| |
Safety and efficacy of REP 2139 and pegylated interferon alfa-2a for treatment-naive patients with chronic hepatitis B virus and hepatitis D virus co-infection (REP 301 and REP 301-LTF): a non-randomised, open-label, phase 2 trial
|
| |
| |
September 27, 2017
Download the PDF here
Summary
Background
REP 2139 clears circulating hepatitis B virus (HBV) surface antigen (HBsAg), enhancing the restoration of functional control of HBV infection by immunotherapy. We assessed the safety and efficacy of REP 2139 and pegylated interferon alfa-2a in patients with chronic HBV and hepatitis D virus (HDV) co-infection.
Methods
In this open-label, non-randomised, phase 2 trial, patients aged 18-55 years, who were treatment naive, hepatitis B e antigen [HBeAg] negative, anti-hepatitis D antigen [HDAg] positive, and HDV RNA positive, with serum HBsAg concentrations of more than 1000 IU/mL, and a history of HDV infection for 6 months or more before treatment, were recruited at Toma Ciorbă Hospital of Infectious Diseases in Chișinău, Moldova. Patients were excluded if they had HDV superinfection, liver infections other than HBV and HDV, or liver cirrhosis. Patients received 500 mg intravenous REP 2139 once per week for 15 weeks, followed by combined therapy with 250 mg intravenous REP 2139 and 180 μg subcutaneous pegylated interferon alfa-2a once per week for 15 weeks, then monotherapy with 180 μg pegylated interferon alfa-2a once per week for 33 weeks. The primary endpoints assessed at the end of treatment were the safety and tolerability of the treatment regimen, analysed in the intention-to-treat population. Secondary outcomes included the proportion of patients with serum HBsAg less than 50 IU/mL, the proportion of patients with suppressed HBV DNA, and the proportion of patients who maintained these responses through follow-up. The REP 301 trial is registered with ClinicalTrials.gov, number NCT02233075. We also did an additional follow-up at 1 year after the end of treatment, as an interim analysis of the REP 301-LTF trial (planned duration 3 years), registered with ClinicalTrials.gov, number NCT02876419, which is ongoing but not recruiting patients.
Findings
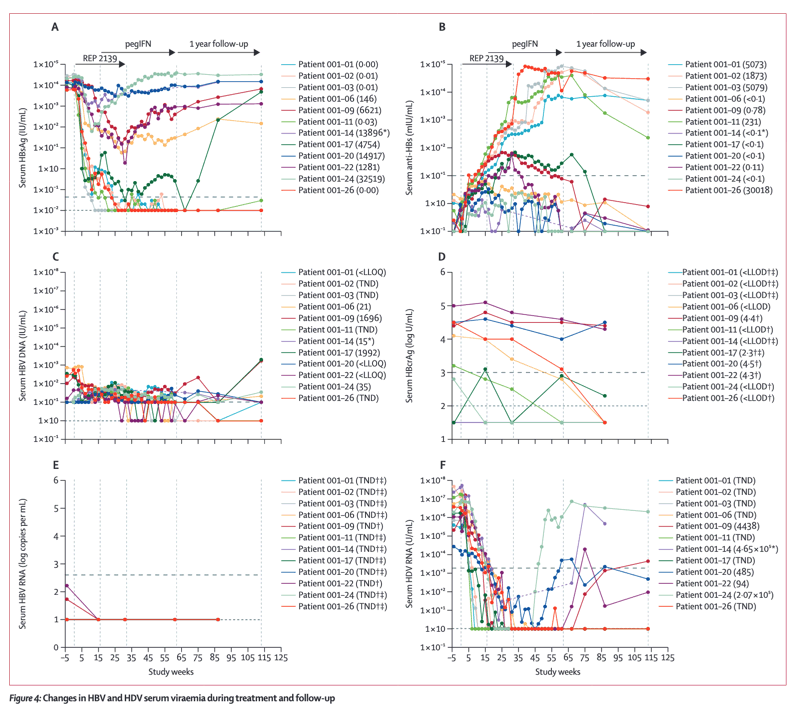
Between Sept 8, 2014, and Jan 27, 2015, we enrolled 12 patients into the REP 301 study. All 12 patients experienced at least one adverse event during treatment: two (17%) patients experienced anaemia, eight (67%) neutropenia, and ten (83%) thrombocytopenia. Five (42%) patients had raised alanine aminotransferase levels, four (33%) had raised aspartate aminotransferase levels, and two (17%) had increased bilirubin concentrations. Four (33%) patients had a serious adverse event, and 12 (100%) patients had treatment-emergent lab abnormalities. Six patients had HBsAg levels less than 50 IU/mL by the end of treatment (all <0⋅05 IU/mL); five maintained this level of suppression at the end of 1 year follow-up. Six patients had hepatitis B surface antibody (anti-HBs) titres above 10 mIU/mL at the end of treatment (five had maximum anti-HBs concentrations of 7681-86 532 mIU/mL during treatment), which were maintained at the end of 1 year follow-up in these five patients. Elevated alanine and aspartate aminotransferase concentrations and profound elevations of anti-HBs titres were restricted to patients who had HBsAg levels of less than <1 IU/mL before the introduction of pegylated interferon alfa-2a. Nine patients had suppressed HBV DNA (<10 IU/mL]) at the end of treatment, which was maintained by seven patients and newly established in an eighth patient at the end of 1 year follow-up. 11 patients became HDV RNA negative during treatment, with nine remaining HDV RNA negative at the end of treatment; seven of these patients remained HDV RNA negative by the end of 1 year follow-up. By the end of 1 year follow-up, normalisation of serum aminotransferases occurred in nine of 12 patients.
Interpretation
Combined REP 2139 and pegylated interferon alfa-2a therapy is safe, well tolerated, and establishes functional control of HBV and HDV co-infection and normalisation of serum aminotransferases in a high proportion of patients 1 year after therapy. This combination therapy approach might provide a new treatment option for patients with HBV and HDV co-infection.
Funding
Replicor.
|
|
| |
| |
|
|
|