| |
Lonafarnib-A new member of the Delta Force? Editorial
|
| |
| |
June 2022 Christopher A. Dietz1Markus Cornberg
Download the PDF here
Chronic HDV requires the envelope (HBsAg) of HBV to enter the hepatocyte, spread intrahepatically, and disseminate. Chronic HDV is the most severe form of viral hepatitis. The risk of HCC and liver cirrhosis associated with chronic HBV is further deteriorated by coinfection with HDV, resulting in a relevant health burden.[1] However, HBV treatment with nucleoside analogues (NAs) has no effect on HDV replication,[2, 3] which explains the unmet need for HDV-specific treatment.
Until the recent past, the only therapeutic option for chronic HDV infection was interferon alfa (IFN) or pegylated IFN (PEG-IFN). Treatment of viral hepatitis with IFN is not a specific direct antiviral therapy, but relies on the immune modulation triggered by IFN, which eventually leads to cure. The efficacy of IFN treatment is limited, with approximately 25%-30% achieving virologic response (HDV RNA-negative) after 48 or 96 weeks of therapy, although relapses may occur during the follow-up period (even years later) (reviewed in Sandmann and Cornberg[4]). Nevertheless, long-term follow-up of patients infected with HDV revealed that the risk of developing clinical endpoints such as hepatic decompensation was lower in patients treated with IFN-based therapy than in patients receiving NAs alone.[5]However, due to the side effects of IFN therapy, its use is limited, and IFN is not specifically approved for the treatment of chronic HDV.
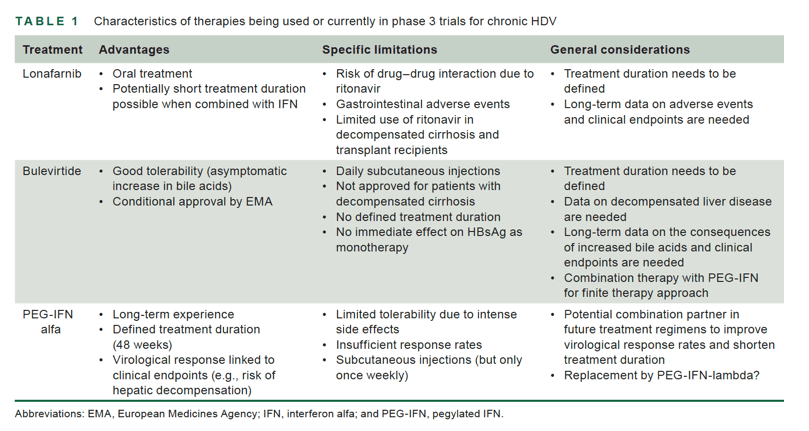
Thus, to date, no specific medical treatment for this severe form of viral hepatitis has been fully approved by the relevant authorities. However, with the conditional approval of bulevirtide by the European Medicines Agency in 2020, an important step has been taken toward the treatment of chronic HDV infection. Bulevirtide is a myristoylated peptide derived from the pre-S domain of the HBsAg that inhibits HDV entry into hepatocytes by blocking the bile acid cotransporter NTCP.[6] Preliminary approval of bulevirtide was based on promising results from phase 2 clinical trials, but these have not yet been fully published (data summarized in Refs. 4, 7). In a nutshell, bulevirtide was investigated in the combination with tenofovir or PEG-IFN, and clinical endpoints such as alanine aminotransferase (ALT) normalization or HDV-RNA decline were achieved more frequently in those treated with bulevirtide (data summarized in Refs. 4, 7). The safety profile appears to be excellent, and the described increase in bile acids is asymptomatic and not associated with clinical findings to date. Data from larger phase 3 studies are expected in mid 2022. However, certain limitations must be considered when treating with bulevirtide. Clinically important long-term endpoints such as prevention of HCC, decompensation, liver transplantation, or liver-related death have not been investigated. In addition, treatment endpoints that determine treatment duration remain unclear. Long-term observational studies are needed to verify that an effect on these clinical endpoints can be achieved and that the therapy does not have side effects in the long-term course. In addition, administration by daily subcutaneous injection for a long period of time may not be tolerated by all patients and could lead to compliance issues in daily practice.
