 |
 |
 |
| |
Voxilaprevir/Velpatasvir/Sofosbuvir Efficacy in hepatitis C
patients previously treated with direct-acting antiviral agents (DAA)
VOX/VEL/SOF Effective Rescue After DAA Failure-With Limits
|
| |
| |
EASL International Liver Congress 2022, London, June 22-26, 2022
Mark Mascolini
Voxilaprevir/velpatasvir/sofosbuvir (VOX/VEL/SOF) yielded sustained virologic response (SVR) in 94% of 427 people in whom a direct-acting antiviral (DAA) had already failed [1]. But SVR rates were lower in people with liver transplant, cirrhosis, or previous hepatocellular carcinoma (HCC). Researchers who conducted this study recommended identifying people with cirrhosis or previous HCC before trying VOX/VEL/SOF rescue.
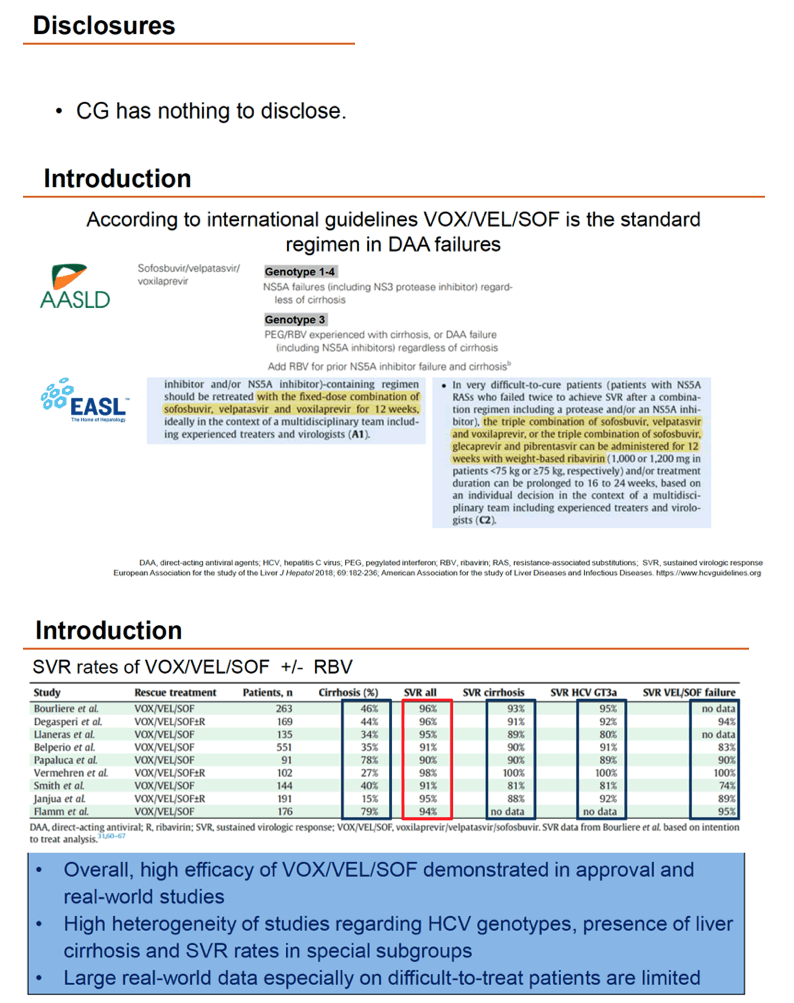
Guidelines from EASL and AASLD both favor fixed-dose VOX/VEL/SOF after DAA failure based on high overall efficacy demonstrated in licensing and real-world studies. But researchers who conducted this analysis noted the SVRs have varied by HCV genotype, presence of cirrhosis, and other factors.
