 |
 |
 |
| |
Integrated efficacy analysis of 24-week data from two phase 2 and one phase 3 clinical trials of bulevirtide monotherapy given at 2 mg or 10 mg dose level for treatment of chronic hepatitis delta
|
| |
| |
One Third With HDV Respond to Bulevirtide Monotherapy Through 24 Weeks
EASL International Liver Congress 2022, London, June 22-26, 2022
Mark Mascolini
Pooled 24-week results from three phase 2 or 3 trials showed that one third of participants receiving 2 or 10 mg of subcutaneous bulevirtide once daily attained an undetectable HDV RNA or at least a 100-fold drop in HDV RNA IU/mL from baseline and alanine aminotransferase (ALT) normalization [1]. Response rates proved consistent across most subgroups.
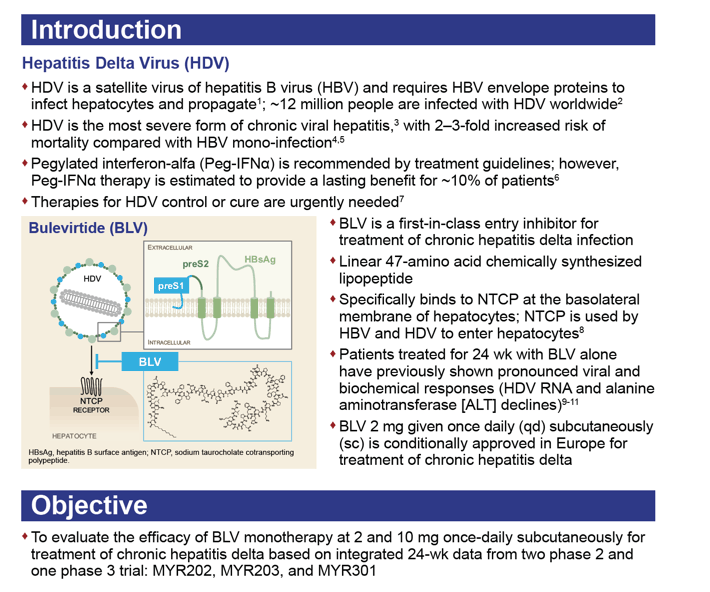
Appropriating hepatitis B virus (HBV) envelope proteins to infect liver cells, HDV is the most severe form of chronic viral hepatitis, increasing death risk 2- to 3-fold over HBV infection alone. Working as an entry inhibitor, bulevirtide has received provisional clearance from European regulators at a subcutaneous dose of 2 mg once daily for chronic HDV infection.
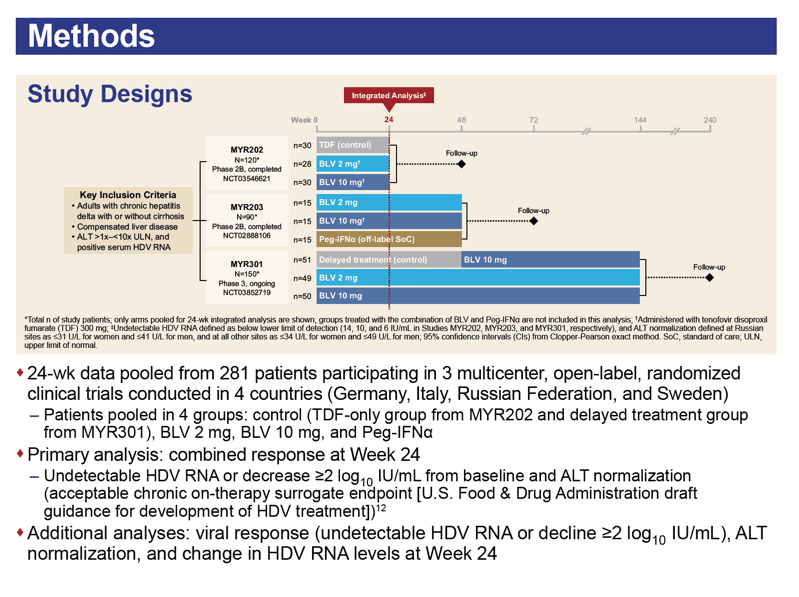
This analysis combined 24-week data from two phase 2 trials and one phase 3 trial of bulevirtide monotherapy at 2 or 10 mg once daily in Germany, Italy, Russia, and Sweden. Participants had to have chronic HDV infection (with or without cirrhosis), compensated liver disease, ALT between 1 and 10 times the upper limit of normal, and positive serum HDV DNA. In the three trial comparison arms, participants got tenofovir, PEG-IFNα, or bulevirtide delayed by 48 weeks. The primary efficacy analysis pooled 24-week data on undetectable HDV RNA or a 100-fold or greater drop in viral load from baseline and a return to normal ALT.
