were observed at 24 weeks after switch and maintained through week 144. The mean weight change from switch to week 144 was +1.4 kg for ISG and +1.2 kg for DSG. The most common adverse events were nasopharyngitis (16.2%), headache (12.3%), and diarrhea (9.1%). Overall, 4.1% discontinued because of adverse events, and no deaths occurred.
Conclusions:
These results confirm that switching to once-daily DOR/3TC/TDF is a generally well-tolerated option for maintaining viral suppression in adults considering a change in therapy.
|
|
| |
| |
|
|
|