 |
 |
 |
| |
Integration of HIV and HCV Services with Medication for Opioid Use Disorder in the U.S.
|
| |
| |
CROI 2022 Feb 11-16
Eshan U. Patel 1, Becky L. Genberg 1, Xianming Zhu 2, Noa Krawczyk 3, Shruti H. Mehta 1, Aaron A.R. Tobian 1
1 Johns Hopkins Bloomberg School of Public Health, Baltimore, MD, United States, 2 The Johns Hopkins University School of Medicine, Baltimore, MD, United States, 3 New York University, New York City, NY, United States

program abstract
Background:
The US National Strategic Plan to End the HIV Epidemic (EHE) includes recommendations to integrate programs combatting the syndemics of HIV, viral hepatitis, and substance use disorders (SUDs). Facilities that offer medication for opioid use disorder (MOUD) are ideal settings to co-locate HIV/HCV services, as they serve a high-risk population and MOUD is associated with improved HIV/HCV treatment outcomes. We examined HIV/HCV testing and treatment availability in SUD treatment facilities that offer MOUD in the US.
Methods:
The 2017-2020 National Survey of Substance Abuse Treatment Services is an annual census of US SUD treatment facilities (response rates=89-92%). The analysis was restricted to facilities that offer MOUD (methadone/buprenorphine/naltrexone) and stratified by federal Opioid Treatment Program (OTP) status, with non-OTPs being facilities that only offer buprenorphine/naltrexone. Prevalence differences (PD) in HIV/HCV testing availability between 2017 and 2020 were examined using binomial regression. We also assessed HIV/HCV testing and treatment availability by facility factors in 2020.
Results:
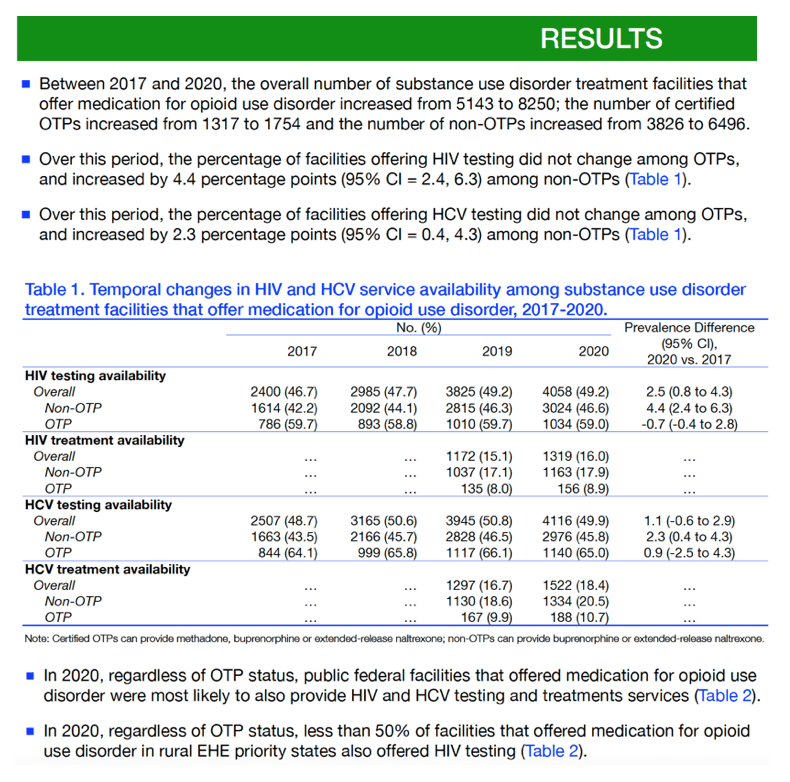
Between 2017 and 2020, the no. of facilities that offered MOUD increased from 5143 to 8250 and the no. of OTPs increased from 1317 to 1754.
Over this period, HIV testing availability only increased from 42.2% to 46.6% in non-OTPs (PD=4.4 [95%CI=2.4, 6.3) and there was no change among OTPs (59.7% to 59.0%; PD=-0.7 [95%CI=-0.4, 2.8]).
HCV testing availability only increased from 43.5% to 45.8% in non-OTPs (PD=2.3 [95%CI= 0.4, 4.3]) and there was no change among OTPs (64.1% to 65.0%; PD=0.9 [95%CI=-2.5, 4.3]). Of the non-OTPs in 2020, 17.9% (n=1,163) offered HIV treatment and 20.5% (n=1,334) offered HCV treatment.
Of the OTPs in 2020, 8.9% (n=156) offered HIV treatment and 10.7% (n=188) offered HCV treatment.
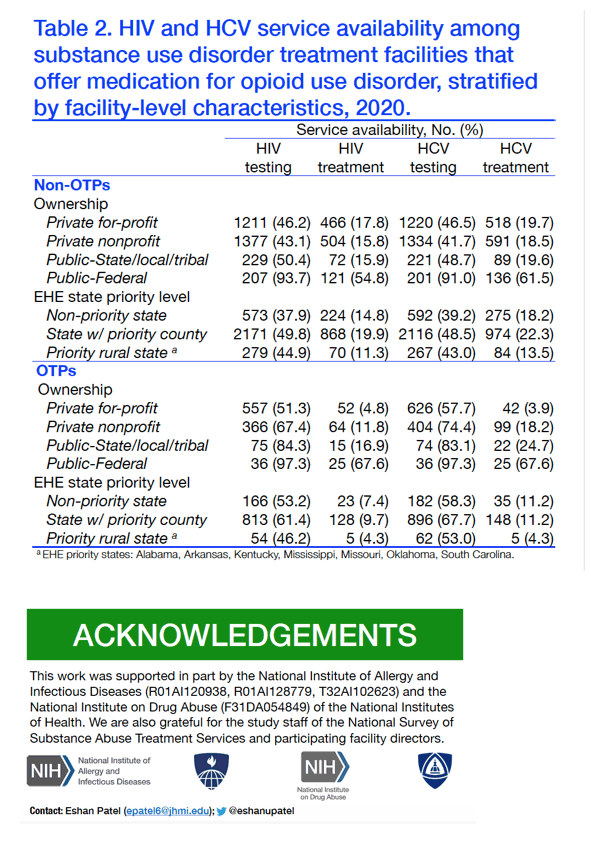
Regardless of OTP status, private for-profit facilities were less likely than federal facilities to offer HIV/HCV testing and treatment services.
In the EHE high-priority rural states, less than half of non-OTPs (44.9%; n=279) and OTPs (46.2%; n=54) offered HIV testing, and only 11.3% (n=70) of non-OTPs and 4.3% (n=5) of OTPs offered HIV treatment.
Conclusion:
Despite increases in the number of facilities providing MOUD in the US, integration of HIV and HCV services remains suboptimal, particularly in EHE high-priority rural states. This represents a missed opportunity to engage at risk marginalized populations in HIV and HCV care, which will be critical for achieving EHE goals.

|
| |
|
 |
 |
|
|