 |
 |
 |
| |
GS-9822, a preclinical LEDGIN, displays a block-and-lock phenotype in cell culture
|
| |
| |
IAS 2021 July 18-22
Presenter: Anne Bruggemans
Authors: A. Bruggemans * {1 ), G. Vansant (1 ), M. Balakrishnan (2), M.L. Mitchell (2), R. Cai (2), F. Christ (1 ), Z. Debyser (1 ).
Institutions: (1) KU Leuven, Molecular Virology and Gene Therapy, Leuven, Belgium, (2) Gilead Sciences Inc., Foster City, United States
Abstract:
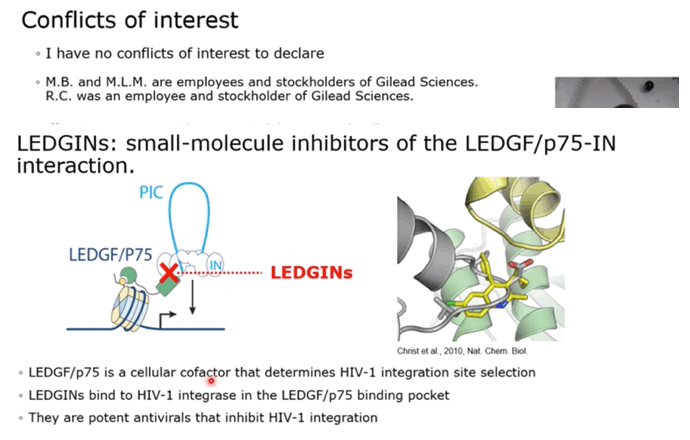
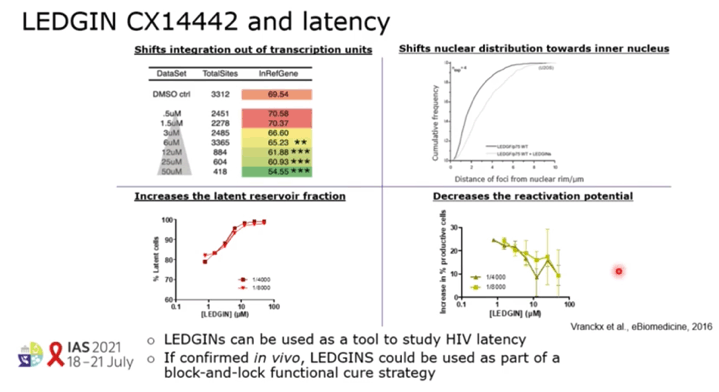
BACKGROUND: The ability of HIV to integrate into the host genome and establish latent reservoirs is the main hurdle towards an HIV cure. LEDGINs are small-molecule integrase that target the binding pocket of LEDGF/p75, a cellular cofactor that substantially contributes to HIV integration site selection. They are potent antivirals that inhibit HIV integration and maturation. In addition, they retarget residual integrants away from transcription units towards a more repressive chromatin environment. A previous study also demonstrated that after CX14442 treatment, residually integrated proviruses are more latent and refractory to reactivation, supporting the use of LEDGINs in a functional cure strategy.
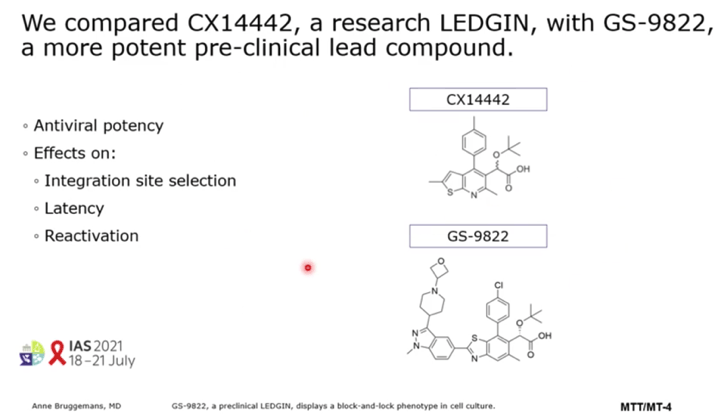
METHODS: In this study we compared GS-9822, a potent, pre-clinical lead compound, with the research compound CX14442 with respect to antiviral potency, integration site selection, latency and reactivation. Using AlphaScreen and multiple round HIV-1 replication in MT-4 cells we compared the activities of CX14442 and GS-9822. In addition, integration sites after LEDGIN treatment were sequenced (lllumina Miseq) and the surrounding chromatin environments were compared using the INSPIRED platform. Using established double reporter viruses we studied both latency and reactivation after treatment with either LEDGINs or the integrase inhibitor raltegravir as a control.
