 |
 |
 |
| |
SARS-CoV-2 CSF N Antigen Detection is Associated with CNS Inflammation in Neuro-COVID
|
| |
| |
CROI 2022 Feb 11-16
Arvid Eden
Department of Infectious Diseases, Institute of Biomedicine,
Sahlgrenska Academy, University of Gothenburg,
Gothenburg, Sweden

program abstract
Arvid Eden1, Anna Grahn1, Daniel Bremell1, Pradeepthi Bathala2, Dietmar Fuchs3, Johanna Gostner4, Salvia Mishagian2, Navaratnam Manjula2, Staffan Nilsson1, Michael Schöll1, George Sigal2, Erika Stentoft1, Martin Stengelin2, Henrik Zetterberg1, Magnus Gisslen1
1Sahlgrenska Academy at the University of Gothenburg, Gothenburg, Sweden, 2Meso Scale Diagnostics, LLC, Rockville, MD, USA, 3Institute of Biological Chemistry, Medical University of Innsbruck, Biocenter, Innsbruck, Austria, 4Institute of Medical Biological Chemistry, Medical University of Innsbruck, Biocenter, Innsbruck, Austria
Background:
The underlying CNS pathogenesis in COVID-19 is not clear and viral RNA is rarely detected in cerebrospinal fluid (CSF). We measured viral antigen and biomarker profiles in CSF in relation to neurological symptoms and disease severity.
Methods:
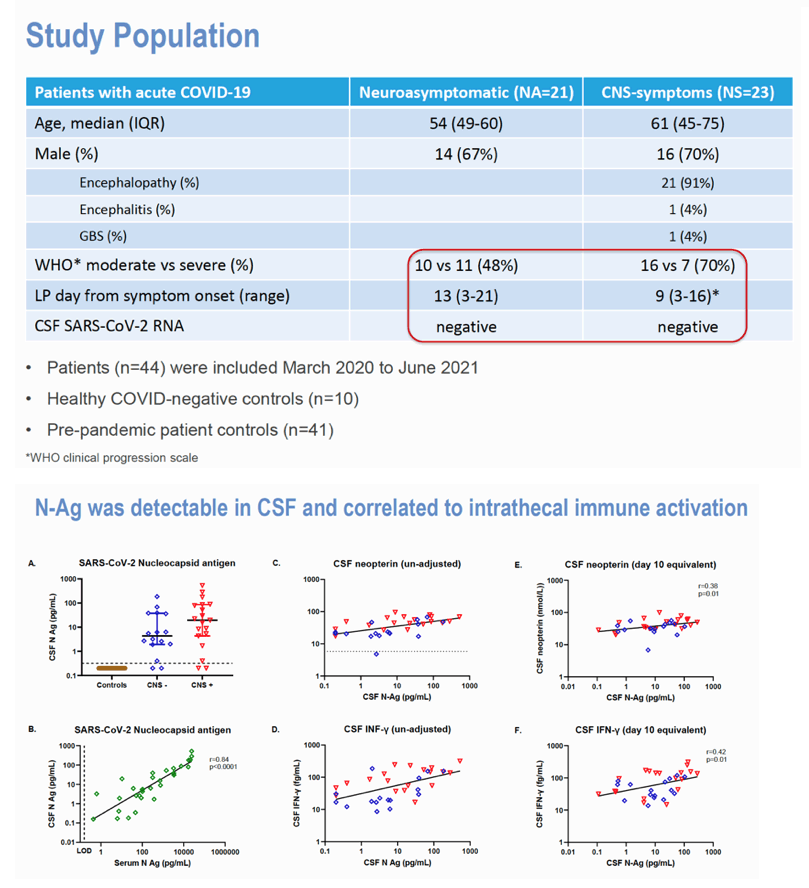
We included 44 (32% female) hospitalized patients (26 moderate, 18 severe COVID-19) and 10 healthy controls (HC). 21 patients were neuroasymptomatic (NA), 23 neurosymptomatic (NS; encephalopathy=21, encephalitis=1, GBS=1). For antigen and cytokine analyses, a patient control (PC; n=41) group (COVID-negative with no sign of CNS infection in clinical CSF samples) was used. CSF nucleocapsid antigen (N-Ag) was analyzed using an ultrasensitive antigen capture immunoassay platform, S-PLEX direct detection assay, S-PLEX SARS-CoV-2 N Kit (MesoScale Diagnostics, LLC. Rockville, MD). Additional analyses included CSF neopterin, β2-microglobulin, cytokines and neurofilament light (NfL).
