 |
 |
 |
| |
Only 1 More Virologic Failure on CAB + RPV in Weeks 96 to 124 of FLAIR
|
| |
| |
IAS 2021, 11th IAS Conference on HIV Science, July 18-21, 2021
Mark Mascolini
Once-monthly injected cabotegravir plus rilpivirine (CAB + RPV) continued to control HIV in 80% of adults switching from a suppressive pill regimen in a phase 3 trial [1]. FLAIR researchers recorded only 1 new confirmed virologic failure after week 96 and no new drug-related serious adverse events.
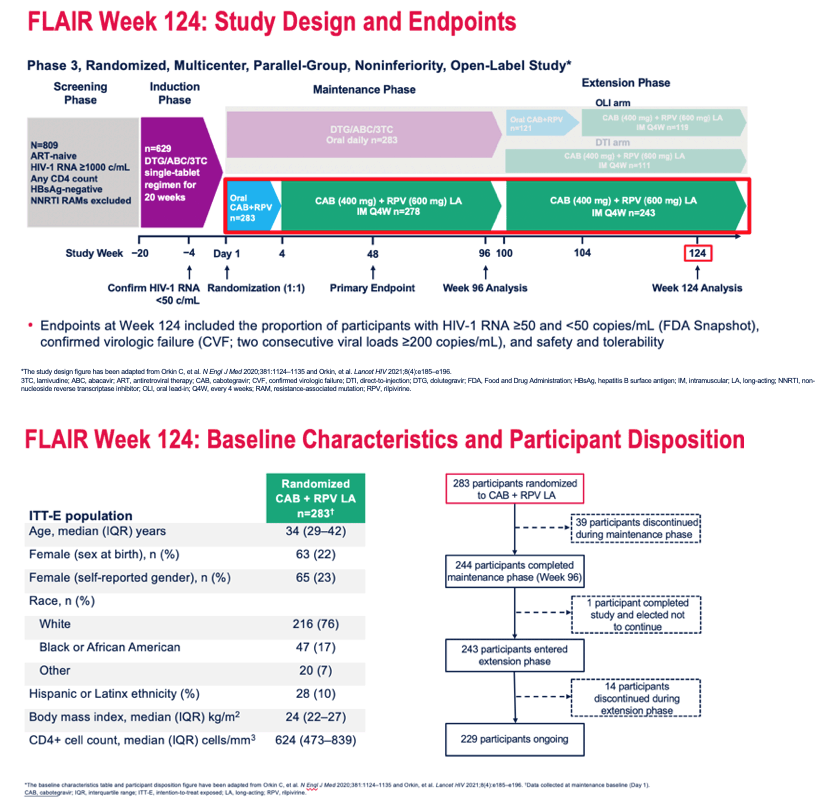
FLAIR, a phase 3 randomized open-label trial [2], helped establish CAB + RPV as a licensed once-monthly injectable regimen (after oral lead-in dosing) to maintain HIV suppression induced by another oral combination. At IAS 2021 FLAIR investigator updated findings from study week 96 through week 124.
The trial began with 809 antiretroviral-naive adults with a viral load at or above 1000 copies and any CD4 count who started oral single-tablet dolutegravir/abacavir/lamivudine. People who reached a confirmed viral load below 50 copies by week 20 got randomized evenly to continue the oral combination (n = 283) or switch to oral CAB + RPV for 4 days followed by injected CAB + RPV every 4 weeks (n = 283). Week-48 results determined that CAB + RPV is virologically noninferior to the continued oral dolutegravir regimen [3].
Of the 283 people randomized to CAB + RPV, median age stood at 34, 23% were women, 76% white, 17% black, and 10% Hispanic. Median body mass index measured 24 kg/m2 and median CD4 count 624. Of the 243 people who entered the extension phase of the trial at week 96, 229 remained in the protocol to this week-124 analysis.