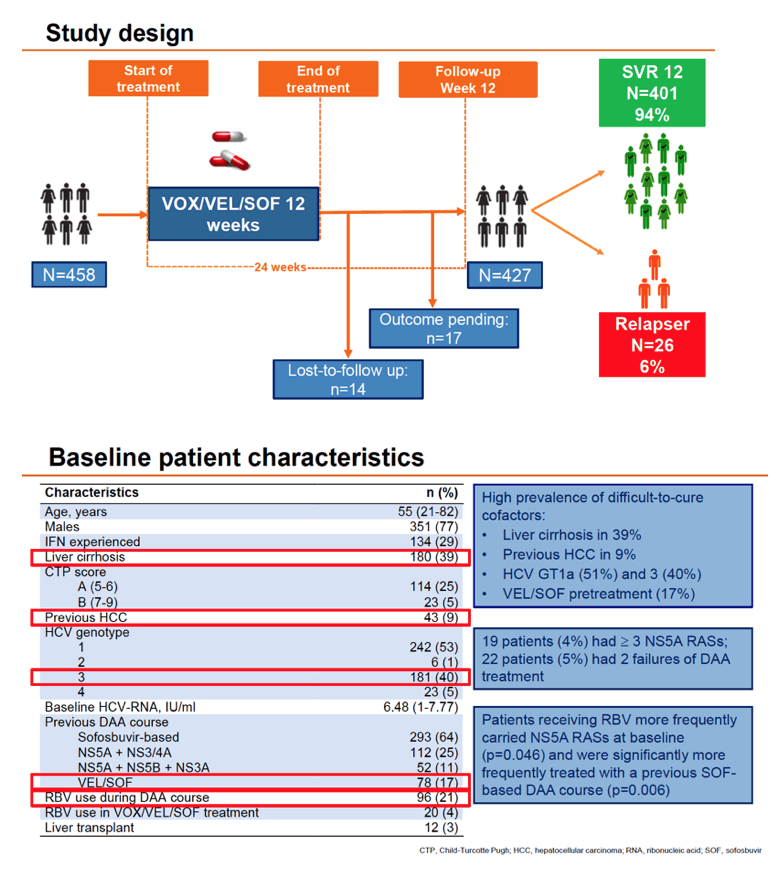
The investigators included adults in the European Resistance Database with HCV infection with or without cirrhosis (Child A or B) in whom 2 or 3 DAAs in an interferon-free regimen had failed. They were treated between May 2015 and November 2020 at 352 centers in Germany, Austria, Switzerland, and Belgium, taking VOX/VEL/SOF with or without ribavirin for 12 weeks. All participants had direct sequencing for baseline and treatment-emergent resistance-associated substitutions (RASs). The European collaborators used multivariate analysis to pinpoint independent predictors of SVR, with a special focus on subgroups identified by HCV genotype 1a or 3, cirrhosis, HCC, VEL/SOF pretreatment, and RASs.
