 |
 |
 |
| |
HCV Cure Rate 92% in Hard-to-Reach Vietnam Drug-Injecting Group
|
| |
| |
IAS 2021, 11th IAS Conference on HIV Science, July 18-21, 2021
Mark Mascolini
More than 9 in 10 HCV-infected drug injectors in a Vietnamese city attained sustained virologic response 12 weeks after treatment ended (SVR12) with a standard direct-acting antiviral (DAA) regimen [1]. DRIVE-C study researchers attribute the high success rate in this 1000-person analysis to free treatment and to avid recruitment, follow-up, and counseling by drug community members.
Researchers at Hai Phong’s Viet Tiep Hospital and others involved with the ANRS 12380 DRIVE-C study noted that identifying models to reach and treat HCV-positive people in challenging populations, like urban drug injectors, would be a strong step toward HCV elimination. They tested one such model in Hai Phong’s drug injectors—community-based HCV mass screening and follow-up coupled to simplified integrated hospital care. DRIVE-C involved drug users themselves in every stage of the strategy: HCV screening, linkage to care, treatment adherence support, social support, and harm reduction to prevent reinfection.
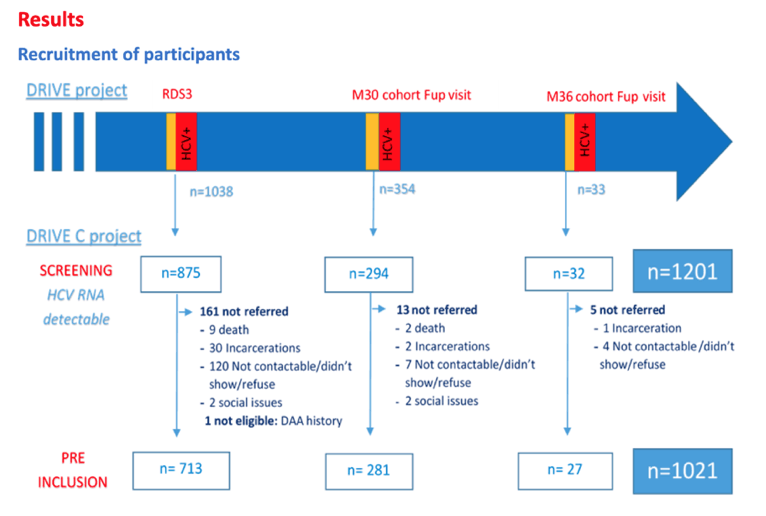
Hai Phong, a major port city in northeastern Vietnam, has a population of 2 million. The HCV study used two strategies to recruit Hai Phong drug injectors for HCV screening: a respondent-driven sampling survey and cohort follow-up visits of people enrolled in DRIVE-C, a community-based research program in Vietnam.
If initial HCV serology screening proved positive, a person got tested for HCV RNA. People with detectable HCV RNA (10 IU/mL or more) got referred by community members to one of three public hospitals for free pangenotypic treatment with 12 weeks of sofosbuvir/daclatasvir, plus ribavirin if Fibroscan indicated cirrhosis.
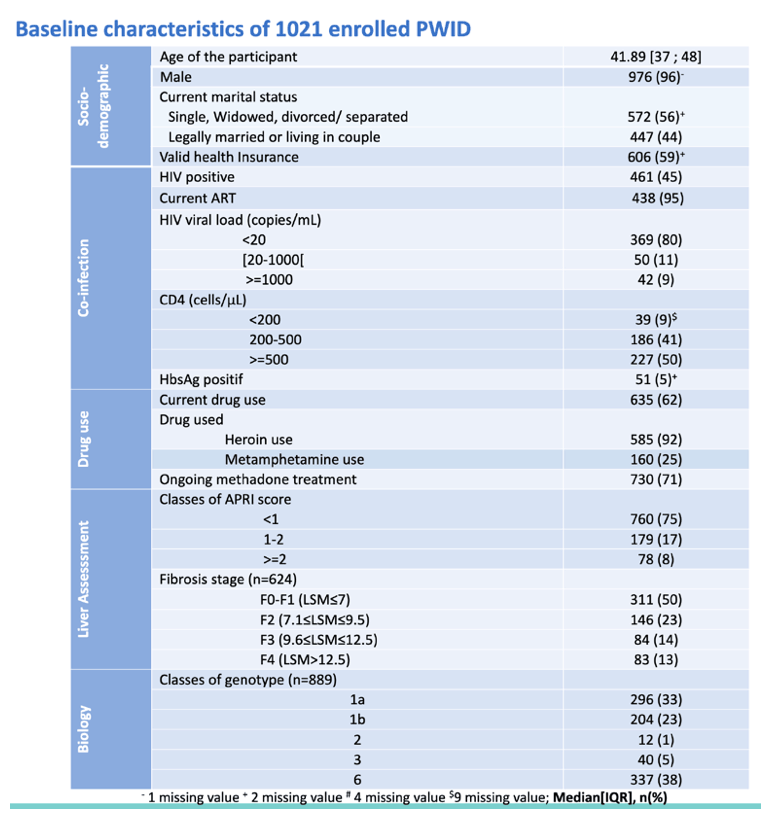
The study enrolled 1021 people who inject drugs with a median age of 41.9 years, 96% of them men. Of the 45% with HIV infection, 95% currently took antiretroviral therapy. Almost two thirds of the group, 62%, currently used illicit drugs, including heroin in 92% and methamphetamine in 25%. Most study participants, 71%, were enrolled in methadone maintenance.
