 |
 |
 |
| |
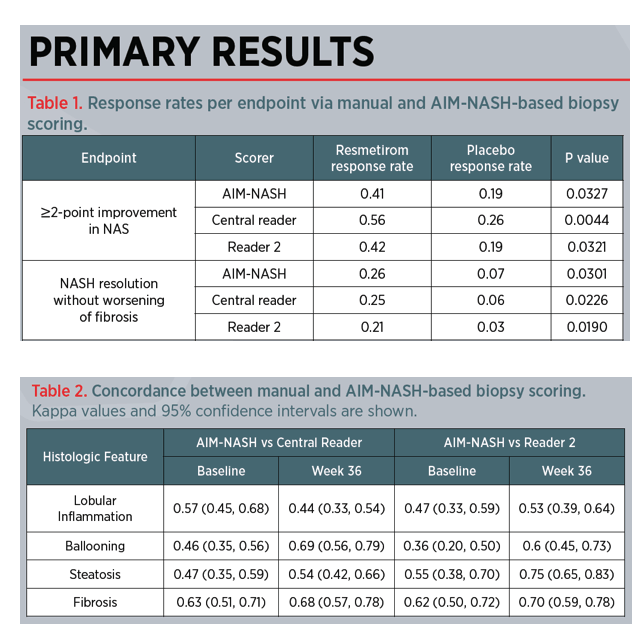
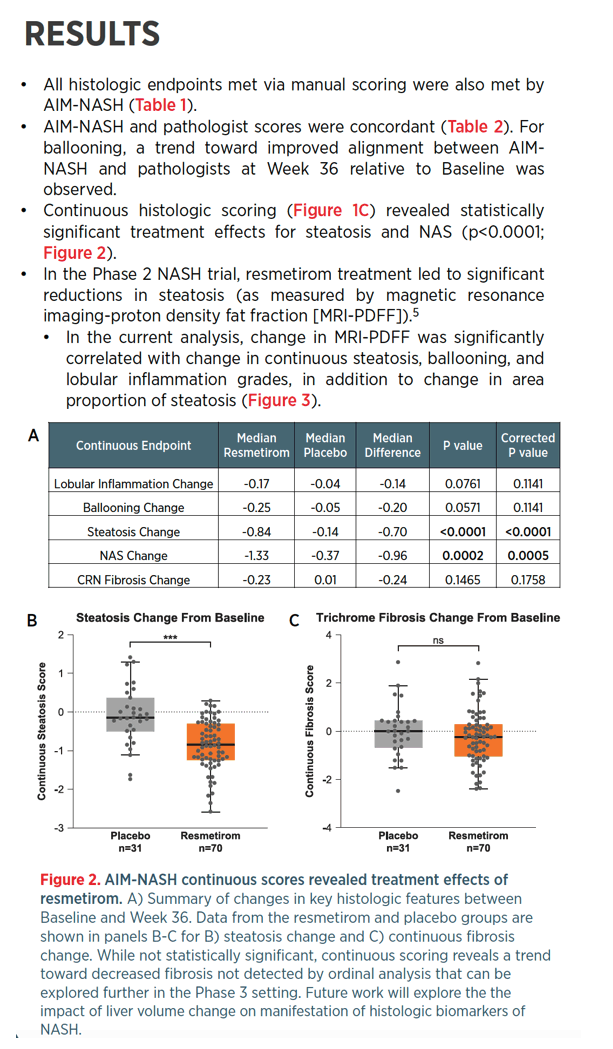
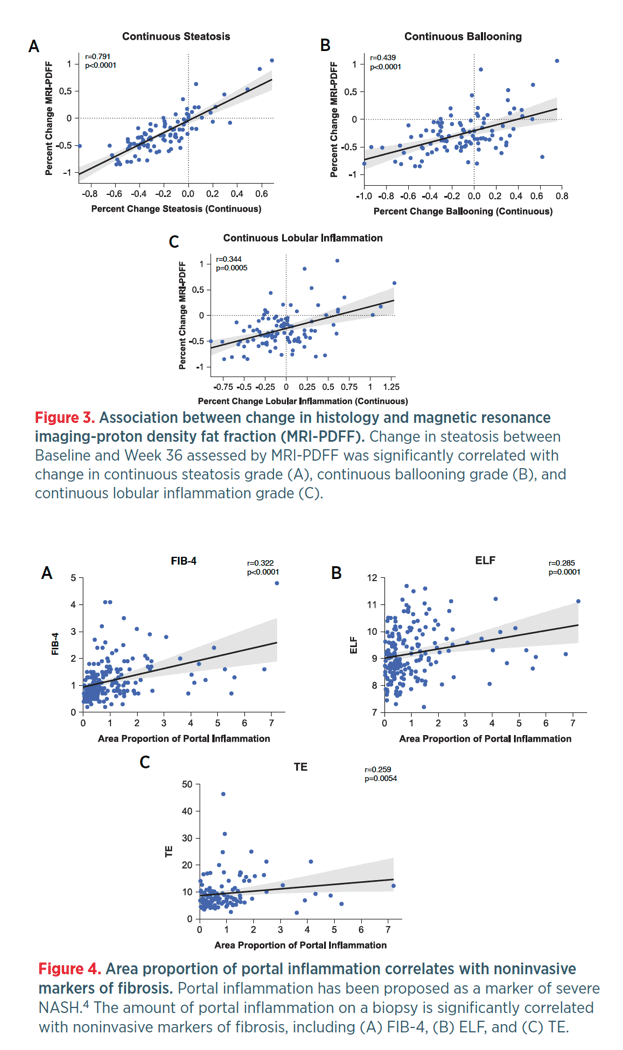
Retrospective AI-based Measurement of NASH Histology (AIM-NASH) Analysis of Biopsies From Phase 2 Study of Resmetirom Confirms Significant Treatment-induced Changes in Histologic Features of Nonalcoholic Steatohepatitis
|
| |
| |
EASL: Madrigal Pharmaceuticals Presents Late-Breaking Phase 3 NASH Data and Multiple Oral Abstracts at EASL's International Liver Congress™ - (06/27/22)
EASL: Primary Data Analyses of MAESTRO-NAFLD-1:a 52-week Double-blind, Placebo-controlled Phase 3 Clinical Trial of Resmetirom in Patients With NAFLD - (06/27/22)
EASL: The Global Burden of Liver Cancer (LC) and Chronic Liver Diseases (CLD) is Driven by Nonalcoholic Steatohepatitis (NASH) and Alcohol Liver Disease (ALD) - (06/27/22)
EASL: Fibroscan-AST (FAST) score predicts liver-related outcomes in 1683 HIV-infected patients at risk for NAFLD - (06/24/22)
EASL 2022 June 22-26 London
Stephen A. Harrison1, Janani S. Iyer2, Pierre Bedossa3, Cynthia Guy4, Charles Biddle-Snead2, Sara Hoffman2, Quang Le2, Victoria Mountain2, Murray Resnick2, Katy Wack2, James K. Hennan5, Rebecca A. Taub5 1Pinnacle Clinical Research, San Antonio, TX, USA; 2PathAI Inc., Boston, MA, USA; 3LIVERPAT, Paris, France; 4Duke University Health System, Durham, NC, USA; 5Madrigal Pharmaceuticals, Conshohocken, PA, USA



|
| |
|
 |
 |
|
|