 |
 |
 |
| |
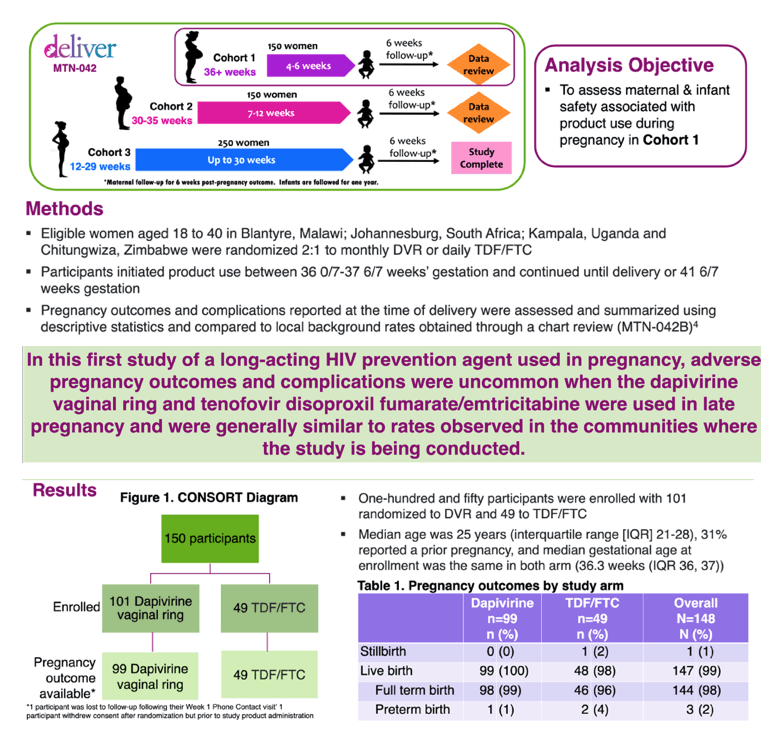
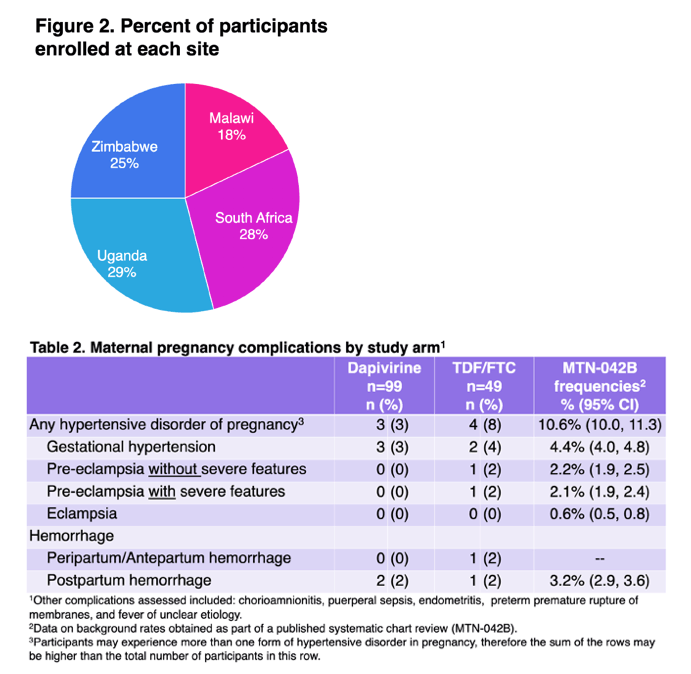
Prioritizing the evaluation of HIV prevention interventions in pregnancy: Interim results from a randomized, open label safety trial of dapivirine vaginal ring and oral tenofovir disoproxil fumarate/emtricitabine use in late pregnancy
|
| |
| |
IAS 2021 July 18-22
IAS: Good Adherence to Vaginal Ring and Daily PrEP in Half of Young African Women - (07/23/21)
Bonus Makanani, MBBS, FCOG(SA)1, Lee Fairlie, MBChB, FCPaeds2, Jennifer E. Balkus PhD3,4, MPH, Daniel Szydlo, MS4, Nyaradzo M. Mgodi, MBChB, MMed5, Clemensia Nakabiito, MBChB, MMed6, Ashley J. Mayo, MSPH7, Jeanna M. Piper, MD8 , Nahida Chakhtoura, MD, MsGH9, Sharon L. Hillier, PhD10, Katherine Bunge, MD, MPH10, for the MTN-042/DELIVER Team.
1Malawi College of Medicine-Johns Hopkins University Research Project; 2Wits Reproductive Health and HIV Institute, University of the Witwatersrand; 3Department of Epidemiology, University of Washington School of Public Health; 4Statistical Center for HIV/AIDS Research and Prevention, Fred Hutchinson Cancer Research Center; 5University of Zimbabwe Clinical Trials Research Centre (UZ-CTRC) 6Makerere University - Johns Hopkins University (MU-JHU) Research Collaboration; 7FHI 360; 8National Institute of Allergy and Infectious Diseases, National Institutes of Health; 9Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health; 10Department of Obstetrics, Gynecology and Reproductive Sciences, University of Pittsburgh


|
| |
|
 |
 |
|
|