| |
Global Hepatitis D 37% Prevalence in IVDUs
|
| |
| |
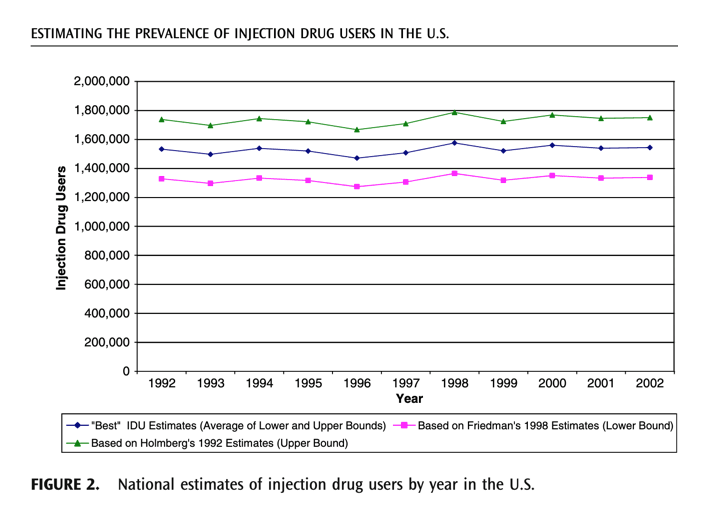
Based on graph below there might be 300,000-400,000 IVDUs with Hep D in USA ?????
Download the PDF here
Gilead Submits Biologics License Application to U.S. Food and Drug Administration for Bulevirtide, an Investigational Treatment for People Living With Chronic Hepatitis Delta - (05/26/22)
PREVALENCE OF HEPATITIS DELTA VIRUS (HDV) INFECTION IN THE UNITED STATES: RESULTS FROM AN ICD-10 REVIEW - 25% of HDV in USA is In NYC (Bklyn, Bronx) & NYC Suburb - (06/06/22) 11.8% of patients with chronic HBV may be HDV infected. Only 4.7% of chronic HBV patients (2.5 mill in USA?) are tested for HDV - 100,000+.
HDV infects up to 72 million people worldwide and is associated with an increased risk of severe and rapidly progressive liver disease.
From 2001-2016, the anti-HBc positivity prevalence was 19.7% (95% confidence interval [CI] 16.0-24.0%) among those with IDU histories, compared with 4.6% (95% CI 4.3-5.0%) in the general population. The HBV surface antigen positivity prevalence was 0.4% (95% CI 0.3-0.5%) in the general population.
We estimate that there are 15⋅6 million (95% uncertainty interval [UI] 10⋅2-23⋅7 million) PWID aged 15-64 years globally, with 3⋅2 million (1⋅6-5⋅1 million) women and 12⋅5 million (7⋅5-18⋅4 million) men. Gender composition varied by location: women were estimated to comprise 30⋅0% (95% UI 28⋅5-31⋅5) of PWID in North America and 33⋅4% (31⋅0-35⋅6) in Australasia, compared with 3⋅1% (2⋅1-4⋅1) in south Asia. Globally, we estimate that 17⋅8% (10⋅8-24⋅8) of PWID are living with HIV, 52⋅3% (42⋅4-62⋅1) are HCV-antibody positive, and 9⋅1% (5⋅1-13⋅2) are HBV surface antigen positive; there is substantial geographic variation in these levels. Globally, we estimate 82⋅9% (76⋅6-88⋅9) of PWID mainly inject opioids and 33⋅0% (24⋅3-42⋅0) mainly inject stimulants. We estimate that 27⋅9% (20⋅9-36⋅8) of PWID globally are younger than 25 years, 21⋅7% (15⋅8-27⋅9) had recently (within the past year) experienced homelessness or unstable housing, and 57⋅9% (50⋅5-65⋅2) had a history of incarceration. https://www.thelancet.com/journals/langlo/article/PIIS2214-109X(17)30375-3/fulltext
Seroprevalence was 10.58% (95% CI 9.14 to 12.11) in mixed population without risk factors of intravenous drug use (IVDU) and high-risk sexual behaviour (HRSB). It was 37.57% (95% CI 29.30 to 46.20) in the IVDU population and 17.01% (95% CI 10.69 to 24.34) in HRSB population.- Past or present HBV infection was highest among non-Hispanic Asian adults (21.1%) compared with non-Hispanic white (2.1%), non-Hispanic black (10.8%), and Hispanic (3.8%) adults, and was greater among adults born outside of the United States than those who were U.S. born. https://www.cdc.gov/nchs/products/databriefs/db361.htm
The seroprevalence of HDV among HBsAg-positive persons in the USA was estimated to be 5.9% (95% CI 3.0-9.8%) [1], but the metanalyses highlighted that US data were sparse. A recent analysis of the 2011-2016 National Health and Nutrition Examination Survey (NHANES), which is representative of the noninstitutionalized U.S. civilian population, reported an anti-HDV prevalence of 42% among hepatitis B surface antigen positive carriers [5], a rate that was substantially higher than previous estimates from NHANES covering the period 1999-2012 [6]. While a true increase in HDV seroprevalence in the USA is possible, other explanations include assay variability and differences in patient sampling. Of note, the DiaSorin anti-HDV ELISA assay has lower specificity in comparison with the quantitative microarray antibody capture (QMAX) assay [7].