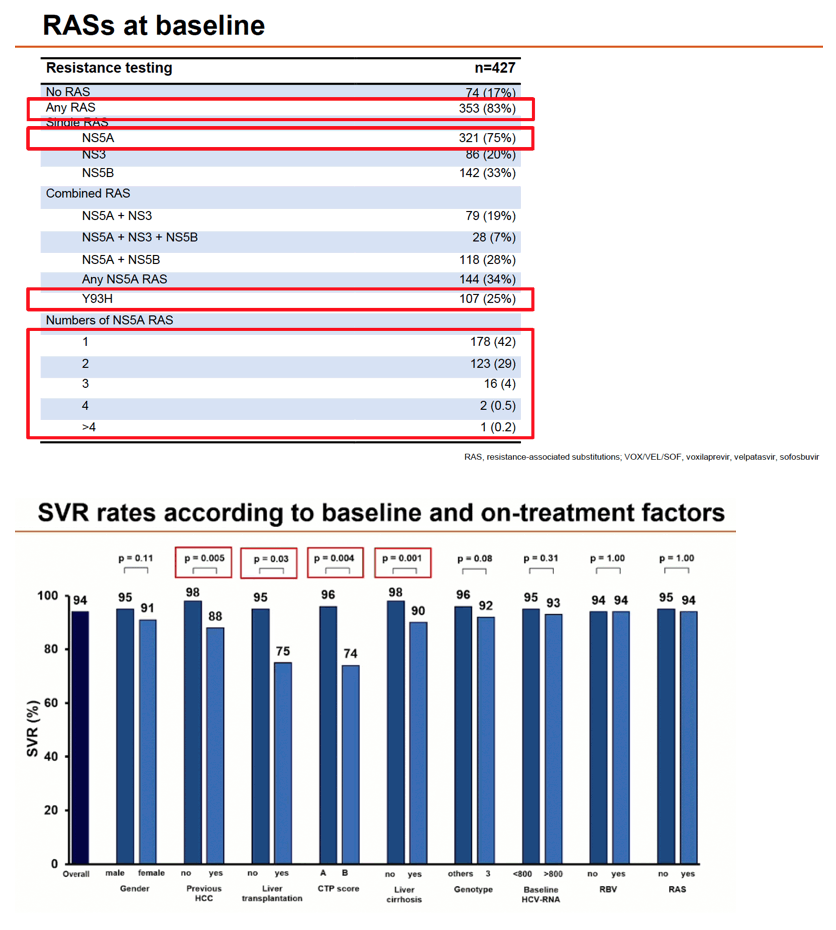
Median age of the 458 participants stood at 55 years, 77% were men, 29% had taken interferon, 64% had taken a SOF-based regimen, and 17% had taken VEL/SOF. While 39% had cirrhosis, 9% had previous HCC. Baseline HCV RNA stood at 6.48 log IU/mL. Rates of hard-to-treat genotypes were 51% for GT1a and 40% for G3. Twenty-two people (5%) had 2 previous DAA failures on their charts, 19 (4%) had 3 or more NS5A RASs, and 353 (83%) had any RAS.
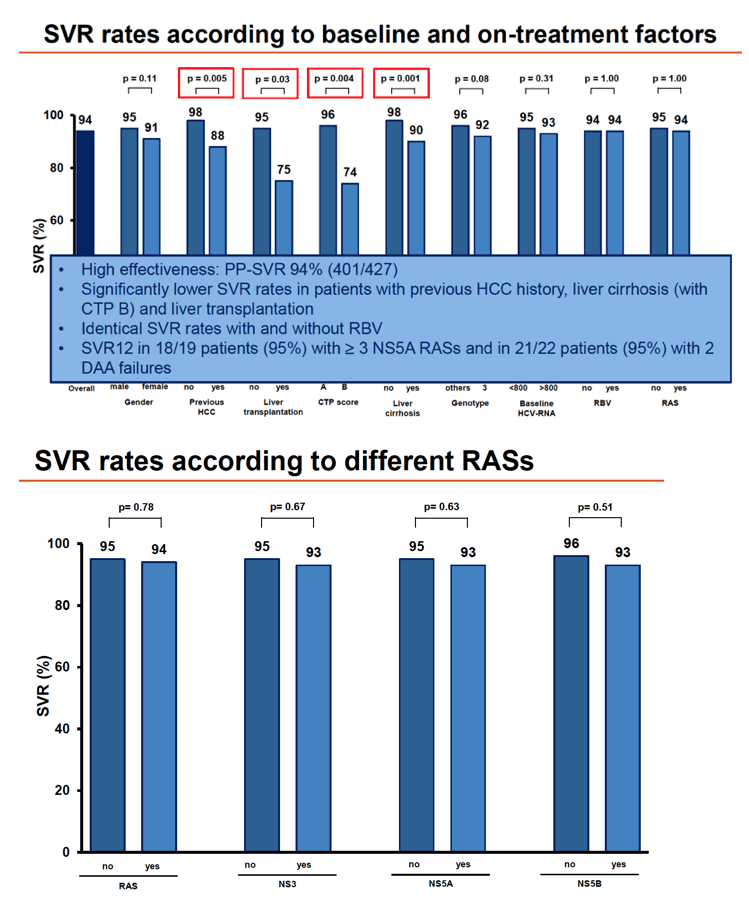
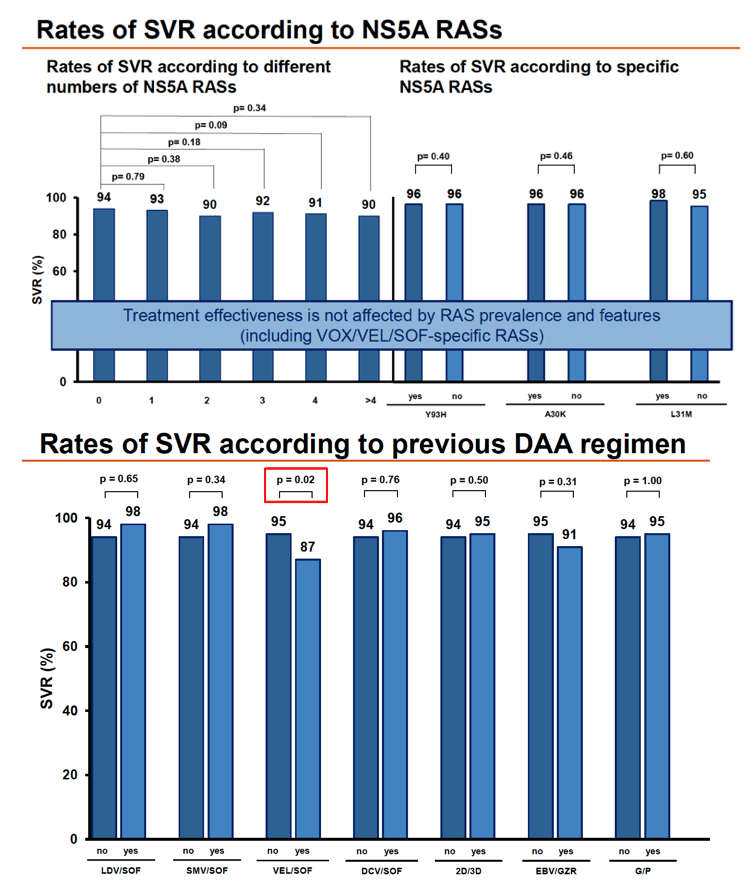