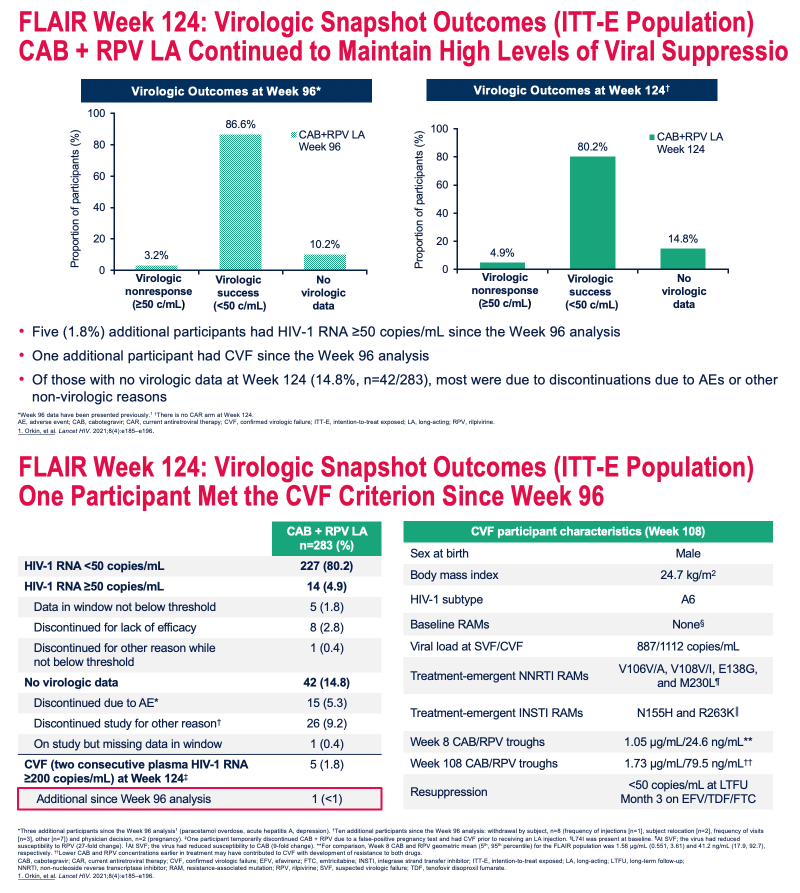
Rates of virologic nonresponse (viral load at or above 50 copies) were 3.2% at week 96 and 4.9% at week 124. Five additional people (1.8%) had virologic nonresponse after week 96, including 1 additional person with confirmed virologic failure. Rates of virologic success (below 50 copies) were 86.6% at week 96 and 80.2% at week 124. Of the 42 people (14.8% of 283) not recorded as below 50 copies at week 124 (13 more participants than at week 96), most failed to make the grade for nonvirologic reasons.
The 1 additional person with confirmed virologic failure (2 confirmed viral loads at or above 200 copies) at week 124 was a man with HIV-1 subtype A6, no pretreatment resistance mutations, and 5 treatment-emergent nonnucleoside or integrase inhibitor mutations at failure. Low CAB and RPV concentrations earlier in treatment may have contributed to virologic failure. This man regained a viral load below 50 copies with efavirenz plus tenofovir/emtricitabine.
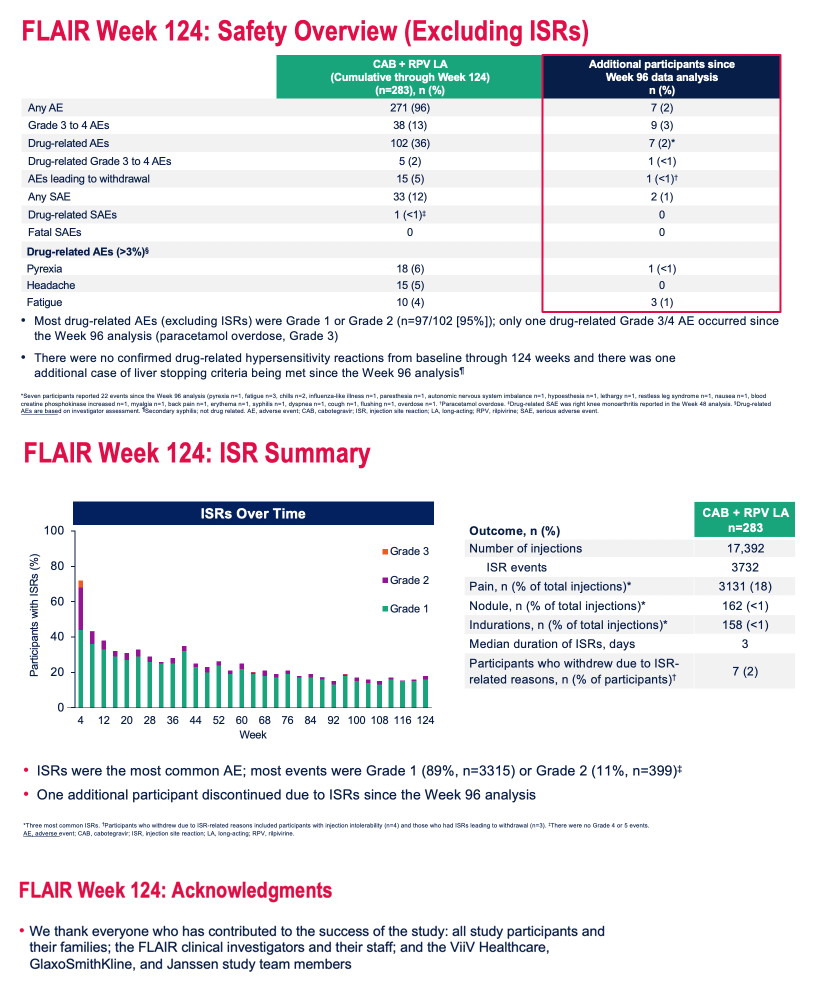
Among 283 people analyzed through week 124, 102 (36%) had any drug-related adverse event, 5 (2%) had any drug-related grade 3 or 4 adverse event, 15 (5%) had any adverse event leading to withdrawal from the study, 33 (12%) had any serious adverse event, and 1 had a drug-related serious adverse event (right knee arthritis at week 48). There were no serious drug-related adverse events after week 96. This adverse event analysis excluded injection site reactions.
