 |
 |
 |
| |
Sub-40-Copy Virologic Hints May Favor
DTG/3TC Over Multidrug Combos in TANGO
|
| |
| |
IAS 2021, 11th IAS Conference on HIV Science, July 18-21, 2021
Mark Mascolini
Deep probes for hints at HIV replication below the standard undetectable cutoff offered some signals favoring switching to dolutegravir/lamivudine (DTG/3TC) over continuing a 3- or 4-drug combination including tenofovir alafenamide (TAF) in the TANGO trial [1]. For example, target not detected (TND) readings proved more frequent with DTG/3TC than with a TAF regimen, and people taking the TAF combination had more episodes of intermittent viremia (blips). But the clinical impact of these seeming advantages awaits further study.
The open-label TANGO trial found that switching to once-daily DTG/3TC is virologically noninferior to staying with a 3- or 4-drug TAF-based regimen [2]. Like most antiretroviral trials, TANGO measured virologic response with an assay that has a lower detection limit of 40 or 50 HIV RNA copies/mL. Yet researchers have long delved below those standard cutoffs for clues to low-level viral replication or portents of full-fledged rebounds. Taking the same tack, researchers from ViiV Healthcare and colleagues at other centers dissected 96 weeks of data from TANGO to see if DTG/3TC differs from 3- or 4-drug regimens in these realms below the standard detection limit.
Below its undetectable cutoff of 40 copies/mL, the Abbott RealTime HIV-1 assay also provides a qualitative reading of viral target detected (TD) or target not detected (TND). The ViiV team tracked TANGO participants’ TD/TND status over time, proportions of participants with a viral load ever above 40 copies, and frequency of elevated viral load categories including blips (transient jumps in measurable viremia).
