| |
Immunogenicity and safety of the BNT162b2 mRNA Covid-19 vaccine in people living with HIV-1
|
| |
| |
Download the PDF here
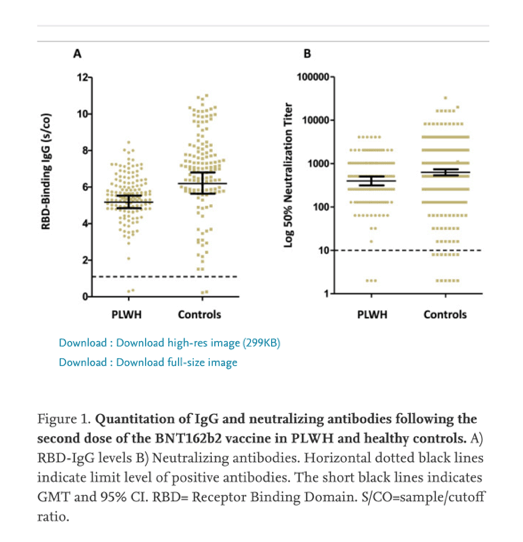
"Following the second dose, immune sera neutralized SARS-CoV-2 pseudo-virus in 97% and 98% of PLWH and HCW, respectively."
"Only four patients did not develop NAs, specifically, a 66-year-old man with a kidney transplant treated with mycophenolate, tacrolimus and prednisone, a 58-year-old man on hemodialysis, a 72-year-old man with ischemic heart disease, and a 64-year-old woman with nonspecific arthritis who was treated with colchicine and developed severe COVID-19 four weeks after the second dose of the vaccine.
In our cohort, only three patients had a current CD4 count of <200 cells per μL, but all developed high levels of RBD -IgG antibodies and NAs in response to vaccination.
In our study, the BNT162b2 vaccine was found to be immunogenic and safe in PLWH. However our study population included stable and older HIV-1 positive subjects, mostly with longstanding HIV, treated with integrase inhibitor, have undetectable viral load and current high CD4 + cell count.
Importantly, although it seems that PLWH may have lower RBD IgG antibody levels compared to immunocompetent HCWs, 97-98% of PLWH did develop antibodies after second vaccination and their neutralizing activity was similar to the control.
Although we still do not know the significance of the small discrepancy between RBD IgG levels in PLWH relatively to controls, most of the PLWH developed neutralizing activity similar to controls, and this serves better as a correlate of immunity than total anti RBD IgG antibodies4. Nevertheless, in our study there was a high correlation between RBD IgG antibodies and NAs.
In our study, HIV-1 viral load increased in 3 subjects from undetectable (<40 copies/mL) to LLV (<100 copies/ml) immediately after vaccination. These patients had nadir CD4+ counts of < 200 cells per μL and also had viral failures, which may imply an increased reservoir. These "blips"12are usually not considered as viral failure, and are not rare among PLWH, occurring in about a quarter of patients on stable ART with undetectable viral loads.13 A transient increase in HIV-1 viral load was also detected two weeks following influenza vaccination in well-controlled patients with HIV14; that study found a concomitant decrease in proviral DNA and memory phenotype CD4+ cells and claimed that the elevated viral load could suggest mobilization of a latent reservoir."
-----------------------------------
Immunogenicity and safety of the BNT162b2 mRNA Covid-19 vaccine in people living with HIV-1
Clinical Microbiology and Infection Aug 24 2021 - Itzchak Levy, Anat Wieder - Finesod, Vladyslav Litchevsky, Asaf Biber, VictoriaIndenbaum, Liraz Olmer, Amit Huppert, Orna Mor, May Goldstein, Einav Sapir,Tammy Hod, Carmit Cohen, Yaniv Lustig, Galia Rahav
Abstract
Objectives
The immunogenicity and safety of the Pfizer-BioNTech BNT162b2 mRNA vaccine in people living with HIV-1 (PLWH) are unknown. We thus aimed to assess the immunogenicity and safety of this vaccine in PLWH.
Methods
In this prospective open study, we enrolled 143 PLWH, aged ≥18 years, who attended our clinic and 261 immunocompetent health care workers (HCWs). SARS-CoV-2 receptor binding domain (RBD) IgG and neutralizing antibodies were measured. Adverse events, viral load and CD4 cell counts were monitored.
Results
At a median of 18 days (IQR 14-21) after the second dose, anti-RBD IgG was positive in 139/141 (98%) PLWH. Among HCWs, 258/261 (98.9%) developed anti-RBD IgG at a median of 26 (IQR 24-27) days after the second dose. Following the second dose, immune sera neutralized SARS-CoV-2 pseudo-virus in 97% and 98% of PLWH and HCW, respectively. Adverse events were reported in 60% of PLWH, mainly pain at the injection site, fatigue, and headache. AIDS-related adverse events were not reported. HIV viral load increased in 3/143 (2%) patients from < 40 copies/mL to ≤ 100 copies/mL. CD4+ T cell count decreased from a geometric mean of 700 (95% CI 648-757) cells per μL to 633.8 (95% CI 588-683) cells per μL (P<0.01).
Conclusions
BNT162b2 mRNA vaccine appears immunogenic and safe in PLWH who are on ART with unsuppressed CD4 count and suppressed viral load.
