 |
 |
 |
| |
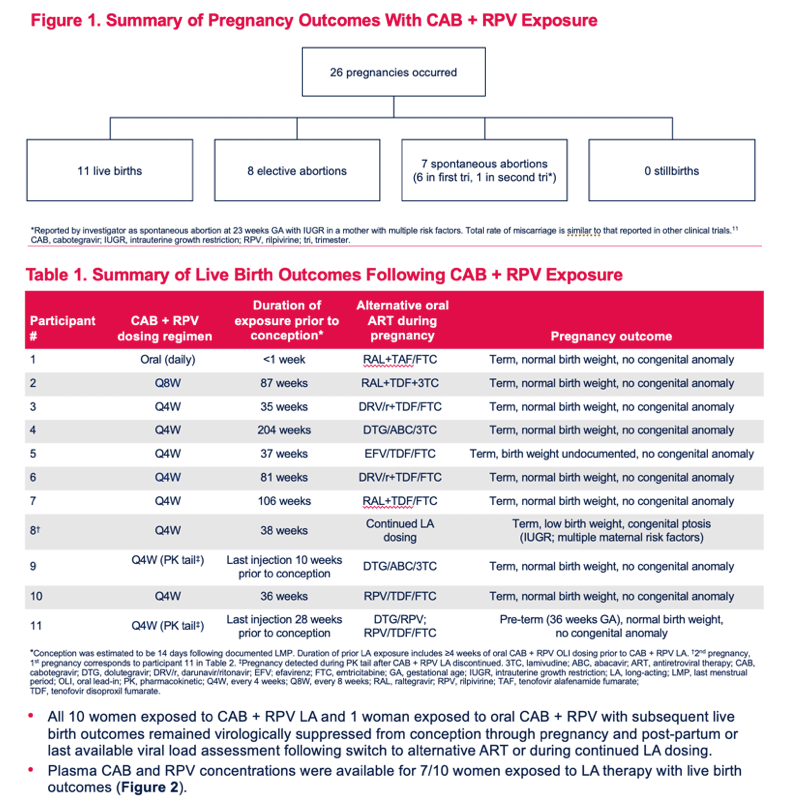
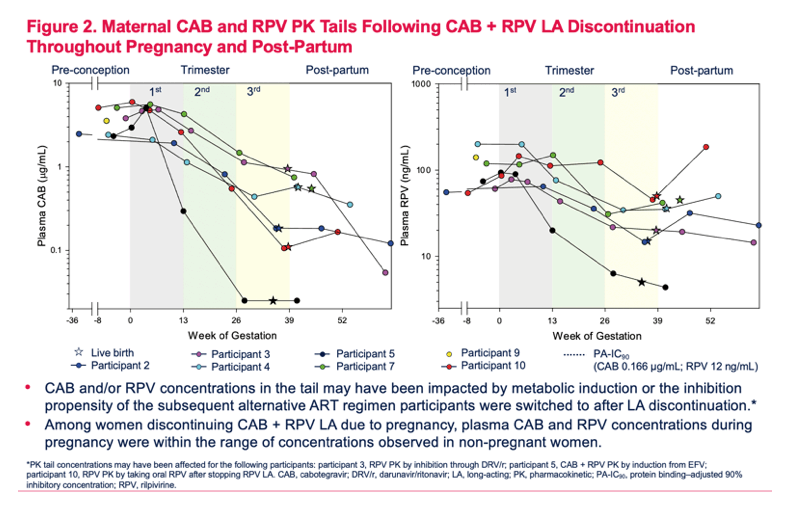
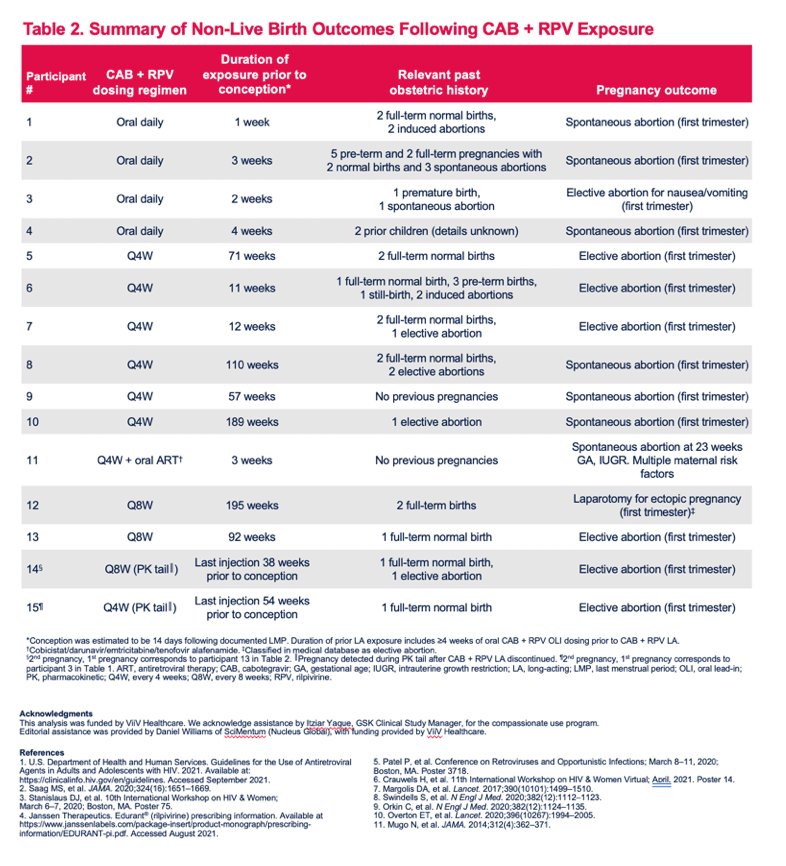
Pregnancy Outcomes and Pharmacokinetics in Pregnant Women Living With HIV Exposed to Long-Acting Cabotegravir and Rilpivirine in Clinical Trials
|
| |
| |
IDWeek 2021 Sept 29-Oct 3
Parul Patel1, Susan L. Ford2, Mark Baker3, Claudia Meyer4, Louise Garside4, Ronald D'Amico1, Rodica Van Solingen-Ristea5, Herta Crauwels5, Joseph W. Polli1, Ciara Seal6, Shanker Thiagarajah4, Eileen Birmingham7, William R. Spreen1, Bryan Baugh8, Matt Bosse9, Vani Vannappagari1
1ViiV Healthcare, Research Triangle Park, NC, United States; 2GlaxoSmithKline, Research Triangle Park, NC, United States; 3ViiV Healthcare, Nyon, Switzerland; 4GlaxoSmithKline, London, United Kingdom; 5Janssen Research & Development, Beerse, Belgium; 6GlaxoSmithKline, Upper Providence, PA, United States; 7Janssen Research & Development, Raritan, NJ, United States; 8Janssen Research & Development, Titusville, NJ, United States; 9ViiV Healthcare, San Diego, CA, United States


|
| |
|
 |
 |
|
|