 |
 |
 |
| |
Immunogenicity and reactogenicity to COVID-19 mRNA vaccine additional dose in PLWH
|
| |
| |
CROI 2022 Feb 11-16
Vergori Alessandra1, Cicalini Stefania1, Cozzi Lepri Alessandro2, Matusali Giulia3, Bordoni Veronica4, Lanini Simone1, Colavita Francesca3, Cimini Eleonora4, Iannazzo Roberta1, De Pascale Lydia1, Castilletti Concetta3, Agrati Chiara4, Girardi Enrico5, Vaia Francesco6, Antinori Andrea1 for the HIV-VAC study group.
1 HIV/AIDS Unit, National Institute for Infectious Diseases Lazzaro Spallanzani IRCCS, Rome, Italy, 2 Centre for Clinical Research, Epidemiology, Modelling and Evaluation (CREME), Institute for Global Health, UCL, London, UK; 3 Laboratory of Virology; 4 Laboratory of Cellular Immunology and Clinical Pharmacology; 5 Scientific Direction; 6Health Direction; National Institute for Infectious Diseases Lazzaro Spallanzani IRCCS, Rome, Italy

RESULTS
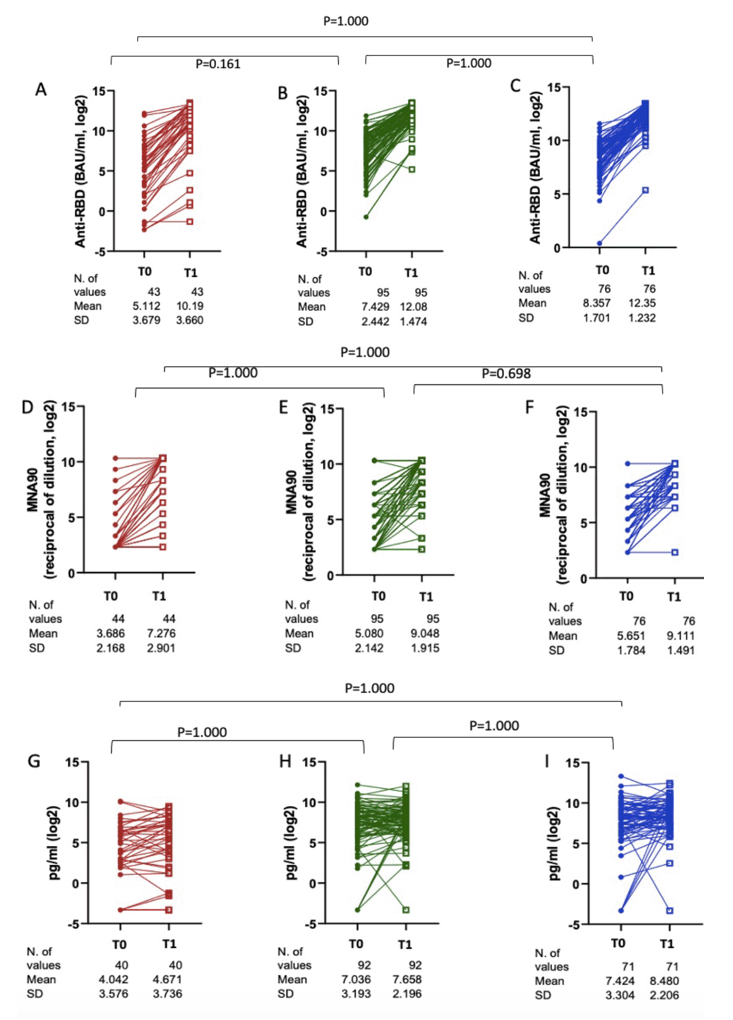
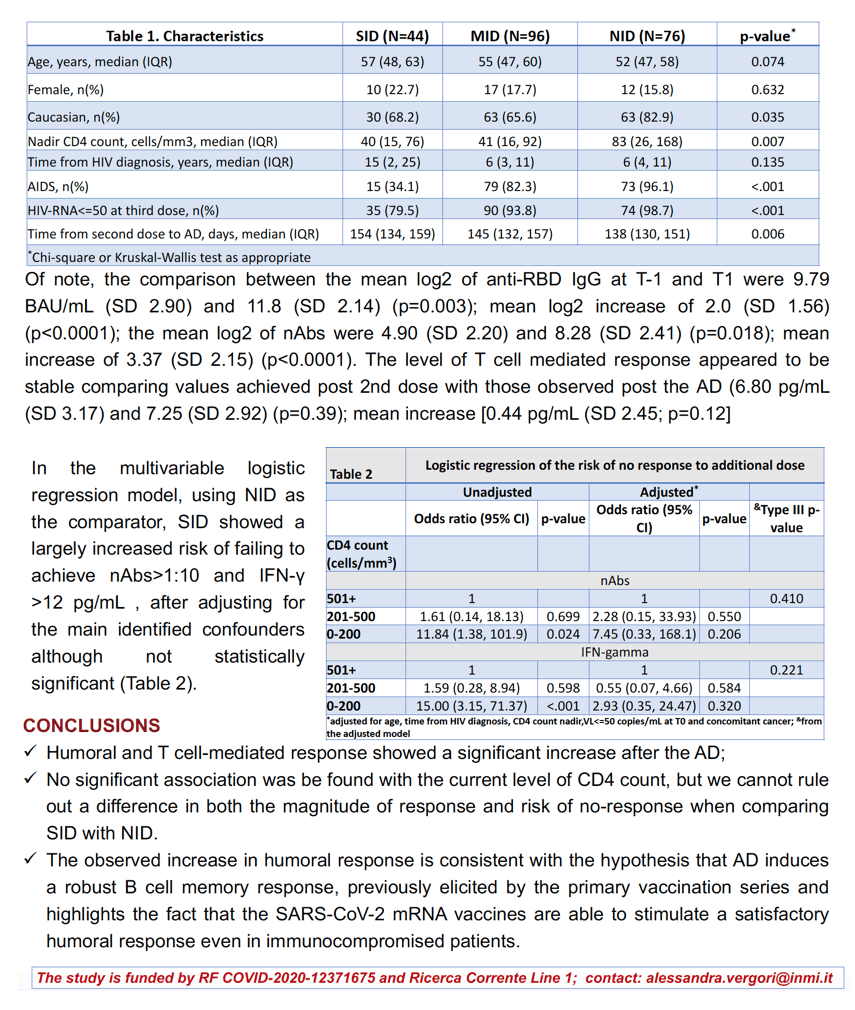
We enrolled 216 PLWH on ART (n=44 SID, n=96 MID, n=76 NID): median age 54 yrs (IQR 47-59), median CD4 nadir 45 cell/mm3 (20-122), 93% HIV-RNA <50 c/mL, 7yrs (3-12) since HIV diagnosis and 5yrs (2-8) since AIDS, if diagnosed (see Table 1). Response rate was 95.5% in SID, 100% in MID, 100% in NID for anti-RBD (p=0.02); 86.3%, 97.9% and 98.7% for nAbs (p=0.002), and 70%, 95.6% and 97.2% for IFN-γ (p<0.0001) , respectively. A significant increase of AD immunogenicity was observed in all CD4 count groups (Fig. 1 A-I). 95% reported mild symptoms, 14% moderate symptoms and 2% severe symptoms not requiring hospitalization.
DEFINITIONS: IgG anti RBD/S positive if >7.1 BAU/mL; MNA positive if titres of neutralizing antibodies (nAbs) >=1:10, IFN-γ positive if > 12 pg/mL;
Severe immunodeficiency, SID: <200/mm3; minor immunodeficiency, MID: 200-500/mm3; no immunodeficiency, NID: >500/mm3
PROGRAM ABSTRACT
Background:
In Italy in September 2021, administration of a booster shot (BS) of COVID-19 vaccine was approved for PLWH with advanced disease (current CD4 count<200 cell/mm3 and/or previous AIDS). The aim of this analysis was to investigate the degree of immunogenicity after BS by current CD4 count.
Methods:
In PLWH attending INMI Spallanzani Hospital in Rome, Italy and receiving a BS of BNT162b2 or mRNA-1273 >28 days after a complete mRNA vaccination cycle, immunogenicity was assessed at time of BS (T0) and at day 15 (T1) by anti-RBD CLIA, microneutralization assay [MNA90] and IFNγ production. Participants were stratified by CD4 count at T0 (severe immunodeficiency, SID: <200/mm3; minor immunodeficiency, MID: 200-500/mm3; no immunodeficiency, NID: >500/mm3). Immune response was defined: anti-RBD >7.1 BAU/mL, MNA90 titres >1:10 and IFNγ >12 pg/mL. A paired t-test was used to test overall changes (log2 scale) over T0-T1. ANOVA and truncated regression models were used to compare change in titers from T0 to T1, association between current CD4 count and the lack of immune response was determined by fitting a multivariable logistic regression adjusted for age, time from HIV diagnosis, CD4 nadir, cancer and HIV-RNA a T0.
Results:
We included 216 PLWH on ART (n=76 SID, n=96 MID, n=44 NID): median age 54 yrs (IQR 47-59), median CD4 nadir 45 cell/mm3 (20-122), 93% HIV-RNA <50 c/mL, 7yrs (3-12) since HIV diagnosis and 5yrs (2-8) since AIDS if diagnosed. Participants received BS after a median of 142 (132-156) days from second dose. Response rate was 95.5% in SID, 100% in MID, 100% in NID for anti-RBD (p=0.02); 86.3%, 97.9% and 98.7% for nAbs (p=0.002), and 70%, 95.6% and 97.2% for IFNγ (p<0.0001). Overall we observed a significant increase of BS immunogenicity [anti RBD: mean Log2 4.5 (SD 1.9),p<0.0001; nAbs: 3.7 (2.2),p<0.0001; IFNγ: 0.77 (2.9),p=0.0003]. However, there was no evidence for a difference in mean change of humoral immunogenicity, anti-RBD, nAbs and IFNγ changes by CD4 count groups (Figure 1 A-C). A current CD4 count <200 cell/mm3 was not associated with the risk of failing to elicit neutralizing and cell-mediated response by logistic regression (Figure1D).
Conclusion:
A mRNA BS strongly boosted humoral response in PLWH with advanced disease, regardless of CD4 count at the time of booster. Although clinical implications of the observed immunological response remain uncertain, our data support the usefulness of BS in PLWH with immune dysregulation.


|
| |
|
 |
 |
|
|