 |
 |
 |
| |
Safety, tolerability, pharmacokinetics (PK), and antiviral activity of the 3rd generation capsid inhibitor AB-836 in healthy subjects (HS) and subjects with chronic hepatitis B (CHB)
|
| |
| |
EASL 2022 June 22-26 London
E. Gane1, A Jucov2, L Kolomiichuk3, A Leerapun4, E Sripariwuth5, P Tangkijvanich6, SI Strasser7, T Suttichaimongkol8, C Cooper9, M Elkhashab10, Y-S Lim11, T Eley12, J Brown12, Y King12, T Varughese12, E Medvedeva12, M Chernyakhovskyy12, N Mani13, AG Cole13, S Kultgen13, R Pamulapati13, EP Thi13, T Harasym13, R Dugyala13, S Chaudhari13, M Sofia13, M Child12, G Picchio12, KD Sims12, MF Yuen14
1 Auckland Clinical Studies, New Zealand, 2 Arensia Exploratory Medicine, Moldova, 3 Arensia Exploratory Medicine, Ukraine, 4 Maharaj Nakhon Chiang Mai Hospital, Thailand, 5 Naresuan University Hospital, Thailand, 6 King Chulalongkorn Memorial Hospital, Thailand, 7 Royal Prince Alfred Hospital, Australia, 8 ACRO Khon Kaen University, Thailand, 9 Ottawa Hospital Research Institute, Canada, 10 Toronto Liver Centre, Canada, 11 Asan Institute for Life Science, South Korea, 12 Arbutus Biopharma Clinical Development, Warminster PA, USA, 13 Arbutus Biopharma Discovery and Research, Warminster, PA , USA 14 Queen Mary Hospital, Hong Kong


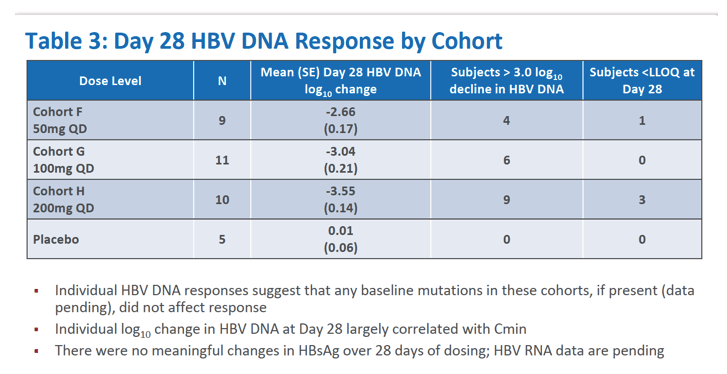
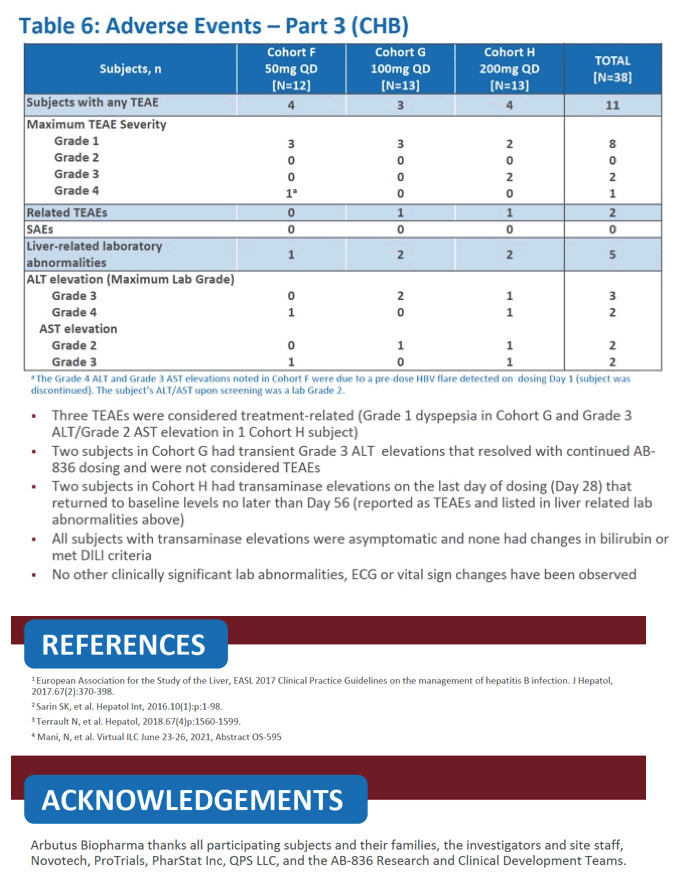

|
| |
|
 |
 |
|
|