 |
 |
 |
| |
HDV Load Drops, Liver Markers
Improve With Bulevirtide for 48 Weeks
|
| |
| |
AASLD-The Liver Meeting, November 4-8, 2022, Washington, DC
Mark Mascolini
In 48 weeks bulevirtide monotherapy for HDV-positive people with advanced compensated cirrhosis lowered viral load by a median 3.1 log10 IU/mL while improving markers of liver health in a 20-person single-center study [1]. Seven people reached an undetectable HDV RNA load.
Bulevirtide, a hepatitis delta virus (HDV) entry inhibitor, has received conditional approval in Europe for people with compensated chronic cirrhosis. The US FDA has not yet given bulevirtide the green light, citing concerns about production and delivery of the novel agent [2], which would be the first anti-HDV drug in the United States.
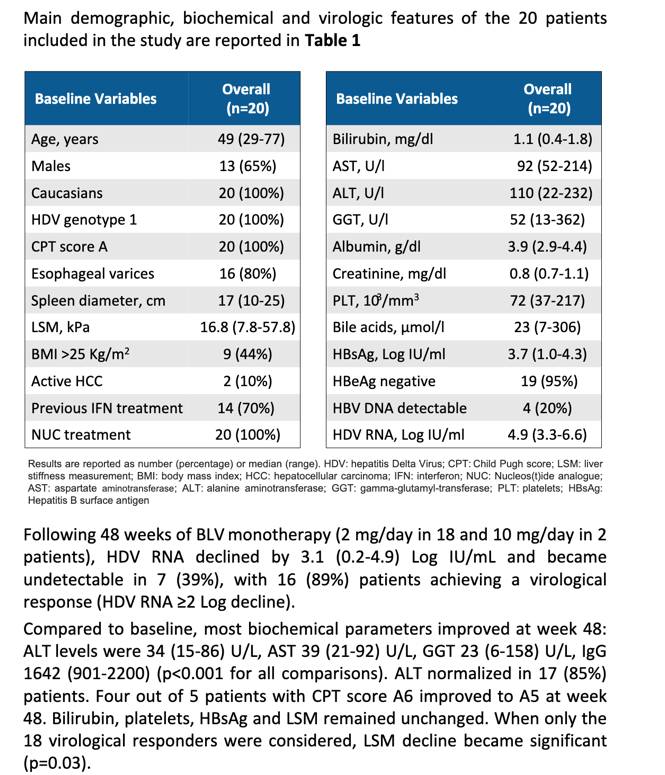
Because little is known about long-term response to bulevirtide monotherapy, clinical researchers at Milan's Foundation IRCCS Ca'Granda Ospedale Maggiore Policlinico and other centers conducted this single-site study of the anti-HDV drug in people with compensated cirrhosis and clinically significant portal hypertension. They consecutively enrolled 20 people with this condition who took bulevirtide monotherapy for 48 weeks.
The study group had a median age of 49 (range 29 to 77), all 20 were white, and 13 were men. Sixteen participants had esophageal varices, 9 had a body mass index in or above the overweight range, all had taken a nucleos(t)ide analog, and 14 had interferon therapy. Median pretreatment HDV RNA load stood at 4.9 log IU/mL, and 4 people had detectable HBV DNA. Median values for some standard liver markers were liver stiffness measure (LSM) 16.8 kPa, bilirubin 1.1 mg/dL, AST 92 U/L, ALT 110 U/L, and gamma-glutamyl transferase (GGT) 52 U/L. Nineteen of 20 participants tested negative for HBeAg, a signal of ongoing HBV replication.
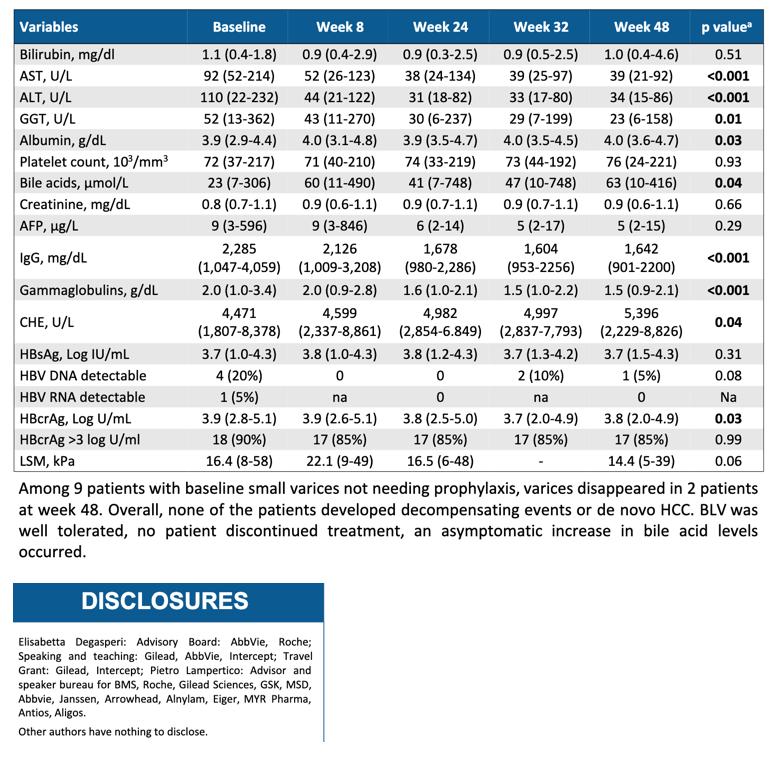
For 48 weeks, 18 people took bulevirtide monotherapy at a dose of 2 mg/day, and 2 people took 10 mg/day. No one stopped treatment during the 48 weeks, and no one had decompensating events or newly diagnosed hepatocellular carcinoma. Among 9 people with small esophageal varices before they started bulevirtide, varices disappeared in 2 people by treatment week 48.
Through 48 weeks HDV DNA dropped by a median 3.1 log10 IU/mL and became undetectable in 7 people (35%). Sixteen people had an HDV RNA decline of 2 log10 or more.
Median ALT improved to 34 U/L, AST to 39 U/L, and GGT to 23 U/L (P < 0.001 for all changes from pretreatment levels). ALT reached a normal level in 17 people (85%)). Bilirubin, platelets, HBsAg, and LSM did not change substantially during 48 weeks of treatment. When the researchers looked only at virologic responders, they charted a significant improvement in LSM (P = 0.03). In 5 people with a pretreatment CPT score (a gauge of cirrhosis severity) of A6, 4 people improved to A5 at week 48.
References
1. Degasperi E, Anolli MP, Colonia S, et al. Improvement of clinical parameters in HDV patients with advanced compensated cirrhosis treated with bulevirtide monotherapy for 48 week. AASLD-The Liver Meeting, November 4-8, 2022, Washington, DC. Abstract 1011.
2. Brooks M. FDA rejects bulevirtide for hepatitis D. Medscape Medical News. October 31, 2022. https://www.medscape.com/viewarticle/983305

|
| |
|
 |
 |
|
|