| |
Renal impairment in a large-scale HIV pre-exposure prophylaxis implementation cohort
|
| |
| |
Download the PDF here
AIDS July 22 2021 - Douglas DRAK1,2, Hamish MCMANUS3, Tobias VICKERS3, Jack E HERON4, Stefanie VACCHER3, Iryna ZABLOTSKA5,6,7, Rebecca GUY3, Benjamin BAVINGTON3, Fengyi JIN3, Andrew E GRULICH3, Mark BLOCH8, Catherine C O’CONNOR1,3,4, David M GRACEY1,4, for the Expanded PrEP Implementation in Communities New South Wales
(EPIC-NSW) research group
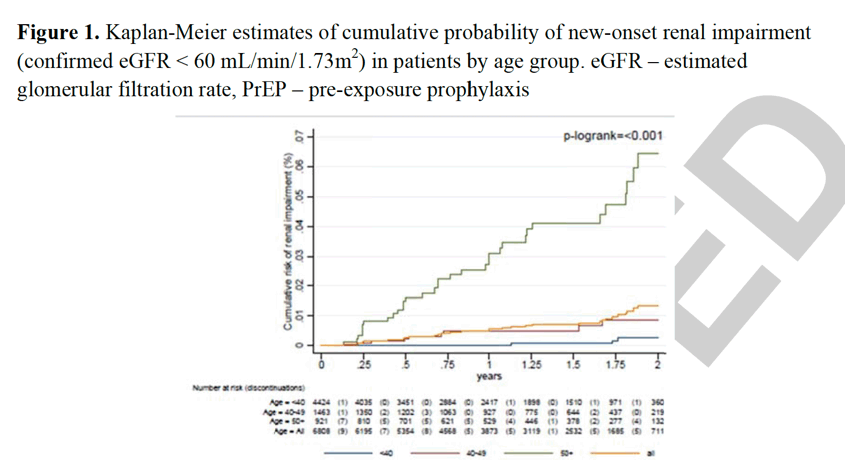
Data from 6808 participants were eligible for inclusion in this analysis of renal function, with 7874 person-years of follow-up and a median follow-up time of 1.2 years (IQR: 0.6 to 1.7). There were 46 instances of new onset renal impairment over the study period, corresponding to a rate of 5.8 instances per 1000 person-years (95%CI: 4.4 to 7.8). Rates of renal impairment are shown by participant subgroup in Table 2. Confirmatory testing occurred in a median of 65 days (IQR: 28 to 91) after renal impairment was first detected. The cumulative risk of renal impairment increased in a roughly linear manner (Figure 1) and, at two years of follow-up, was 1.34% (95%CI: 0.95 to 1.89). Participants that were ≥ 50 years of age at enrolment, however, had more than a four-fold higher cumulative risk of developing renal impairment at two years than younger participants (6.46%, 95%CI: 4.4 to 9.4, p<0.001). Similar trends were seen in the sensitivity analysis where renal impairment also included patients with a single eGFR measure < 60 mL/min/1.73m2 and were either lost to follow-up or censored (Appendix A, http://links.lww.com/QAD/C250).
6808 participants were eligible for inclusion. Almost all were male (99%), with a median age of 35 years (IQR: 28 to 44). Approximately one-quarter (26%) had a baseline eGFR < 90 mL/min/1.73m2. Over a median follow-up period of 1.2 years (IQR 0.6 to 1.7), the rate of renal impairment was 5.8 episodes per 1000 person years (95%CI: 4.0 to 7.8). In multivariable Cox regression, there was higher risk of renal impairment in participants aged ≥50 years (HR 14.7, 95%CI: 5.0 to 43.3, p< 0.001) and those with an eGFR < 90 mL/min/1.73m2 (HR 28.9, 95%CI: 6.9 to 121.9) at baseline.
Discussion
In this retrospective post-hoc analysis of 6,808 EPIC-NSW study participants, there were 46 instances of new onset renal impairment during 7,874 person-years of follow-up, corresponding to a rate of 5.6 instances per 1,000 person-years. The risk of renal impairment was substantially higher in patients with pre-existing renal dysfunction and older patients, particularly those aged 50 or older.