Because the clinical impact of these viral readings remains hazy, the researchers focused on average change from baseline in standard inflammation markers. Most authorities see inflammation as an unwanted residue of imperfectly controlled HIV that may eventually have grave clinical consequences, like predisposition to cardiovascular disease or cancer.
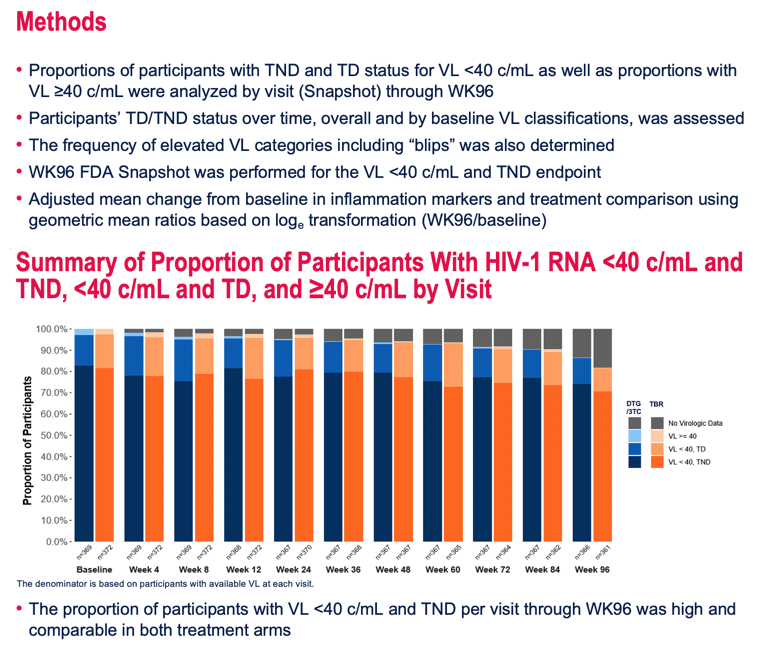
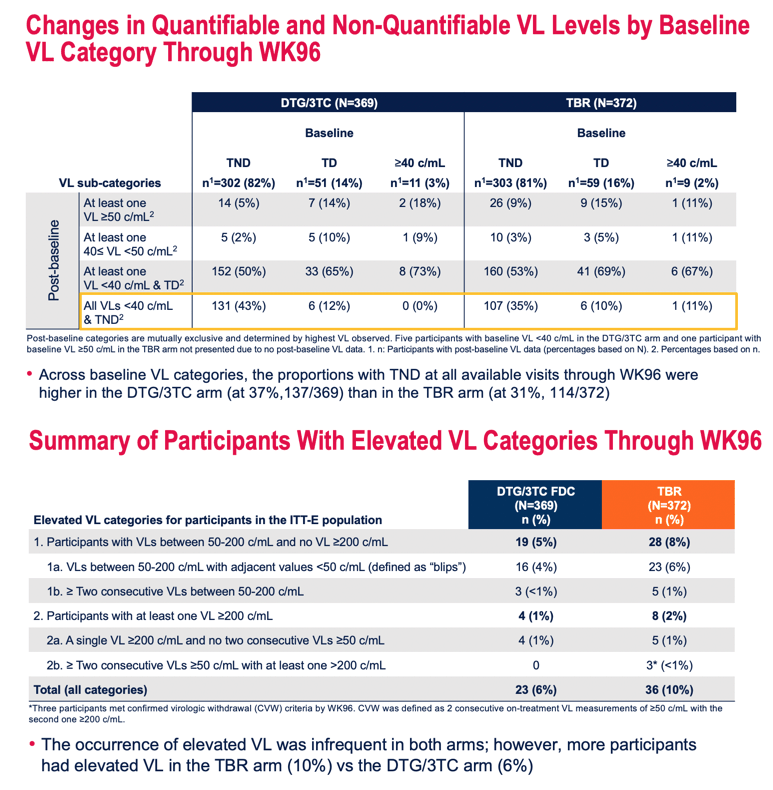
Looking at viral readings from the baseline TANGO visit plus 10 more visits during randomized treatment through week 96, the researchers found that proportions of participants with a viral load below 40 copies and TND per visit did not differ meaningfully between treatment arms. But scrutinizing all available visits through week 96, the researchers found that people taking DTG/3TC had a higher proportion of TND results (37%, 137 of 369 visits) than did people keeping their TAF regimen (31%, 114 of 372 visits).
By several other viral load measures through 96 weeks, people assigned to DTG/3TC did better than those assigned to keep their 3- to 4-drug regimen:
- Viral load between 50 and 200 copies but never above 200: 5% DTG/3TC, 8% control
- Viral load between 50 and 200 with adjacent reading under 50 (a blip): 4% DTG/3TC, 6% control
- Two consecutive viral load between 50 and 200: less than 1% DTG/3TC, 1% control
- At least 1 viral load at or above 200 copies: 1% DTG/3TC, 2% control
- Single viral load at or above 200 with no consecutive loads at or above 50: 1% DTG/3TC, 1% control
- Two or more consecutive loads at or above 50 copies and at least one above 200 copies: 0 DTG/3TC, less than 1% control
Summing all those novel metrics of low-level viral control, the researchers found a lower overall rate with DTG/3TC than with the TAF-based control regimen: 6% vs 10%.
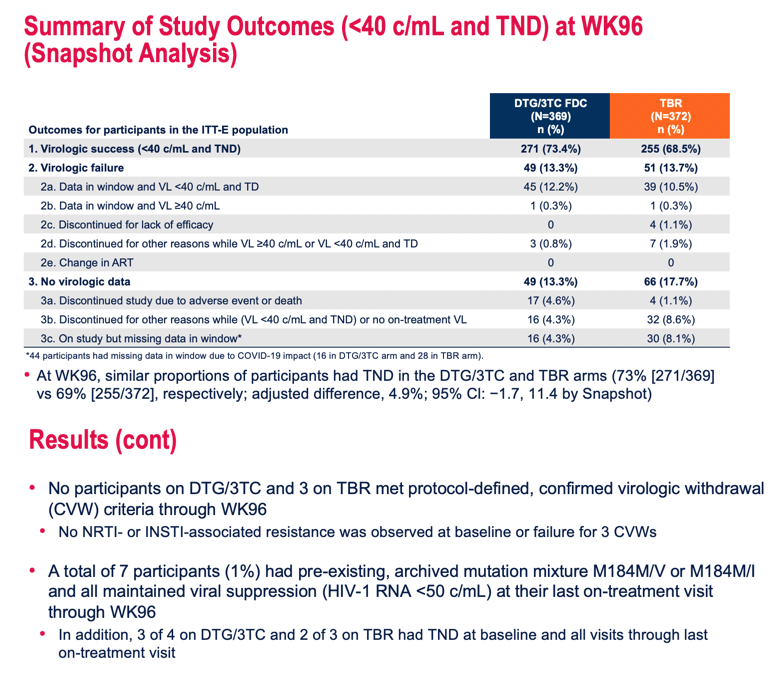
No one assigned to DTG/3TC versus 3 people assigned to continue their TAF regimen met protocol-defined confirmed virologic withdrawal criteria through week 96.