Results:
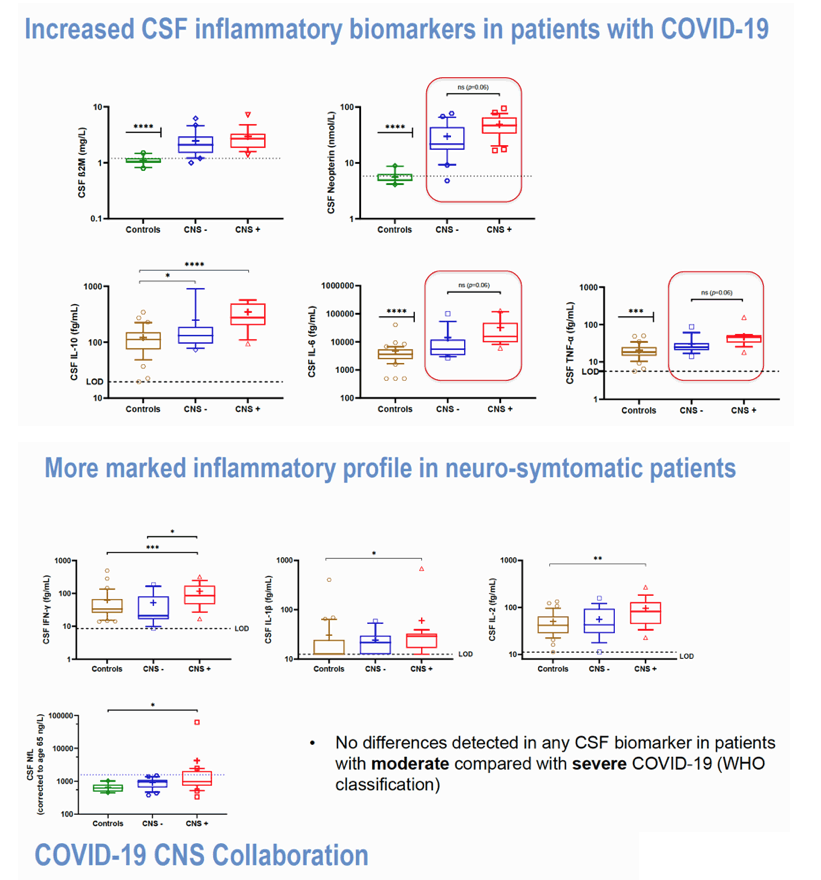
CSF N-Ag was detected in 31/35 patients (0/41 controls) while viral RNA was negative in all. CSF N-Ag was significantly correlated with CSF neopterin (r=0.38; p=0.03) and IFN-γ (r=0.42; p=0.01) adjusted for sampling day. No differences in CSF N-Ag concentrations were found between patient groups. All patient groups had markedly increased CSF neopterin, β2M, IL-6, IL-10 and TNF-α compared to controls, while IL-2, IL-1β and IFN-γ were significantly increased only in the NS group. CSF biomarkers were associated with time from symptom onset to CSF sampling. After adjusting for time of sampling, the NS group had significantly higher CSF IFN-γ (p=0.03), and showed a statistical trend towards significantly higher CSF neopterin, IL-6 and TNF-α (p=0.056-0.06) than the NA group. Additionally, age-adjusted CSF NfL was higher in the NS compared to the HC (p=0.01) group. No differences were seen in any CSF biomarkers in moderate compared to severe disease.
Conclusion:
Viral antigen is detectable in CSF in a majority of patients with COVID-19 despite the absence of detectable viral RNA, and is correlated to CNS immune activation markers. Patients with neurological symptoms had a more marked immune activation profile compared to NA patients, as well as signs of neuroaxonal injury compared to controls. These observations could not be attributed to a difference in COVID-19 severity. Our results highlight the importance of neurological symptoms and indicate that the CNS immune response and CNS pathogenesis can be initiated by viral components without direct viral invasion of the CNS.

|
| |
|
 |
 |
|
|