Conclusion
In EPIC-NSW, a large-scale PrEP implementation study, the incidence of renal impairment was low, but substantially higher than reported in previous clinical trials. New onset renal impairment in our study was largely confined to patients with pre-existing renal dysfunction and/or those aged 50 years or older, suggesting that daily dosing of TDF-containing PrEP in young, healthy patients is safe from a renal perspective. For those with risk factors, more frequent clinical monitoring may be warranted.
Compared to those < 40 years of age, participants aged 40 - 49 or ≥ 50 years at study enrolment had a 4-fold and 14-fold high risk of developing renal impairment during the study period respectively. A baseline eGFR < 90 mL/min/1.73m2 was independently associated with 29-fold increased risk of renal impairment. Age and pre-existing renal dysfunction have been consistently identified as risk factors for renal impairment across PrEP studies with the relative risk/odds ratios ranging from less than ten-fold increases in implementation studies from the United States to over 30-fold in the iPrEx open label extension.[7, 9-11]
The higher rate of renal impairment in our study, as compared to previous PrEP studies, is likely explained by a greater proportion of participants with these risk factors. In the iPrEx open label extension, for example, only 8.6% of participants were aged 50 or older and 8.5% had pre-existing renal impairment. This compares to 14% aged over 50 and 29% with pre-existing renal impairment in our study. Higher prevalence of these risk factors appears to be a common feature of PrEP implementation studies,[7, 10, 22] suggesting that the “real world” incidence of TDF-induced renal impairment would be greater than that reported in earlier randomised controlled trials and their open label extensions.[5, 6, 9, 21] Our findings support current recommendations that patients with these risk factors should receive more frequent monitoring of their renal function and that clinicians could consider alternatives to daily dosing of TDF-containing PrEP.[12, 13]
Intermittent dosing of TDF-containing PrEP has been shown to be similarly efficacious to daily regimens and has the theoretical benefit of reduced renal toxicity from lower TDF exposure. this benefit remains to be convincingly demonstrated by trial data, however.[1] In IPERAGY, while the risk of serious adverse renal events was similar between the intermittent dosing and placebo groups, those on PrEP were sill at significantly increased risk of creatinine elevations.[21] Head-to-head data comparing daily-dosing and intermittent PrEP regimens are limited,[25] and further study, particularly in relation to high-risk groups, may be warranted.
TAF, a novel prodrug of the active metabolite tenofovir, may represent a safer alternative for patients at risk of TDF-induced nephrotoxicity. TAF appears to be associated with lower rates of renal impairment among people living with HIV.[26] DISCOVER, the first trial comparing TDF to TAF for PrEP is ongoing, with early results suggesting lower rates of renal and bone toxicity.[16] The lower cost of generic TDF is likely to justify its continued use in lower risk groups.
Markers of renal proximal tubular dysfunction, including proteinuria and urinary phosphate excretion, were not measured in this study. TDF exposure is associated with proteinuria among people living with HIV,[26] however, the association is less robust when TDF is used for PrEP. In Partners PrEP and iPrEx, two large placebo-controlled studies, TDF exposure was associated with proteinuria in the former, but not the latter trial.[5, 27] Although, tubular proteinuria was noted in the iPrEx open label extension study.[27] New onset proteinuria during PrEP, however, does not appear to increase the risk of new onset renal impairment.[5,
11]
Data from 6808 participants were eligible for inclusion in this analysis of renal function, with 7874 person-years of follow-up and a median follow-up time of 1.2 years (IQR: 0.6 to 1.7). There were 46 instances of new onset renal impairment over the study period, corresponding to a rate of 5.8 instances per 1000 person-years (95%CI: 4.4 to 7.8). Rates of renal impairment are shown by participant subgroup in Table 2. Confirmatory testing occurred in a median of 65 days (IQR: 28 to 91) after renal impairment was first detected. The cumulative risk of renal impairment increased in a roughly linear manner (Figure 1) and, at two years of follow-up, was 1.34% (95%CI: 0.95 to 1.89). Participants that were ≥ 50 years of age at enrolment, however, had more than a four-fold higher cumulative risk of developing renal impairment at two years than younger participants (6.46%, 95%CI: 4.4 to 9.4, p<0.001). Similar trends were seen in the sensitivity analysis where renal impairment also included patients with a single eGFR measure < 60 mL/min/1.73m2 and were either lost to follow-up or censored (Appendix A, http://links.lww.com/QAD/C250).