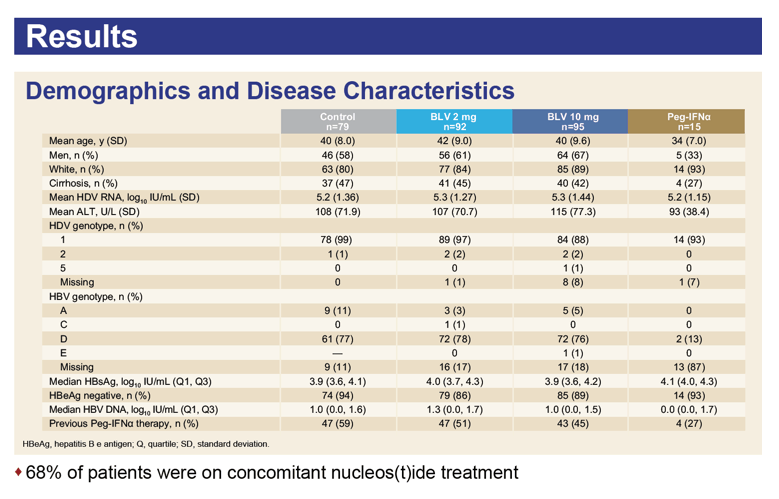
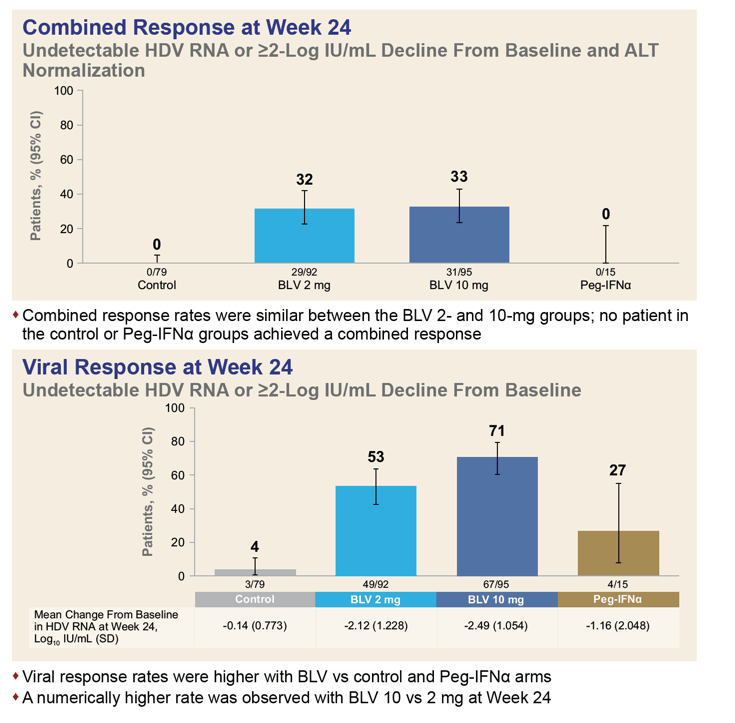
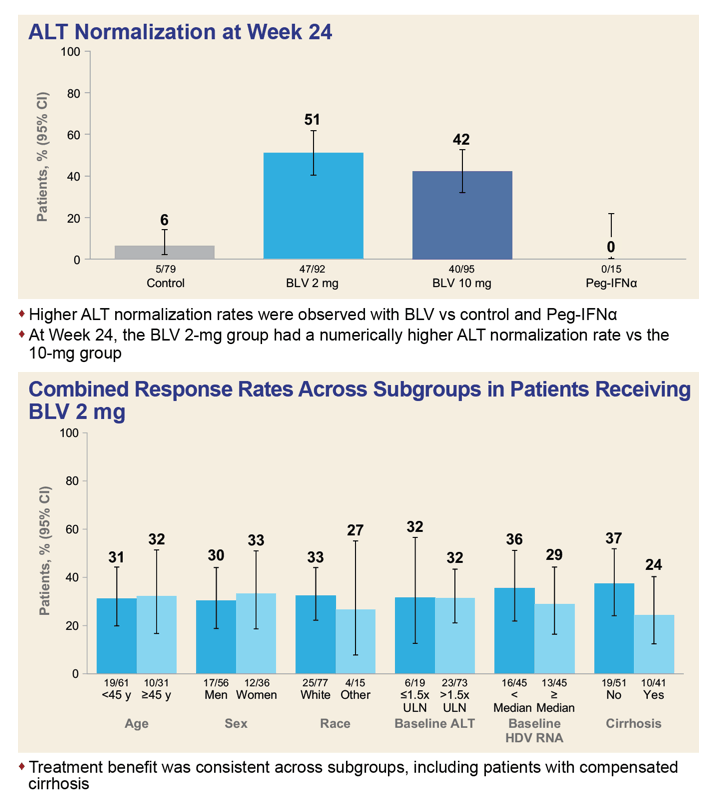
The combined data set involved 92 people receiving 2 mg of bulevirtide daily, 95 getting 10 mg, 79 control participants, and 15 receiving PEG-IFNα. Two thirds of participants also took nucleos(t)ides. At week 24 one third of participants (32%) getting 2 mg of bulevirtide daily and one third getting 10 mg (33%) had attained a protocol-defined response. No one in the comparison arms had a response.
When the endpoints excluded ALT normalization, response rates improved to 53% with 2 mg of bulevirtide, 71% with 10 mg of bulevirtide, 27% with PEG-IFNα, and 4% in other control arms. Looking only at ALT normalization at week 24, researchers recorded respective response rates of 51%, 42%, 6%, and 0%.
The investigators also figured response rates to 2 mg of bulevirtide daily in six subgroups. Those rates differed hardly at all by age (under 45 or older), sex, race (white or other), baseline ALT (above or below 1.5 times the upper limit or normal), and baseline HDV RNA (above or below the median value). A slightly higher proportion of participants without versus with baseline cirrhosis had a response at 24 weeks (37% vs 24%).
Reference