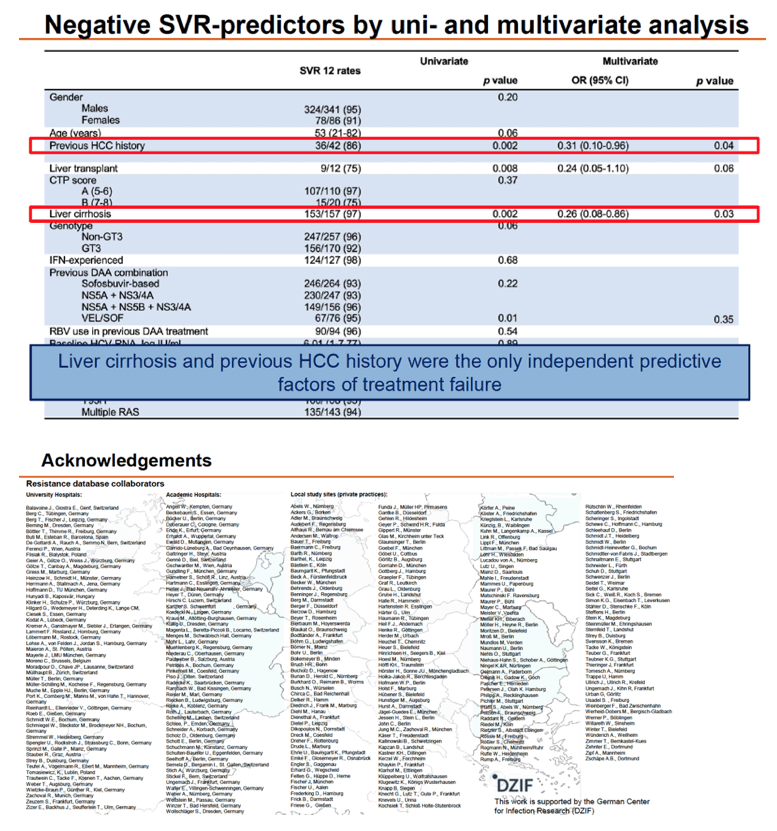
Overall SVR in 427 people analyzed stood at 94%, with no difference by baseline HCV RNA above versus below 800, by ribavirin use, or by RAS detection. SVR rates did differ significantly between people who did versus did not have previous HCC (88% vs 98%, P = 0.005), did versus did not have cirrhosis (90% vs 98%, P = 0.001), did versus did not have liver transplantation (75% vs 95%, P = 0.03), and had CTP score B versus A (74% vs 96%, P = 0.004). SVR rate differences approached significance for females versus males (91% vs 95%, P = 0.11) and for genotype 3 versus other genotypes (92% vs 96%, P = 0.08).