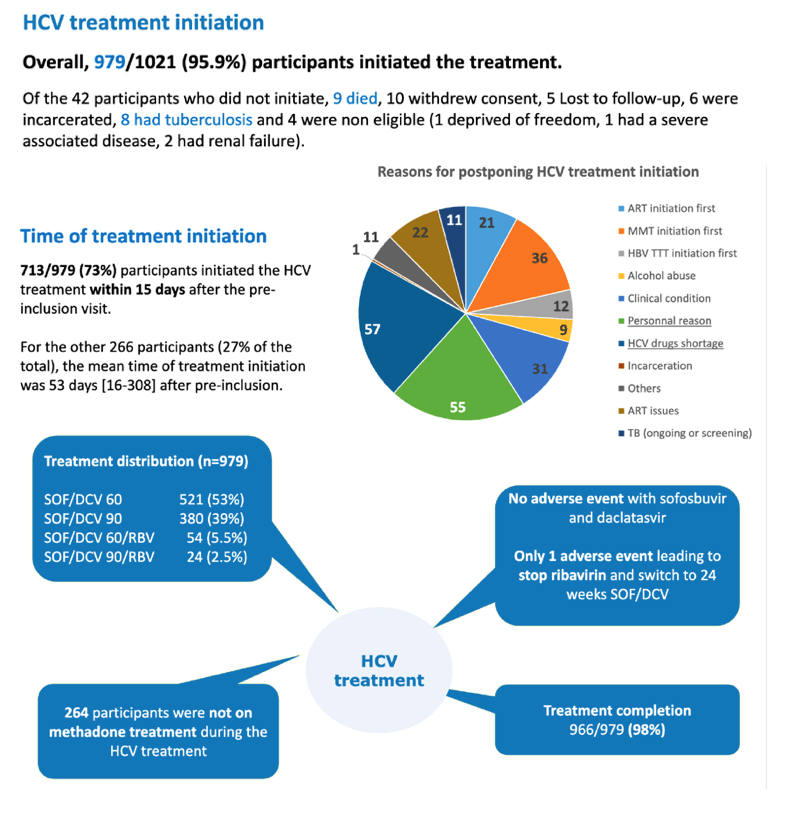
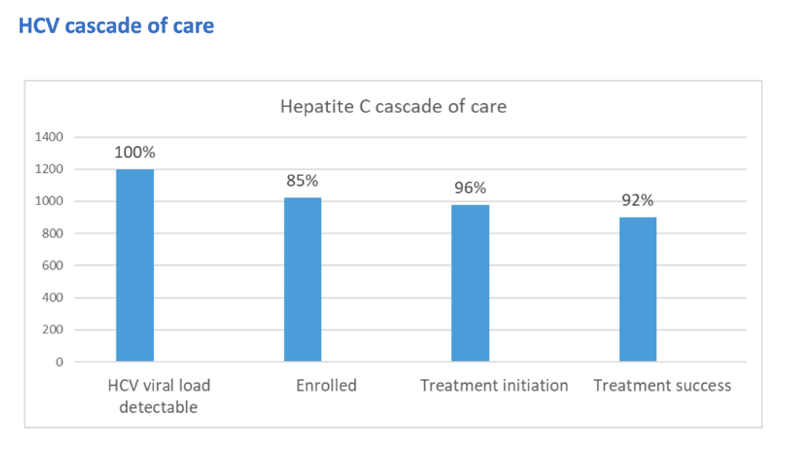
Of the 1021 participants, 979 (95.9%) started HCV therapy. Among the 42 people who did not start treatment, 10 withdrew consent, 9 died, 8 had tuberculosis, 4 were not eligible for other reasons, 6 were incarcerated, and 5 were lost to follow-up (stopped keeping study visits). Among the 979 people who began treatment, 713 (73%) did so within 15 days of their preinclusion visit. The remaining 266 people averaged 53 days to starting therapy.
Among the 979 people who started therapy, 924 (94%) kept their SVR12 visit. The most frequent reasons for missing the SVR12 visit were incarceration in 25, loss to follow-up in 9, and death in 5. Among all 979 participants who began treatment, 901 (92%) achieved SVR12. Among 23 people who kept the SVR12 visit and had a detectable HCV load, 10 had a different HCV genotype from their screening genotype, a finding suggesting early reinfection.
The DRIVE-C investigators concluded that a hard-to-reach drug-injecting population can achieve a high SVR rate in a program combining free treatment with step-by-step community group involvement.
Reference
1. Vinh VH, Binh NT, Khue PM, et al. Achieving an extremely high hepatitis C treatment success rate in people who inject drugs in Hai Phong, Vietnam: results of the ANRS 12380 DRIVE-C study. IAS 2021, 11th Conference on HIV Science, July 18-21, 2021. Abstract P.EB090.

|
| |
|
 |
 |
|
|