1. Lampertico P, Alernan S, Blank A, et al. Integrated efficacy analysis of 24-week data from two phase 2 and one phase 3 clinical trials of bulevirtide monotherapy given at 2 mg or 10 mg dose level for treatment of chronic hepatitis delta. EASL International Liver Congress 2022, London, June 22-26, 2022. Abstract SAT351.
----------------------

Pietro Lampertico,1,2 Soo Aleman,3 Antje Blank,4 Pavel Bogomolov,5 Vladimir Chulanov,6 Nina Mamonova,6 Viacheslav Morozov,7 Olga Sagalova,8 Tatyana Stepanova,9 Vithika Suri,10 Dmitry Manuilov,10 Lei Ye,10 John F. Flaherty,10 Anu Osinusi,10 Markus Cornberg,11 Maurizia Brunetto,12 Heiner Wedemeyer11
1Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico, Milano, Italy; 2 Università degli Studi di Milano; 3Karolinska Universitetss jukhuset/Karolinska Institutet, Stockholm, Sweden; 4Heidelberg University Hospital, Heidelberg, Germany; 5Moscow Regional Research Clinical Institute Named After M.F. Vladimirsky, Moscow, RussianFederation; 6NationalMedical Research Center of Phthisiopulmonology and Infectious Diseases, Moscow; 7Hepatolog, Samara, Russian Federation; 8SouthernUral State Medical University of the Ministry of Health of the Russian Federation, Chelyabinsk; 9Clinic of Modern Medicine, Moscow, Russian Federation; 10Gilead Sciences, Inc., Foster City, California, USA; 11Medizinische Hochschule Hannover, Germany; 12Hepatology Unit, University Hospital of Pisa, Italy
|
| |
|
 |
 |
|
|