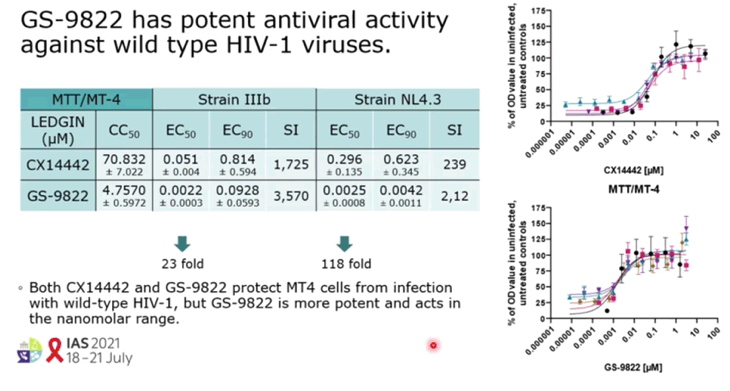
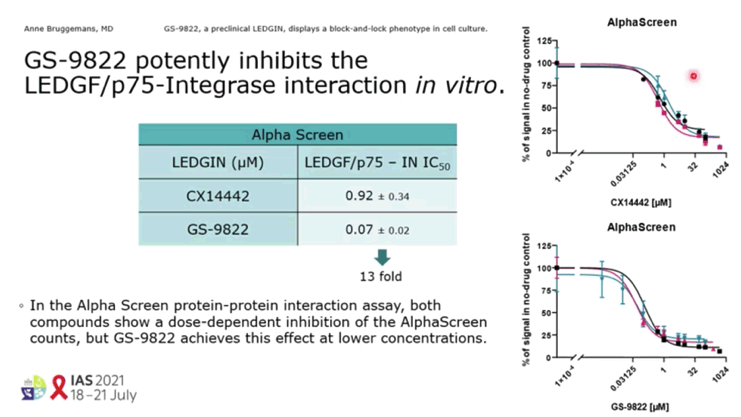
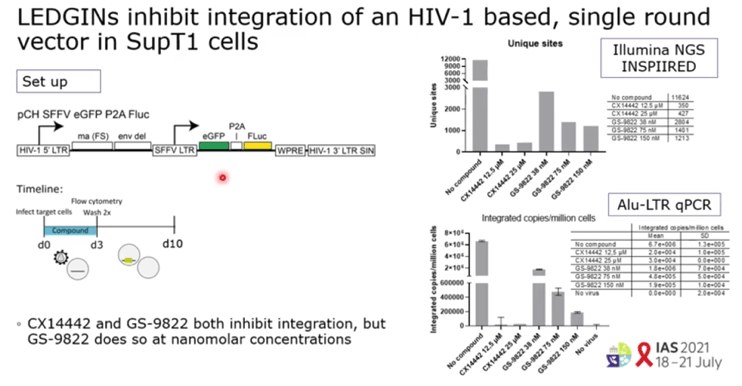
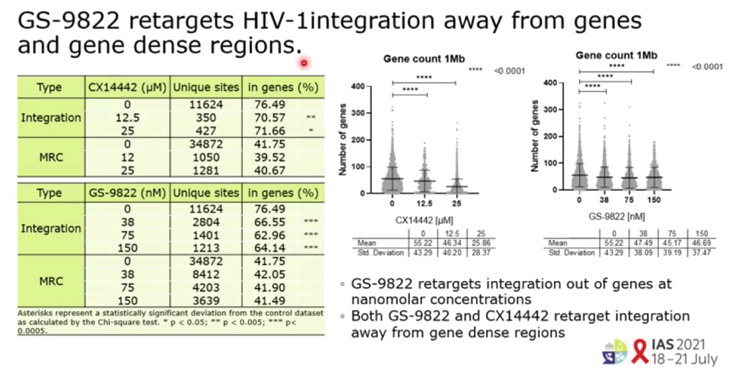
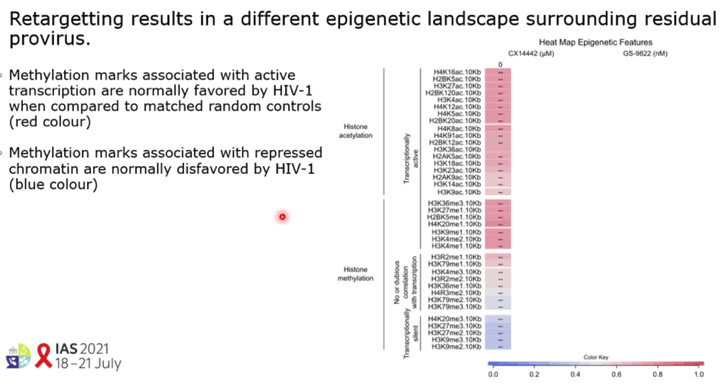
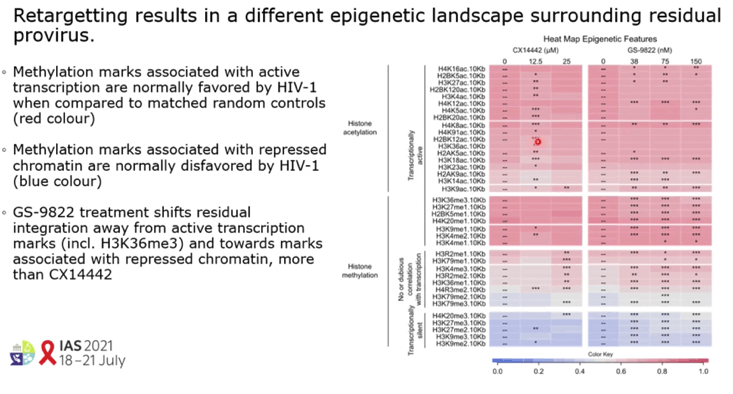
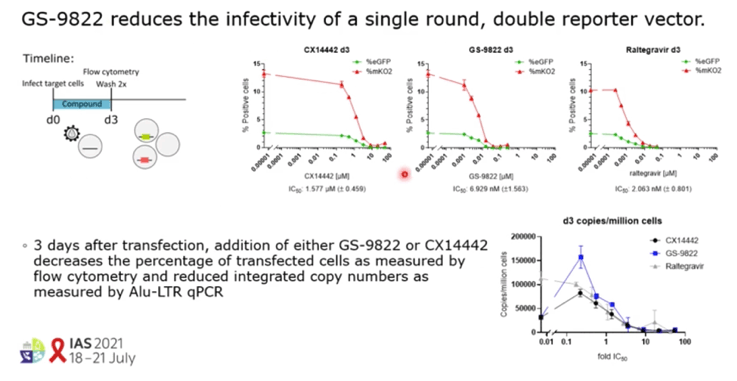
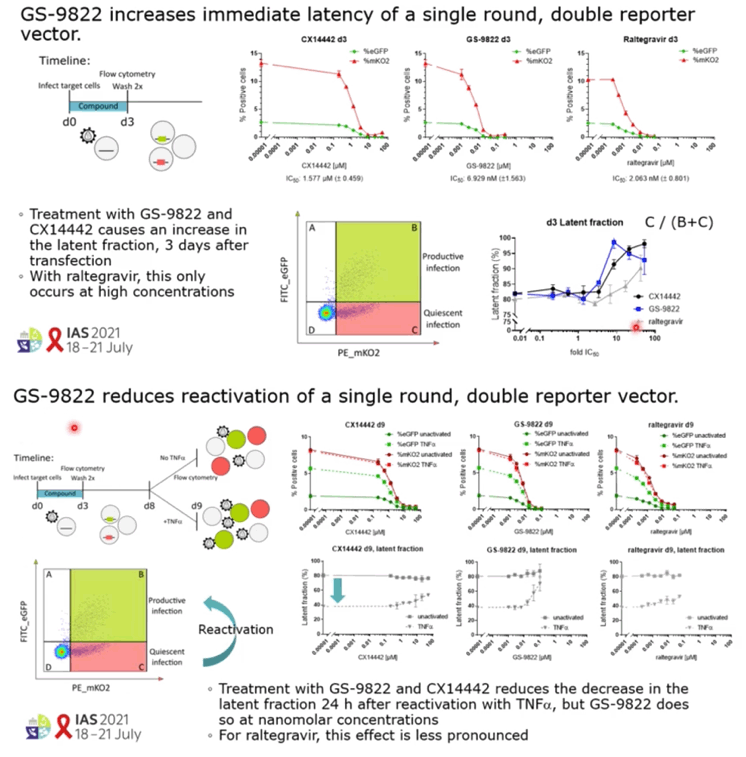
RESULTS: GS-9822, a pre-clinical LEDGIN, is a potent antiviral with nanomolar activity against wild type HIV-1. GS-9822 inhibits the LEDGF/p75-integrase interaction and reduces HIV-1 integration. Much like CX14442, GS-9822 was able to retarget integration of residual proviruses away from active genes and gene dense regions, resulting in a more repressive epigenetic landscape. Finally, when using a double reporter construct, CX14442 and GS-9822 were shown to reduce HIV-1 infectivity, increase immediate latency and decrease the reactivation potential of residual integrants. Remarkably, GS-9822 induced these effects at 200-300-fold lower concentrations than CX14442.
CONCLUSIONS: The ability to retarget integration sites and induce a deep latent state (block-and-lock) is not specific for a single LEDGIN, CX14442, but a class-effect related to the inhibition of the LEDGF/p75-integrase interaction. Highly potent LEDGIN compounds that inhibit this interaction can induce these effects at doses achievable in the clinic, making LEDGINs an interesting candidate for functional HIV cure research.




|
| |
|
 |
 |
|
|