 |
 |
 |
| |
Maximizing the potential of Cepheid, GeneXpert PCR rapid testing for hepatitis C through use of finger prick blood samples collected into EDTA capillary tubes
|
| |
| |
EASL 2022 June 22-26 London
Anam Choudhry1, Louise Davies2, Nicki Palmer2, Jade Davies3, Bazga Ali4, Rhys Oakley5, James Plant6, Louise Evans6, Chinlye Chng6, Helen Thompson-Jones6, Lorraine Wall7, Sarah Nicholas7,
Brendan Healy4,8. 1Cardiff University School of Medicine, Cardiff, United Kingdom; 2Public Health Wales Cardiff, Cardiff, United Kingdom; 3Public Health Wales Swansea, Swansea, United Kingdom; 4University Hospital Wales, Cardiff, United Kingdom; 5Cardiff and Vale Pharmacy, United Kingdom; 6Swansea Bay UHB BBV Team, Swansea, United Kingdom; 7Cardiff and Vale UHB BBV Team, Cardiff, United Kingdom; 8Public Health Wales, United Kingdom

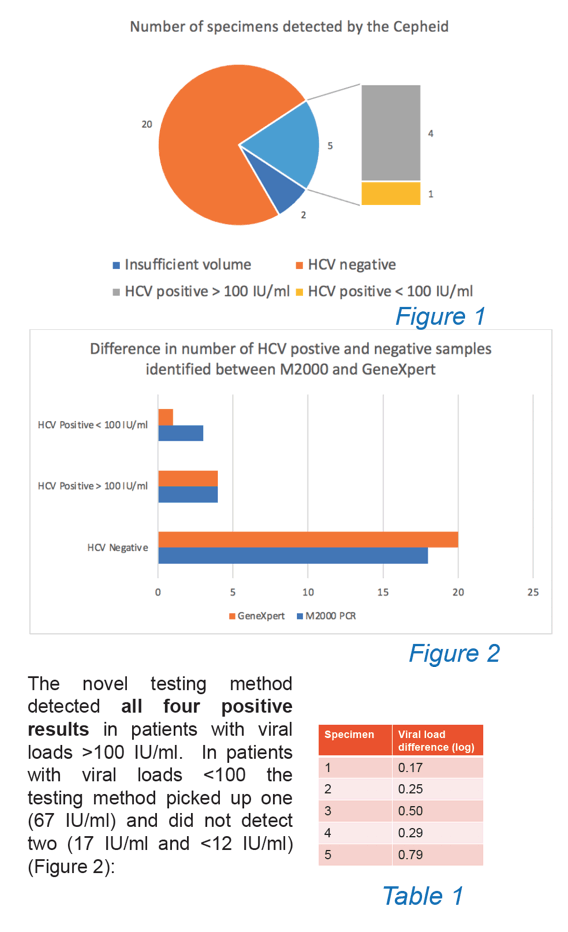
Background and aims: The Cepheid, GeneXpert provides hepatitis C virus (HCV) PCR results <90 minutes. A major limitation is the need to load finger prick samples <15 minutes, as patients must be co-located with the machine. Venous blood collected into EDTA tubes can be stored for up to 72 hours at 2-8°C prior to processing. In this project finger-prick blood collected into EDTA capillary tubes and processed >15 minutes after collection was compared with venepuncture (gold standard) and dried-blood spot testing (DBST) to facilitate testing remote from the machine (e.g. prison clinics on different wings remote from a centrally located machine) but still delivering rapid turn-around times with testing in close proximity to the point of need.




|
| |
|
 |
 |
|
|