Introduction
The Pfizer-BioNTech BNT162b2 mRNA vaccine has been tested for safety and efficacy in a multinational randomized placebo controlled trial with more than 40,000 participants.1 In that trial, 196 immunologically stable HIV-1 positive patients were included, but data for the safety and immunogenicity of the vaccine specifically for that patient group have not been published. Indeed, to the best of our knowledge, there are no data available for the response of people living with HIV (PLWH) to mRNA vaccines.
The BNT162b2 mRNA vaccine was introduced in Israel on December 19th, 2020, and our program to vaccinate PLWH started a day later. Since the initiation of the vaccination program in Israel, more than 52% aged 16 and over have been vaccinated.
In this study, we examined the immunogenicity and safety of the BNT162b2 mRNA vaccine in 143 PLWH.
Discussion
In our study, the BNT162b2 vaccine was found to be immunogenic and safe in PLWH. However our study population included stable and older HIV-1 positive subjects, mostly with longstanding HIV, treated with integrase inhibitor, have undetectable viral load and current high CD4 + cell count.
Importantly, although it seems that PLWH may have lower RBD IgG antibody levels compared to immunocompetent HCWs, 97-98% of PLWH did develop antibodies after second vaccination and their neutralizing activity was similar to the control.
Although we still do not know the significance of the small discrepancy between RBD IgG levels in PLWH relatively to controls, most of the PLWH developed neutralizing activity similar to controls, and this serves better as a correlate of immunity than total anti RBD IgG antibodies4. Nevertheless, in our study there was a high correlation between RBD IgG antibodies and NAs.
For HIV patients, it is imported to collect data on immunogenicity because the cell-mediated and B-cell immune abnormalities that occur as HIV infection advances may reduce the magnitude of the response and the durability of the protection. Nonetheless, several studies have demonstrated the protective benefit of vaccinations against influenza5,6 and Streptococcus pneumonia,7,8 even in advanced HIV patients. Although efficacy data are sparse for other types of vaccine, studies using surrogate endpoints (most commonly post-vaccination antibody levels) have shown that most HIV patients do generate antibody responses post vaccination.
In our cohort, only three patients had a current CD4 count of <200 cells per μL, but all developed high levels of RBD -IgG antibodies and NAs in response to vaccination.
We found a statistically significant decrease in CD4+ T cell count between baseline levels and those measured following the first and second vaccines, as well as four months after the second vaccination. This drop was not associated with any clinical signs or symptoms, but it should be further monitored. It should be noted that the study was conducted among PLWH who were stable on ART with unsuppressed CD4 count and suppressed VL. A drop in CD4 count may be deleterious for people whose CD4 count is already low, and may not recover as rapidly in people who are not stable on ART.
A similar drop in CD4+ T cell count was not reported in other studies on different types of vaccinations; for example, two studies that examined CD 4+ T cell dynamics in PLWH receiving the 7-valent pneumococcal conjugate vaccine (PCV-7) did not find significant changes in CD4+ count in response to the vaccine at 6 months9 and 3-4 months10 post vaccination. In addition, a large study evaluating influenza immunizations in over 30,000 HIV patients found no long-term negative effects on CD4 counts, HIV RNA levels, or progression to AIDS or death.11
In our study, HIV-1 viral load increased in 3 subjects from undetectable (<40 copies/mL) to LLV (<100 copies/ml) immediately after vaccination. These patients had nadir CD4+ counts of < 200 cells per μL and also had viral failures, which may imply an increased reservoir. These "blips"12 are usually not considered as viral failure, and are not rare among PLWH, occurring in about a quarter of patients on stable ART with undetectable viral loads.13 A transient increase in HIV-1 viral load was also detected two weeks following influenza vaccination in well-controlled patients with HIV14; that study found a concomitant decrease in proviral DNA and memory phenotype CD4+ cells and claimed that the elevated viral load could suggest mobilization of a latent reservoir.
We found that the BNT162b2 mRNA Covid-19 vaccine was safe. None of the patients developed an immediate or delayed type hypersensitivity reaction. 40.6% and 25.6% developed local following the first and second doses, respectively, but in most cases they were mild to moderate and subsided after 24-48 hours. Although safety data was not collected for controls at the time that the study was conducted, the rate of adverse events that we found among PLWH was lower than that reported for the Pfizer phase 2/3 trial, with the difference probably being due to the different way in which adverse events were monitored.
The main limitations of this study include lack of appropriated control, small size and limited follow up.
Matched case control study in which PLWH were matched to HCWs according to the exposure was not feasible due to differences in age and sex. Since the blood in the PLWH and the controls was drawn for serologic response at different time points in windows that do not overlap we have adjusted the groups in multivariable linear regression not only for age, sex and comorbidities but also for timing of serology from the second vaccine dose. It should be also stated that in former studies age and sex had a limited effect on antibody production15.
The small size and limited follow up do not enable us to check for efficacy of the vaccine in PLWH.
In conclusion, this prospective study thus demonstrates the immunogenicity and safety of the BNT162b2 mRNA vaccine in a stable cohort PLWH with a preserved immune system.
|
|
| |
| |
|
|
|