 |
 |
 |
| |
PHASE IIA TRIAL OF ISLATRAVIR QM FOR HIV PrEP: WEEK 24 METABOLIC AND RENAL OUTCOMES
|
| |
| |
CROI 2022 Feb 11-16

Program abstract
Pippa J. Macdonald1, Sharon A. Riddler2, Sharlaa Badal-Faesen3, Howard Schwartz4, Johannes Lombaard5, Yoseph Caraco6, Avivit Peer7, Ciaran McMullan8, Keith D. Kaufman8, Brenda Homony8, Katherine Nedrow8, Valerie Teal8, Michael N. Robertson8, Rebeca M. Plank8
1Desmond Tutu HIV Centre, Cape Town, South Africa, 2University of Pittsburgh, Pittsburgh, PA, USA, 3Clinical HIV Research Unit, University of the Witwatersrand, Johannesburg, South Africa, 4Research Centers of America, Hollywood, FL, USA, 5Josha Research, Bloemfontein, South Africa, 6Hadassah Medical Center, Jerusalem, Israel, 7Rambam Health Care Campus, Hafia, Israel, 8Merck & Co, Inc, Kenilworth, NJ, USA
program abstract
Background:
Adverse events (AEs) and long-term toxicities, including weight gain, loss of bone mineral density (BMD), and changes in renal function, have been reported among persons taking approved HIV-1 pre-exposure prophylaxis (PrEP) regimens. Islatravir (ISL) is a nucleoside reverse transcriptase translocation inhibitor under clinical investigation for the treatment and prevention of HIV-1. ISL has a long half-life and a pharmacokinetic profile compatible with extended-duration dosing regimens. Here we present metabolic and renal outcomes through Week 24 of a phase 2a trial of ISL 60 mg, ISL 120 mg, or matching placebo, all dosed once monthly (QM).
Methods:
This randomized, double-blind, placebo-controlled, parallel-group, multi-center trial (Israel, South Africa, and USA) assessed the safety, tolerability, and pharmacokinetics of monthly oral ISL in adults (age 18-65 years) at low-risk for HIV-1 acquisition (NCT04003103). Participants were randomly assigned (2:2:1) to receive a total of 6 QM doses of ISL 60 mg, ISL 120 mg, or matching placebo. Metabolic outcomes (weight, total hip BMD, lumbar spine BMD, peripheral fat, trunk fat) and renal outcomes (serum creatinine, estimated glomerular filtration rate [eGFR], retinol-binding protein/creatinine ratio) were assessed through Week 24. Median percent change from baseline and interquartile range (IQR) were determined.
Results:
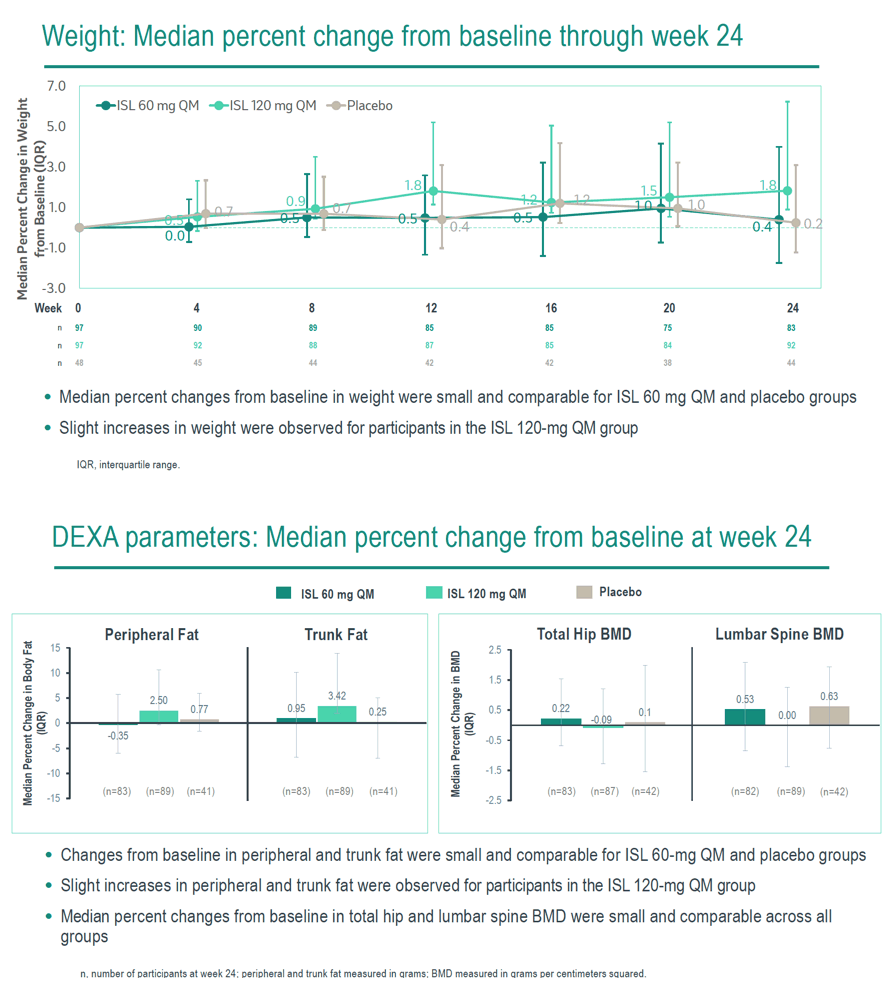
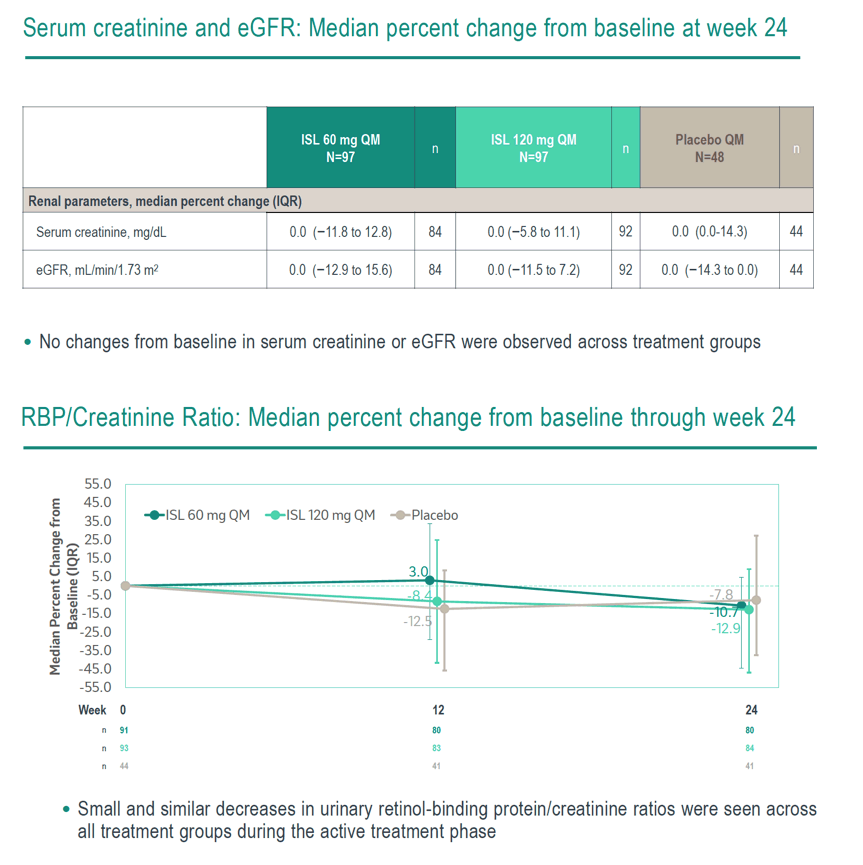
Of 242 randomized participants (median age, 31 years; male, 33%; White, 53%; Black or African American, 42%), 222 completed dosing and 20 discontinued study intervention prior to Week 24; none discontinued due to metabolic or renal AEs. Median body mass index at baseline for ISL 60 mg, ISL 120 mg, and placebo was 26.8 kg/m², 25.8 kg/m², and 26.6 kg/m², respectively. Median percent changes from baseline in weight, total hip BMD, lumbar spine BMD, peripheral fat, and trunk fat were small and comparable for ISL 60 mg QM and placebo groups, with slight increases in weight, peripheral fat, and trunk fat in the ISL 120 mg QM group (Table). No change in eGFR was observed across treatment groups; small and similar decreases in urinary retinol-binding protein to creatinine ratios were seen across all treatment groups (Table).
Conclusion:
No clinically meaningful differences from placebo in metabolic and renal parameters were observed with ISL 60 mg or ISL 120 mg after 6 QM doses. Ongoing phase 3 clinical trials of ISL 60 mg QM for PrEP will provide additional insights into renal and metabolic outcomes with longer ISL exposures.



|
| |
|
 |
 |
|
|