| |
Efficacy of prolonged tenofovir therapy
on hepatitis delta in HIV-infected patients
|
| |
| |
AIDS 2014 Vincent Sorianoa,b, Eugenia Vispoa, Rocio Sierra-Enguitaa,Carmen de Mendozac, Jose V. Ferna Andez-Monteroa,Pablo Labargaa and Pablo Barreiroa,b
Download the PDF here
In summary, thanks to the unique opportunity represented by a population of HIV patients with delta hepatitis, we investigated the long-term effect of tenofovir, which exhibits dual antiviral effect against both HIV and HBV, and was marketed as an antiretroviral agent 10 years ago. Serum HDV-RNA steadily declined in all patients receiving tenofovir and became undetectable in more than a half of them by 5 years. This virological outcome resulted in significant improvements in hepatic fibrosis as measured by transient elastography, with regression of cirrhosis in five out of nine patients. In our knowledge, this study is the first to provide robust evidence in favour of a clinical benefit of oral antivirals on chronic hepatitis delta. In the absence of current, more well tolerated medications, we propose that tenofovirshould be considered as a therapeutic option for HDV infection, acknowledging its benefit would mainly be recognized after prolonged exposure and perhaps not in all patients.
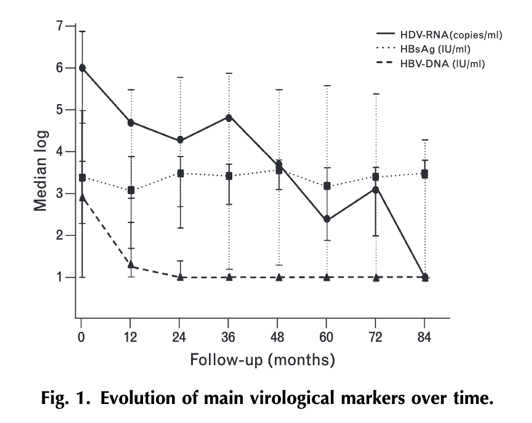
In our study, we report outcomes for viral markers and histological estimates in a relatively large series of chronic hepatitis delta patients treated with tenofovir and followed for a median of nearly 5 years. Complete HBV suppression was achieved in all patients along with significant reductions in HDV replication (with clearance of HDV-RNA in more than a half) along with improvements in liver fibrosis (with regression of cirrhosis in five out of 10). In our knowledge, this is the first study that demonstrates unquestionably a clinical benefit of oral antivirals on chronic hepatitis delta. On the basis of these data, we propose that tenofovir should be considered as a therapeutic option for HDV infection, being aware that its benefit would mainly be recognized in the long term and perhaps not in all patients.
Our results suggest that the benefit of tenofovir on delta hepatitis is most likely the result of an indirect effect that follows the potent antiviral activity of the drug on HBV. Reduction and/or suppression of HDV-RNA occurred over months and followed complete HBV-DNA suppression in all patients. However, concentrations of serum HBsAg did not follow the same trend and remained relatively stable during the whole length of the study.
Similar unaffected kinetics for HBsAg in patients treated with tenofovir have been reported by others [34-39], which argues against a benefit of tenofovir on delta hepatitis throughout reducing the production of the HBV envelope protein. However, we did not check whether a dysbalance in the release of distinct HBsAg fractions under prolonged tenofovir therapy had occurred in our patients. Hypothetically, a reduction in the production of the large HBsAg isoform on long-term tenofovir treatment could impair HDV infectivity of new hepatocytes and in this way ultimately result in HDV-RNA suppression [40].
It is noteworthy that the improvement in liver fibrosis was significantly linked to the achievement of complete suppression of serum HDV-RNA. Although almost all our patients with delta hepatitis experienced declines in serum HDV-RNA on long-term tenofovir, liver fibrosis did not improve in those who did not completely suppress HDV-RNA, suggesting that hepatic damage in delta patients is mainly driven by HDV replication rather than by HBV infection.
We could not identify any predictor of the achievement of HDV-RNA suppression in our series. Whereas HBV-DNA became negative soon after introducing tenofovir in all patients, the median time to reach negative HDV-RNA in the 10 individuals who cleared HDV was 54 months. This observation suggests that any benefit of tenofovir on delta hepatitis would be manifest only after extended periods.
Abstract
Background:
Hepatitis delta virus (HDV) produces the most severe form of chronic viral hepatitis. We explored whether prolonged tenofovir exposure might be beneficial on hepatitis delta in HIV-infected patients.
Methods:
All HIV-infected patients with hepatitis delta followed at our institution since year 2000 were retrospectively examined. Serum HBV-DNA and HDV-RNA were quantified using commercial assays. Liver fibrosis was measured using elastometry.
Results:
A total of 19 HIV/delta patients were identified. All were viremic for HDV and 11 for HBV. After a median tenofovir exposure of 58 months, all had undetectable HBV-DNA and 10 (53%) had undetectable HDV-RNA. The median drop in HDV-RNA in the remaining nine HDV viremic patients at the end of follow-up was 2.4 log copies/ml. A reduction above 30% in liver stiffness occurred in six out 10 (60%) patients who achieved undetectable HDV-RNA, whereas hepatic stiffness did not change in the remaining HDV viremic patients (P = 0.03). Serum HBsAg concentrations did not decline significantly, although HBsAg seroclearance occurred in three patients, all of whom became negative for HDV-RNA.
Conclusion:
Long-term exposure to tenofovir significantly reduced serum HDV-RNA apart from completely suppressing HBV-DNA in HIV-infected patients with hepatitis delta. This virological benefit is accompanied by significant improvements in liver fibrosis.
Introduction
Hepatitis D virus (HDV) is a small unique subviral particle that only infects individuals with hepatitis B virus (HBV) infection [1,2]. More than 15 million persons are estimated to be infected with HDV worldwide [3,4]. HDV transmission overlaps HBV mechanisms, contagion being mainly the result of sexual and parenteral exposures [5]. Large HBV vaccine campaigns in adults and universal HBV vaccination of children in Western countries have translated in dramatic declines of hepatitis delta, being immigration from endemic countries the most frequent source of current HDV cases [6-8].
Infection with HDV produces the most severe form of chronic viral hepatitis [9,10]. There is no specific antiviral treatment for hepatitis delta, although peginterferon alpha given for at least 1 year may improve liver enzymes and reduce viremia [11,12]. This benefit, however, is generally only transient as HDV replication resumes thereafter in most treated patients [13-16]. New hopes for hepatitis delta patients rely on the use of peginterferon lambda, which is better tolerated than alpha, and prenylated inhibitors [17-19].
As HDV replication requires HBsAg and tenofovir is amongst the most potent anti-HBV agents and is widely used as part of many HIV therapeutic combinations, we explored whether prolonged tenofovir exposure might be of benefit on hepatitis delta in HIV-coinfected patients.
HDV genotype 1 was found in all cases. HBV genotypes D and A were the most common (53 and 26%, respectively).
All patients at different time points began tenofovir therapy (time 0 for our analysis). Prior lamivudine exposure (median 59 months) before beginning tenofovir was recognized in nine (47%), being the rest naive to any anti-HBV agent.
Although there was an initial decline in serum HBsAg following initiation of tenofovir therapy, it was not sustained over time (Fig. 1). Overall, there were no significant changes in serum HBsAg concentrations during the whole study period, although HBsAg seroclearance occurred in three patients. Median baseline HDV-RNA was lower in these three individuals than in the rest [2 (1-5.1) vs. 5.8 (4.3-6.7) log copies/ml]. Interestingly, these three patients became all negative for serum HDV-RNA.
|
|
| |
| |
|
|
|