Based on current epidemiologic data, the American Association for the Study of Liver Diseases (AASLD) guidance on hepatitis B recommends risk-based screening for HDV. The "at risk" groups include persons who inject illicit drugs, persons with HIV, men who have sex with men, those at risk for sexually transmitted diseases, and persons who emigrated from countries of high endemicity [8].
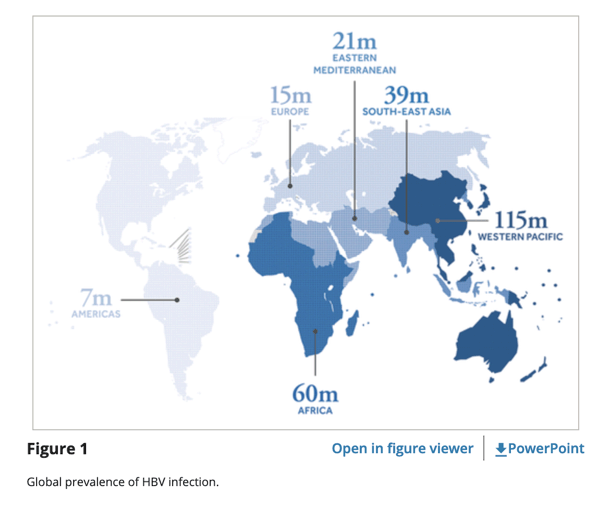
Worldwide, ∼248 to 292 million people are chronically infected with HBV [3], [4]. Based on these estimations, ∼15 to 20 million of these patients were initially thought to be also affected by HDV [5]. These figures were challenged by a recent meta-analysis, proposing that a staggering 62-72 million people may live with HDV worldwide [6],
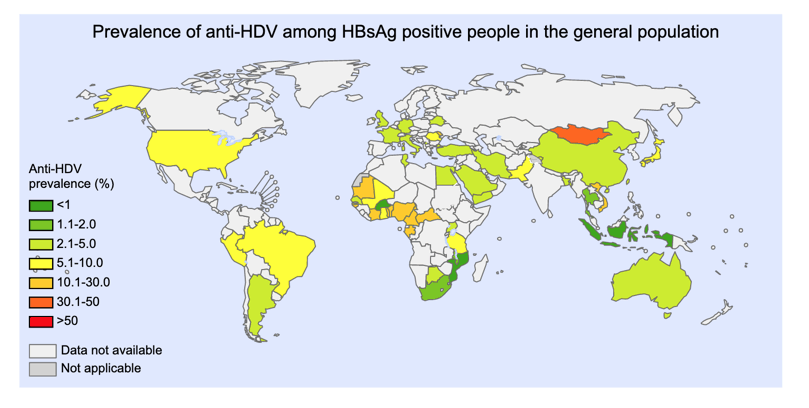
The global prevalence of hepatitis D virus infection: Systematic review and meta-analysis
• HDV infection is common among HBsAg-positive people worldwide.
• Among HBsAg-positive people, estimated HDV prevalence is 4.5% (95% CI 3.6-5.7).
• HDV prevalence in HBsAg-positive hepatology clinic attendees is 16.4% (14.6-18.6).
• HDV prevalence is higher in people who inject drugs and who have HCV or HIV.
• HDV causes an estimated 18% of cirrhosis and 20% of HCC associated with hepatitis B.