Some of these limitations (Table 1) may be overcome by the development of additional new drugs such as lonafarnib. Lonafarnib is an oral farnesyltransferase or prenylation inhibitor that was initially developed to treat progeria, a rare pediatric disease. Glenn et al. discovered that inhibition of prenylation of the large hepatitis delta antigen is a target to interfere with hepatitis D viral particle formation.[8] In a proof-of-concept study published in 2015, Koh et al. showed that HDV viral load decreased significantly in a dose-dependent manner in patients receiving lonafarnib.[9] However, the therapy with lonafarnib is associated with gastrointestinal adverse events such as nausea, vomiting and diarrhea, which are equally dose dependent. To achieve higher drug level, and thus optimal efficacy with lower doses of lonafarnib, the cytochrome P450-3A4 inhibitor ritonavir can be used as already demonstrated in the LOWR-1-study.[10]
In this issue of Hepatology, Yurdaydin et al. present the results of the LOWR-2 trial, which assessed the effect of ritonavir-boosted lonafarnib in different dosing regimens with and without PEG-IFN. The study was designed as a single-center, open-label, uncontrolled trial. A total of 55 patients were enrolled and assigned to three main treatment groups. Two groups were treated for 12 or 24 weeks with different doses of lonafarnib/ritonavir as all-oral regimen. In the remaining group, the combination of ritonavir-boosted lonafarnib with subcutaneous PEG-IFN was investigated. The primary endpoint was a ≥ 2-log decline in HDV RNA or suppression below the lower limit of quantification at the end of treatment. Normalization of ALT levels was one of the secondary endpoints. Another important objective of this study was to evaluate treatment regimens with good overall tolerability, allowing for further investigation in larger, placebo-controlled phase 3 trials.
The authors concluded that the combination of ritonavir-boosted lonafarnib with PEG-IFN was associated with a better virologic response than the all-oral treatment regimens. Seven of the 9 patients (78%) who completed a 24-week therapy with PEG-IFN plus lonafarnib/ritonavir achieved the primary endpoint, while all experienced ALT normalization. This is an important finding that is currently being further investigated in the D-LIVR phase 3 trial (NCT03719313). To date, the duration of treatment with lonafarnib has not been established, and a very long duration of lonafarnib therapy may not be easy to implement, given the side-effect profile. Therefore, a short and finite therapy with PEG-IFN plus lonafarnib would certainly be an important option. Nevertheless, viral rebound may still occur after treatment discontinuation but may result in a transient ALT increase followed by HDV-RNA negativity and ALT normalization, suggesting that positive immune responses are induced. This was observed in 3 patients in the LOWR-2 study and similarly in 2 patients in the LOWR-1 study.[10] This stop-to-flare concept certainly needs further investigation. As observed in previous studies with lonafarnib, gastrointestinal symptoms accounted for the largest proportion of adverse events.[10] Because tolerability was another key endpoint of the presented study, the authors show detailed information on the occurrence of related adverse events. Discontinuation due to adverse events occurred in 7 patients. Weight loss was used as a surrogate marker for gastrointestinal symptoms. Because relevant weight loss occurred more frequently with therapies using high doses of lonafarnib, the investigators decided to exclude high doses from future trials. A new observation was that patients treated with lonafarnib had an asymptomatic increase in platelets. Some of the patients also showed a decrease in hemoglobin, but the authors could not identify a correlation between the changes in platelets and hemoglobin levels. This observation and potential mechanisms certainly need to be further elucidated in further studies. In summary, Yurdaydin et al. provide important insights into potential treatment regimens for lonafarnib, while confirming the drug's antiviral efficacy but also disclosing adverse events of therapy. The findings support the evaluation of lonafarnib in phase 3 trials, as the results of placebo-controlled studies are needed to demonstrate the safety and efficacy of lonafarnib on a larger scale. The data also support further investigation of the combination with PEG-IFN, the drug we would like to avoid in the future as discussed previously. However, if high response rates can be achieved with short-term treatments, this would still be an important option. Meanwhile lonafarnib has also been tested in combination with PEG-IFN-lambda, another drug candidate for treating chronic HDV infection. Type III interferon receptors are expressed primarily in hepatocytes and to a lesser extent in hematopoietic cells and central nervous system cells. Therefore, IFN-lambda therapy should be associated with less side effects, which normally limit the use of interferon alfa (reviewed in Refs. 4, 7).
The study by Yurdaydin et al. presented in this issue of Hepatology illustrates how intensive research into the treatment of chronic HDV infection has led to the fortunate situation that several promising antiviral agents are on their way into clinical application. In the future, it will be of great importance to see to what extent long-term endpoints such as liver-related death, risk of HCC, or need for liver transplantation are affected by antiviral therapy. Surrogate endpoints on viral response are needed before long-term clinical data are available. It remains to be seen to what extent the association between virologic response and clinical endpoints, as described for treatment with PEG-IFN,[5] will translate to emerging HDV-specific treatments. Hopefully, lonafarnib will find its way into the therapeutic landscape of chronic HDV and contribute to the "Delta Force." Once several treatments are in clinical use, it will also be of interest to combine different therapies with a different mode of action.
|
|
| |
| |
|
|
|