New onset renal impairment was defined as an eGFR < 60 mL/min/1.73m2. To account for spurious measures, this needed to be confirmed by the average of that measurement and next also being < 60 mL/min/1.73m2. In contrast, pre-existing renal dysfunction refers to a baseline eGFR < 90 mL/min/1.73m2. A further subgroup analysis of patients with a baseline eGFR < 90 mL/min/1.73m2 was performed, where renal impairment was alternatively defined as a confirmed eGFR decline to < 60 mL/min/1.73m2 or a decrease of > 25% from baseline.
Adherence to PrEP, measured by medication possession ratio, was high. Patients were dispensed PrEP at three-monthly study visits. Of the intervals between these visits, 89.2% had a medication possession ratio associated with a protective level of TDF, equivalent to four tablets per week.[19] There was no PrEP coverage, however, in 10.3% of these intervals due to missed dispensing visits. Delayed study visits resulted in 0.5% of intervals having a medication possession ratio that was less than protective.
In bivariate Cox proportional hazards regressions (Table 2), baseline eGFR < 90 mL/min/1.73m2 (p<0.001), age 40 - 49 years (p=0.002) and age ≥ 50 years (p<0.001) were associated with a higher risk of renal impairment. No such association was found for recent methamphetamine use (p=0.403), positive hepatitis C serology (p=0.553) or previous PrEP use (p=0.654). As compared to low MPR, there was no increased risk associated with either medium (p=0.180) or high (p=0.077) MPR.
In a multivariable model, there was a substantially higher risk of renal impairment in participants aged ≥ 50 years (HR 14.7, 95%CI: 5.0 to 43.3, p<0.001) and those with a baseline eGFR < 90 mL/min/1.73m2 (HR 28.9, 95%CI: 6.9 to 121.9, p<0.001).
A subgroup analysis was performed on participants with baseline eGFR < 90 mL/min/1.73m2, where renal impairment was a composite outcome of confirmed eGFR < 60 mL/min/1.73m2 or a decrease in eGFR of > 25% from baseline (Figure 2). With this definition, there were 22.0 episodes per 1000 patient years (95%CI: 16.6 to 29.1) of new renal impairment. This equated to a cumulative risk of 5.6% (95%CI 3.3 to 6.3) at two years of follow-up.
----------------------------------
Renal impairment in a large-scale HIV pre-exposure prophylaxis implementation cohort
Abstract
Background:
HIV pre-exposure prophylaxis (PrEP) with fixed-dose tenofovir disoproxil fumarate (TDF) and emtricitabine has been associated with low rates of renal impairment in clinical trials. Large-scale PrEP implementation may result in higher rates, as the prevalence of associated risk factors may be higher than in trial populations.
Methods:
A post-hoc analysis of EPIC-NSW, a large Australian multi-centre PrEP implementation trial for patients at high-risk of HIV infection. Participants were eligible for inclusion if they commenced PrEP between 1 March 2016 and of 30 April 2018, and had renal function assessed at baseline and at least once more before the censor date. The primary outcome was new onset renal impairment, defined as an estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73m2.
Results:
6808 participants were eligible for inclusion. Almost all were male (99%), with a median age of 35 years (IQR: 28 to 44). Approximately one-quarter (26%) had a baseline eGFR < 90 mL/min/1.73m2. Over a median follow-up period of 1.2 years (IQR 0.6 to 1.7), the rate of renal impairment was 5.8 episodes per 1000 person years (95%CI: 4.0 to 7.8). In multivariable Cox regression, there was higher risk of renal impairment in participants aged ≥50 years (HR 14.7, 95%CI: 5.0 to 43.3, p< 0.001) and those with an eGFR < 90 mL/min/1.73m2 (HR 28.9, 95%CI: 6.9 to 121.9) at baseline.