The proportion of cirrhosis attributable to HBV varies geographically according to HBV epidemiology, ranging from 6% in North America, to 6-21% in Latin America, 34-38% in sub-Saharan Africa, and 39% in East Asia.[3] Worldwide, HBV is associated with a third of deaths from HCC. [4]
Worldwide, ∼248 to 292 million people are chronically infected with HBV [3], [4]. Based on these estimations, ∼15 to 20 million of these patients were initially thought to be also affected by HDV [5]. These figures were challenged by a recent meta-analysis, proposing that a staggering 62-72 million people may live with HDV worldwide [6], a prevalence almost two-times greater than that of human immunodeficiency virus (HIV) infection (estimated to infect 36.9 million persons in 2017, according to the World Health Organization).
(6) Prevalence and burden of hepatitis D virus infection in the global population: a systematic review and meta-analysis - The pooled prevalence of HDV among population with HBsAg-positive was twofold as what was estimated before. The prevalence of HDV among two special groups (intravenous drug users and people who have high-risk sexual behaviour) was both higher than the mixed population https://gut.bmj.com/content/68/3/512 Results From a total of 2717 initially identified studies, only 182 articles from 61 countries and regions met the final inclusion criteria. The overall prevalence of HDV was 0.98% (95% CI 0.61 to 1.42). In HBsAg-positive population, HDV pooled prevalence was 14.57% (95% CI 12.93 to 16.27): Seroprevalence was 10.58% (95% CI 9.14 to 12.11) in mixed population without risk factors of intravenous drug use (IVDU) and high-risk sexual behaviour (HRSB). It was 37.57% (95% CI 29.30 to 46.20) in the IVDU population and 17.01% (95% CI 10.69 to 24.34) in HRSB population.
Conclusion We found that approximately 10.58% HBsAg carriers (without IVDU and HRSB) were coinfected with HDV, which is twofold of what has been estimated before. We also noted a substantially higher HDV prevalence in the IVDU and HRSB population. Our study highlights the need for increased focus on the routine HDV screening and rigorous implementation of HBV vaccine programme.
---------------------

2019
Highlights
• Hepatitis D virus is a defective virus, dependent on hepatitis B virus for its assembly.
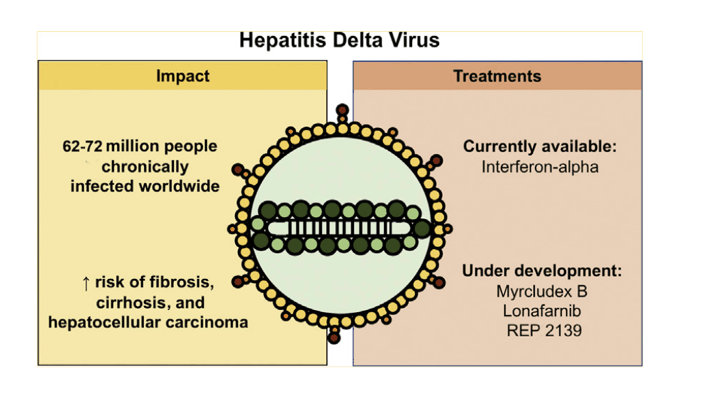
• Hepatitis D virus infection affects 62-72 million people worldwide.
• Chronic hepatitis D is the most severe chronic viral hepatitis.
• Current interferon-based antiviral treatments have dismal efficiency and are poorly tolerated.
• Host-targeting molecules inhibiting the viral life cycle are currently in clinical development.
Abstract
Hepatitis delta virus (HDV) is a defective virus that requires the hepatitis B virus (HBV) to complete its life cycle in human hepatocytes. HDV virions contain an envelope incorporating HBV surface antigen protein and a ribonucleoprotein containing the viral circular single-stranded RNA genome associated with both forms of hepatitis delta antigen, the only viral encoded protein. Replication is mediated by the host cell DNA-dependent RNA polymerases. HDV infects up to72 million people worldwide and is associated with an increased risk of severe and rapidly progressive liver disease. Pegylated interferon-alpha is still the [Myrcludex approved as bulevirtide in Europe; FDA approval request submitted in USA] only available treatment for chronic hepatitis D, with poor tolerance and dismal success rate. Although the development of antivirals inhibiting the viral replication is challenging, as HDV does not possess its own polymerase, several antiviral molecules targeting other steps of the viral life cycle are currently under clinical development: Myrcludex B, which blocks HDV entry into hepatocytes, lonafarnib, a prenylation inhibitor that prevents virion assembly, and finally REP 2139, which is thought to inhibit HBsAg release from hepatocytes and interact with hepatitis delta antigen. This review updates the epidemiology, virology and management of HDV infection.
|
|
| |
| |
|
|
|