Of 17,392 injections through week 124, 3732 (21.5%) caused injection site reactions. Most such reactions were grade 1 (89%) or grade 2 (11%), and the rate of these reactions fell over time, from about 70% at week 4 to 20% at week 124. Seven people overall (2.5% of 283) dropped out of the trial because of injection site reactions, including 1 person between weeks 96 and 124. Injection site reactions lasted for a median of 3 days.
Results indicate that in a closely observed and supported clinical trial, 4 of 5 people can maintain an established undetectable viral load through 124 weeks with once-monthly injected CAB + RPV. Throughout the study 5 people had confirmed virologic failure, including 1 between week 96 and week 124. Researchers recorded no drug-related serious adverse events after week 96.
References
1. Orkin C, Morell EB, Tan DHS, et al. Week 124 results of the randomized, open-label, phase 3 FLAIR study evaluating long-acting cabotegravir + rilpivirine for treatment in adults with HIV-1 infection (ITT-E population). IAS 2021, 11th IAS Conference on HIV Science, July 18-21, 2021. Abstract OAB0302.
2. ClinicalTrials.gov. Study to evaluate the efficacy, safety, and tolerability of long-acting intramuscular cabotegravir and rilpivirine for maintenance of virologic suppression following switch from an integrase inhibitor in HIV-1 infected therapy naive participants. ClinicalTrials.gov identifier NCT02938520. https://clinicaltrials.gov/ct2/show/NCT02938520
3. Orkin C, Arasteh K, Gorgolas Hernández-Mora M, et al. Long-acting cabotegravir and rilpivirine after oral induction for HIV-1 infection. N Engl J Med. 2020;382:1124-1135. doi: 10.1056/NEJMoa1909512.


3TC, lamivudine; ABC, abacavir; ART, antiretroviral therapy; CAB, cabotegravir; CAR, current antiretroviral therapy; DTG, dolutegravir; IM, intramuscular; LA, long-acting; RPV, rilpivirine.
1. U.S. Department of Health and Human Services. Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents with HIV. 2021. Available at: https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv/15/virologic-failure. Accessed March 25, 2021. 2. Saag MS, et al. JAMA. 2020;324(16):1651–1669. 3. ViiV Healthcare. Cabotegravir extended-release injectable suspension; rilpivirine extended-release injectable suspension (Cabenuva) Prescribing Information. US, January 2021. 4. ViiV Healthcare. Vocabria Summary of Product Characteristics. EU, December 2020.
5. ViiV Healthcare. Vocabria (cabotegravir tablets) and Cabenuva (cabotegravir and rilpivirine extended release injectable suspensions) Product Monograph. Canada, March 2020. 6. CABENUVA. Available at: https://viivhealthcare.com/en-au/our-medicines/cabenuva/. Accessed April 9, 2021. 7. Swindells S, et al. N Engl J Med. 2020;382(12):1112–1123. 8. Orkin C, et al. N Engl J Med. 2020;382(12):1124–1135. 9. Orkin, et al. Lancet HIV. 2021;8(4):e185–e196. 10. Overton ET, et al. Lancet. 2020;396(10267):1994–2005.
11. D'Amico R, et al. Glasgow HIV 2020;O414.

|
| |
|
 |
 |
|
|