Conclusion:
In a large-scale implementation study, TDF-containing PrEP was associated with a low risk of renal impairment overall, while older patients and those with pre-existing renal dysfunction were at substantially increased risk.
Baseline characteristics of study participants are detailed in Table 1. Almost all were male
(99%) and resided in major cities (95%). Median age was 35 years (IQR 28 to 44), with 14% of the cohort being 50 years of age or older at enrolment. At baseline, nearly one-third of the cohort had an eGFR < 90 mL/min/1.73m2 (27%) and nine percent had previous exposure to PrEP. Use of methamphetamine within the last three months was reported by one-fifth of participants (19%) and positive hepatitis C virus serology was noted in one percent.
There were 46 instances of new onset renal impairment over the study period, corresponding to a rate of 5.8 instances per 1000 person-years (95%CI: 4.4 to 7.8). Rates of renal impairment are shown by participant subgroup in Table 2. Confirmatory testing occurred in a median of 65 days (IQR: 28 to 91) after renal impairment was first detected. The cumulative risk of renal impairment increased in a roughly linear manner (Figure 1) and, at two years of follow-up, was 1.34% (95%CI: 0.95 to 1.89). Participants that were ≥ 50 years of age at enrolment, however, had more than a four-fold higher cumulative risk of developing renal impairment at two years than younger participants (6.46%, 95%CI: 4.4 to 9.4, p<0.001). Similar trends were seen in the sensitivity analysis where renal impairment also included patients with a single eGFR measure < 60 mL/min/1.73m2 and were either lost to follow-up or censored (Appendix A, http://links.lww.com/QAD/C250).
Introduction
Pre-exposure prophylaxis (PrEP) with fixed-dose tenofovir disoproxil fumarate (TDF) and emtricitabine (FTC) has been shown to be efficacious in preventing HIV infection in clinical trials and, more recently, to be effective at a population level in the large-scale EPIC-NSW implementation study.[1-4] While TDF is generally well tolerated, it is a potential nephrotoxin.
The use of TDF for PrEP is associated with a small and generally non-progressive reduction in estimated glomerular filtration rate (eGFR), with a minority of patients developing clinically significant renal impairment.[5-7] Earlier placebo-controlled trials and subsequent implementation studies have differed, however, in the reported incidence of this TDF-associated renal impairment. A meta-analysis of the former found the risk of creatinine elevations no greater in PrEP patients than those receiving placebo.[8] In contrast, implementation studies have generally reported between two to ten-fold higher rates of renal impairment than in randomised trials, depending on how renal impairment was defined.[5, 7, 9-
11]
These differences in the risk of renal impairment may be due to the placebo-controlled studies, and their open label extensions, having included fewer older patients and/or patients with pre-existing renal dysfunction as compared to later implementation studies, two patient groups being known to be at increased risk of TDF-induced renal impairment.[7, 9-11] The implementation studies have tended to be smaller than the earlier placebo-controlled trials, however.[7, 8, 10] The incidence of renal impairment in “real world” PrEP populations therefore may not be well characterised.
Despite the potential for nephrotoxicity, daily dosing with TDF-containing PrEP is currently recommended by major international societies for HIV prevention and uptake of TDF-containing PrEP is increasing.[12-15] Alternatives to daily dosing with TDF-containing PrEP exist, including event driven PrEP, which offers reduced TDF exposure, and the novel prodrug tenofovir alafenamide (TAF), with less renal toxicity.[16]
With the continued uptake of PrEP and the emergence of alternatives to daily TDF dosing, a more precise estimation of the risk of renal impairment in a “real world” population would aid in clinical decision making. Here we report the incidence and predictors of new onset renal impairment in patients taking once daily PrEP for HIV prevention in EPIC-NSW, a prospective large-scale PrEP implementation study in Australia.
|
|
| |
| |
|
|
|