 |
 |
 |
| |
Chronic morphine administration alters gut-brain homeostasis in SHIV infected rhesus macaques
|
| |
| |
".....morphine abuse results in remarkable neuropathogenesis characterized by neuronal dysfunction/degeneration..."
IAS 2021 July 18-21
Presenter
Omalla Allan Olwenyi
Authors
O. Allan Olwenyi * (1,2), S. D Johnson (1,2), M. Bidokhti (1), A. Acharya (1), S. Uppada (1), K. Pandey (1,2), M. Thurman (1), S. Callen (1), U. Ranga (3), S. J. Buch (1), S. N. Byrareddy (1)
Institutions
(1) University of Nebraska Medical Center, Pharmacology and Experimental Neuroscience, Omaha, United States, (2) University of Nebraska Medical Center, Pathology, Microbiology and Infectious Diseases, Omaha, United States, (3) Jawaharlal Nehru Center for Advanced Scientific Research, HIV-AIDS Laboratory, Molecular Biology and Genetics Unit, Bangalore, India
BACKGROUND: Commonly used opioids such as morphine have been implicated in augmented central nervous system (CNS) pathology. Recently, we have shown that morphine increases replication-competent viral reservoirs in CNS-microglia. However, the extent of myeloid cell polarization and viral persistence in different brain regions remains unclear. To address this, we profiled myeloid cell polarization and viral persistence within diverse brain regions and evaluated the role of morphine dysregulation of gut-brain systems crosstalk.
METHODS: Eight rhesus macaques were ramped-up, and a daily injection of either morphine (n=4) or saline (n=4) for a total of 9 weeks and infected with SHIVAD8EO variants. During necropsy, isolated mononuclear cells from different regions of the brain (frontal lobe, cerebellum, medulla, putamen, hippocampus (HIP) and subventricular zone (SVZ)) and gut (lamina