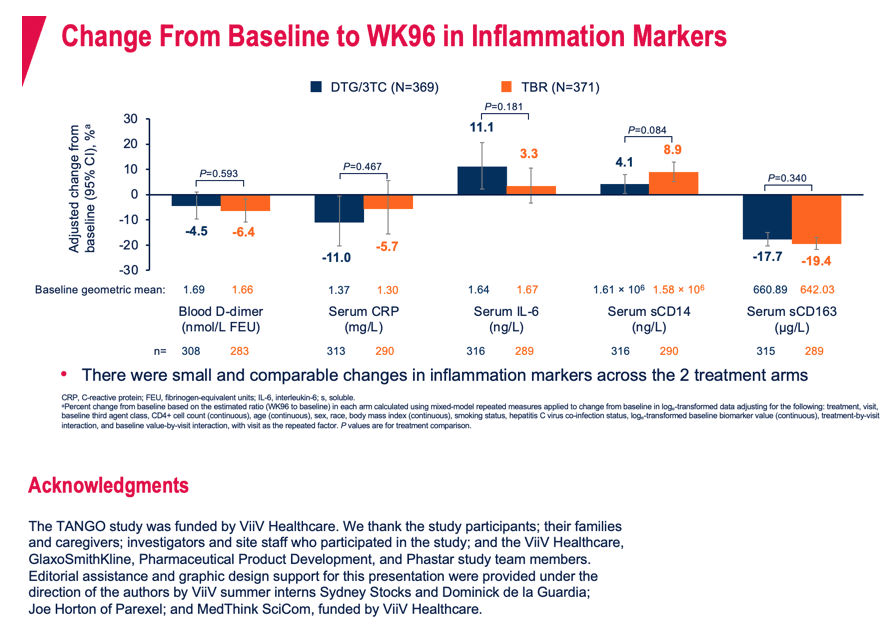
Charting levels of 5 standard inflammation markers through 96 weeks (D-dimer, C-reactive protein, IL-6, sCD14, sCD163), the researchers saw small and comparable changes in the two study arms. One marker, sCD14, rose twice higher over 96 weeks with the TAF control regimen than with DTG/3TC, but that difference stopped short of statistical significance (adjusted mean change from baseline 8.9% vs 4.1%, P = 0.084).
The researchers proposed that their “deep dive” findings bolster standard research showing the antiviral potency of this simple 2-in-1 drug combination, DTG/3TC. But whether these finely sifted virologic differences between DTG/3TC and more complex regimens have a clinical impact remains uncertain.
References
1. Wang R, Wright J, George N, et al. Comparison of viral replication for the 2-drug regimen (2DR) of dolutegravir/lamivudine (DTG/3TC) versus a 3/4-drug tenofovir alafenamide-based regimen (TBR) in the TANGO study through week 96. IAS 2021, 11th IAS Conference on HIV Science, July 18-21, 2021. Abstract OAB0301.
2. van Wyk J, Ajana F, Bisshop F, et al. Efficacy and safety of switching to dolutegravir/lamivudine fixed-dose 2-drug regimen vs continuing a tenofovir alafenamide-based 3- or 4-drug regimen for maintenance of virologic suppression in adults living with human immunodeficiency virus type 1: phase 3, randomized, noninferiority TANGO study. Clin Infect Dis. 2020;71:1920-1929. doi: 10.1093/cid/ciz1243.


|
| |
|
 |
 |
|
|