Detection of any RAS or RASs related to NS3, NS5A, or NS5B did not affect SVR rate. Nor did chances of SVR differ significantly by number of NS5A RASs or presence of specific NS5A RASs (Y93H, A30K, or L31M).
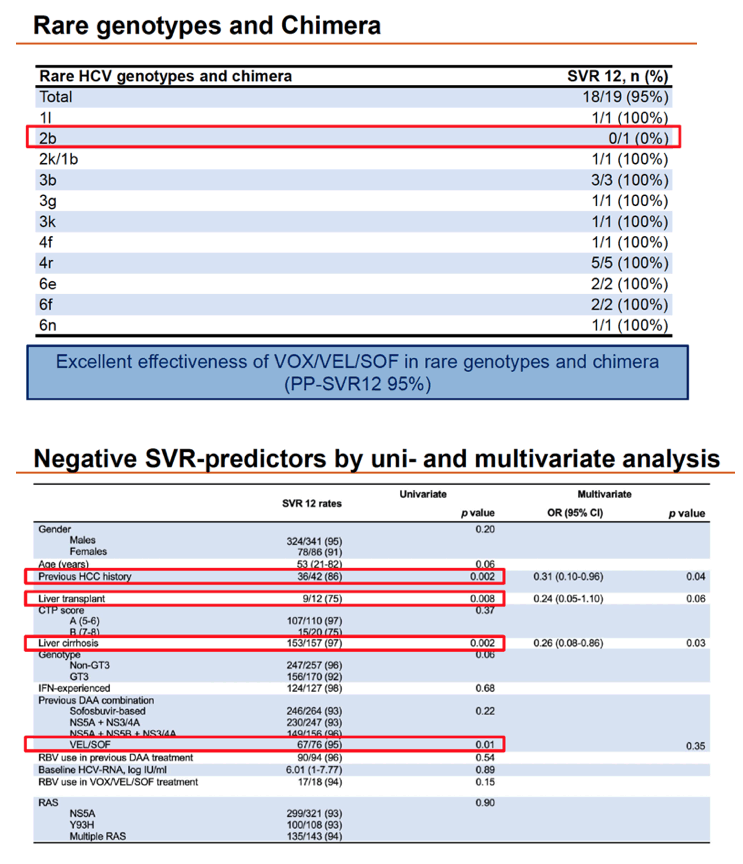
People who had already taken VEL/SOF had a significantly lower SVR rate than those who did not (87% vs 95%, P = 0.02). But chances of SVR did not differ by prior treatment with LDV/SOF, SMV/SOF, DCV/SOF, EBV/GZR, or 2- versus 3-drug regimens. Response rates proved excellent-usually 100%--in people with rare HCV genotypes or chimera (1i, 2k/1b, 3b, 3g, 3k, 4f, 4r, 6e, 6f, and 6n).
Multivariate analysis identified only two independent predictors of failure to attain SVR: previous HCC cut chances about 70% (adjusted odds ratio [aOR] 0.31, 95% confidence interval [CI] 0.10 to 0.96, P = 0.04) and cirrhosis lowered chances about 75% (aOR 0.26, 95% CI 0.08 to 0.86, P = 0.03). There was a trend toward lower odds of SVR in people who had a liver transplant (aOR 0.24, 95% CI 0.05 to 1.10, P = 0.06).
The European team rated overall chances of responding to VOX/VEL/SOF rescue as excellent. SVR rates lagged in people who already took VEL/SOF and in those with previous HCC, liver transplantation, cirrhosis, or a high CTP score.
Reference
1. Graf C, Dietz J, Mulhaupt B, et al. Effectiveness of voxilaprevir/ velpatasvir/ sofosbuvir in hepatitis C patients previously treated with direct-acting antiviral agents (DAA). ASL International Liver Congress 2022, London, June 22-26, 2022. Abstract OS003.
-----------------------


|
| |
|
 |
 |
|
|