propria (LP) and muscularis (MUSC) of ascending colon, duodenum, and ileum) were profiled for myeloid cell polarity/ activation. Levels of SHIV DNA were evaluated using the digital droplet PCR assay. Luminex platform to quantify soluble plasma/ CSF biomarkers and changes in the fecal microbiome was investigated using the Illumina NovaSeq platform.
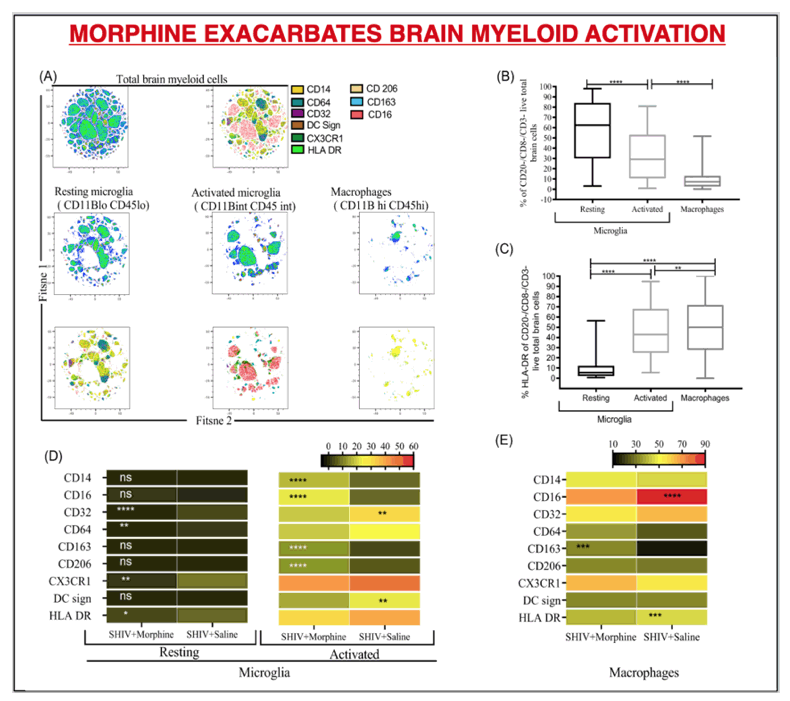
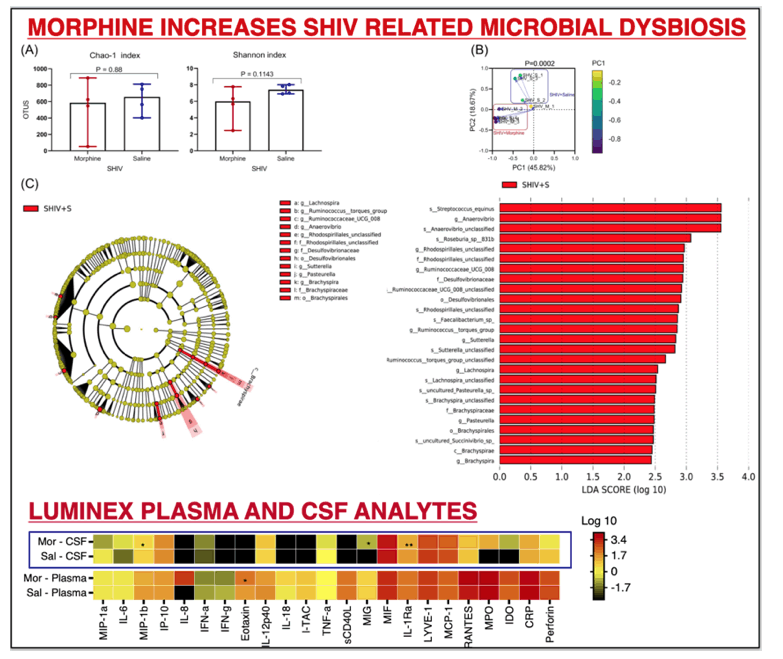
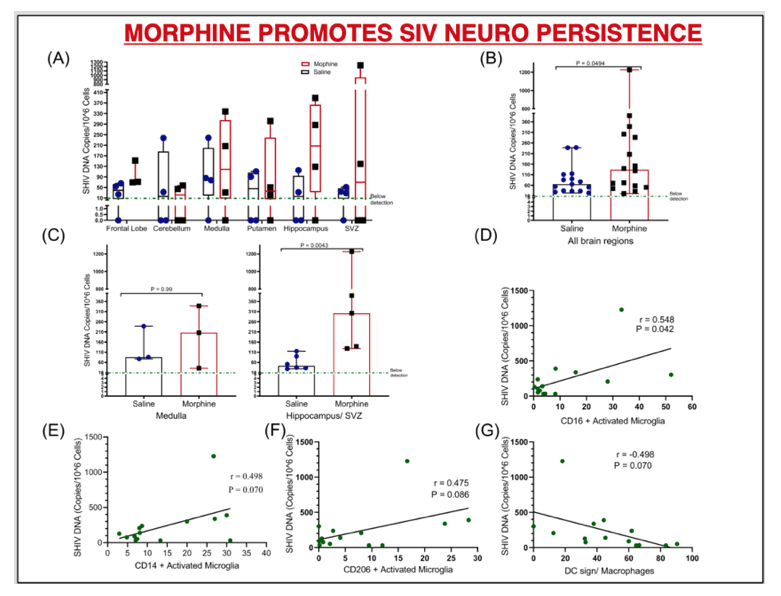
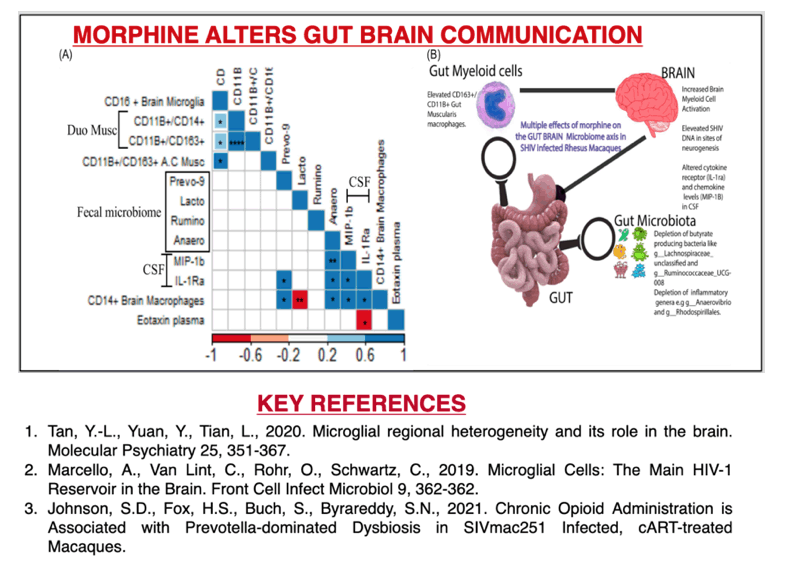
RESULTS: There were no significant differences in the plasma and CSF viral loads between the two groups. Morphine exposure led to exacerbated M1(CD14/ CD16)/ M2 (CD163/ CD206) polarization in activated microglia that spanned across diverse brain regions. This was accompanied by elevated SHIV DNA enriched in sites of neurogenesis (HIP/SVZ). HIP/SVZ CD16+ activated microglia positively correlated with SHIV DNA levels in the brain (r = 0.548, p = 0.042). Simultaneously, morphine depleted butyrate-producing bacteria such as ruminococcus (p = 0.05), lachnospira (p = 0.068) genera and roseburia species (p = 0.008) and enriched CD163+ cells in MUSC macrophages. Morphine also altered the regulation of inflammation in the CNS by reducing levels of CSF IL1Ra.
CONCLUSIONS: These data indicate that morphine 1) elevates CNS inflammation by altering receptor regulation and selective enhancement of SHIV persistence in sites of neurogenesis; 2) depletes crucial genera that support optimal brain function; 3) Increases macrophage activation in sites enriched with enteric neurons (MUSC), warranting further investigation into mechanisms involved in morphine enhanced alternation of the gut-brain axis during SHIV infection.


|
| |
|
 |
 |
|
|