| |
NASH Pipeline
|
| |
| |
Therapeutic pipeline in nonalcoholic
steatohepatitis - PDF
Drugs for Non-alcoholic Steatohepatitis (NASH): Quest
for the Holy Grail - PDF
Abstract
Nonalcoholic fatty liver disease (NAFLD) is a global epidemic that is likely to become the most common cause of chronic liver disease in the next decade, worldwide. Though numerous drugs have been evaluated in clinical trials, most of them have returned inconclusive results and shown poorly-tolerated adverse effects. None of the drugs have been approved by the Food and Drug Administration for treating biopsy-proven non-alcoholic steatohepatitis (NASH). Vitamin E and pioglitazone have been extensively used in treatment of biopsy-proven nondiabetic NASH patients. Although some amelioration of inflammation has been seen, these drugs did not improve the fibrosis component of NASH. Therefore, dietary modification and weight reduction have remained the cornerstone of treatment of NASH; moreover, they have shown to improve histological activity as well as fibrosis. The search for an ideal drug or ‘Holy Grail’ within this landscape of possible agents continues, as weight reduction is achieved only in less than 10% of patients. In this current review, we summarize the drugs for NASH which are under investigation, and we provide a critical analysis of their up-to-date results and outcomes.
Introduction
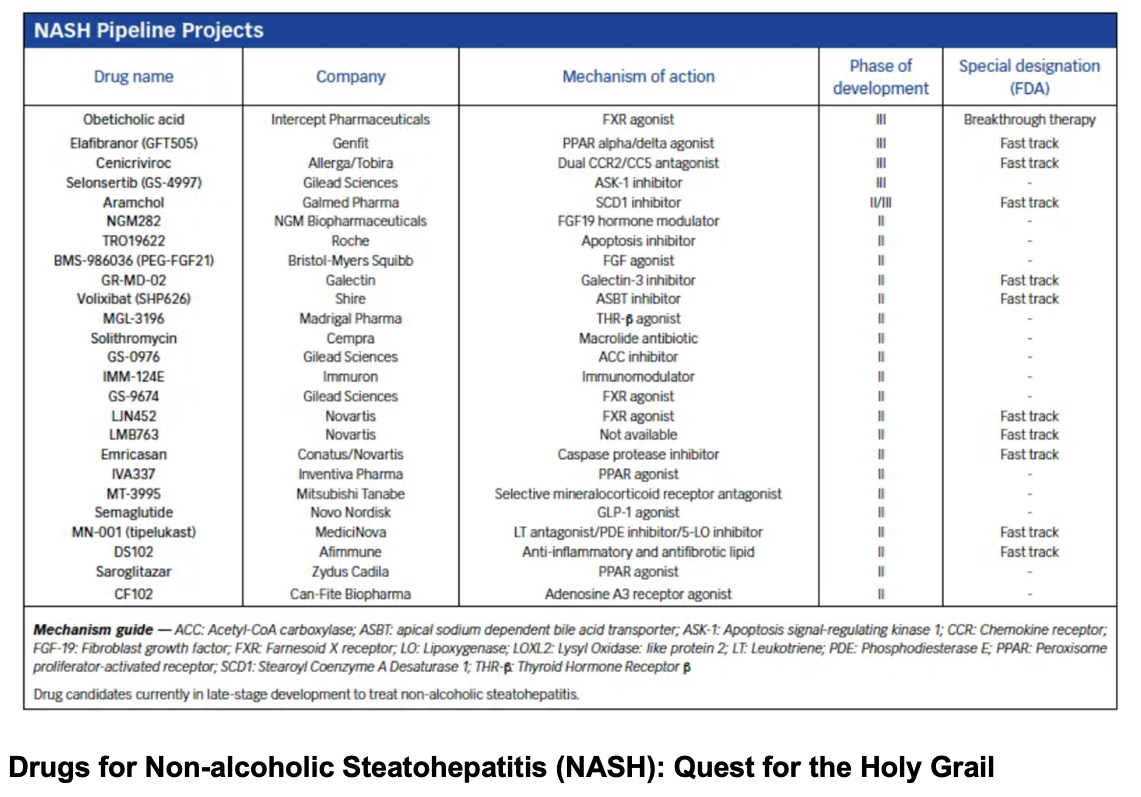
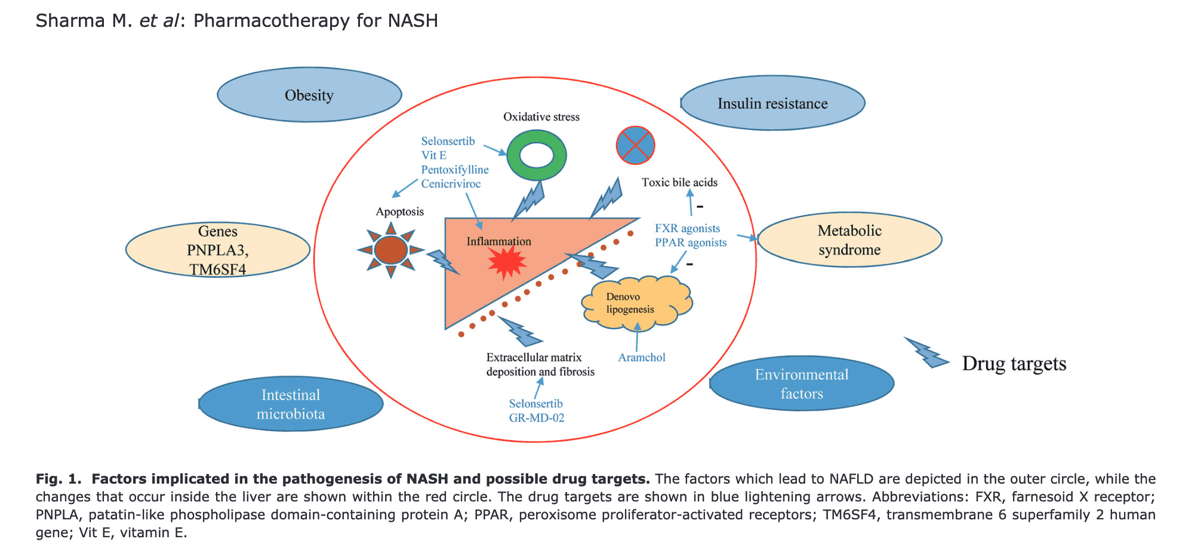
Nonalcoholic fatty liver disease (NAFLD) is an emerging global epidemic. It is the most common cause of chronic liver disease in the western world.1 The pooled prevalence of NAFLD in Asia is 30% (95% confidence interval [CI]: 28.13-31.15).2 The term nonalcoholic steatohepatitis (NASH) was coined almost 4 decades ago.3 Recently, NAFLD was renamed as “metabolic (dysfunction)-associated fatty liver disease”, or “MAFLD”.4 However, the change in terminology is unlikely to alter the management of these patients and hence will not be referred to in this manuscript. In this review, instead, we will focus on the drugs used for NASH. There have been several trials to find an ideal drug for the treatment of NASH.5-10 Despite several advances in the understanding of the pathophysiology underlying this common disease, to date, there are no drugs approved by the Food and Drug Administration (FDA) for the treatment of either simple steatosis or NASH. Recently, FDA has accepted a new drug application (not approved) for OCA, a farnesoid X receptor (FXR) agonist, which was shown to improve fibrosis in NASH.11 In this review, we outline the mechanisms and targets of such, and discuss some of the major drug trials in NASH.
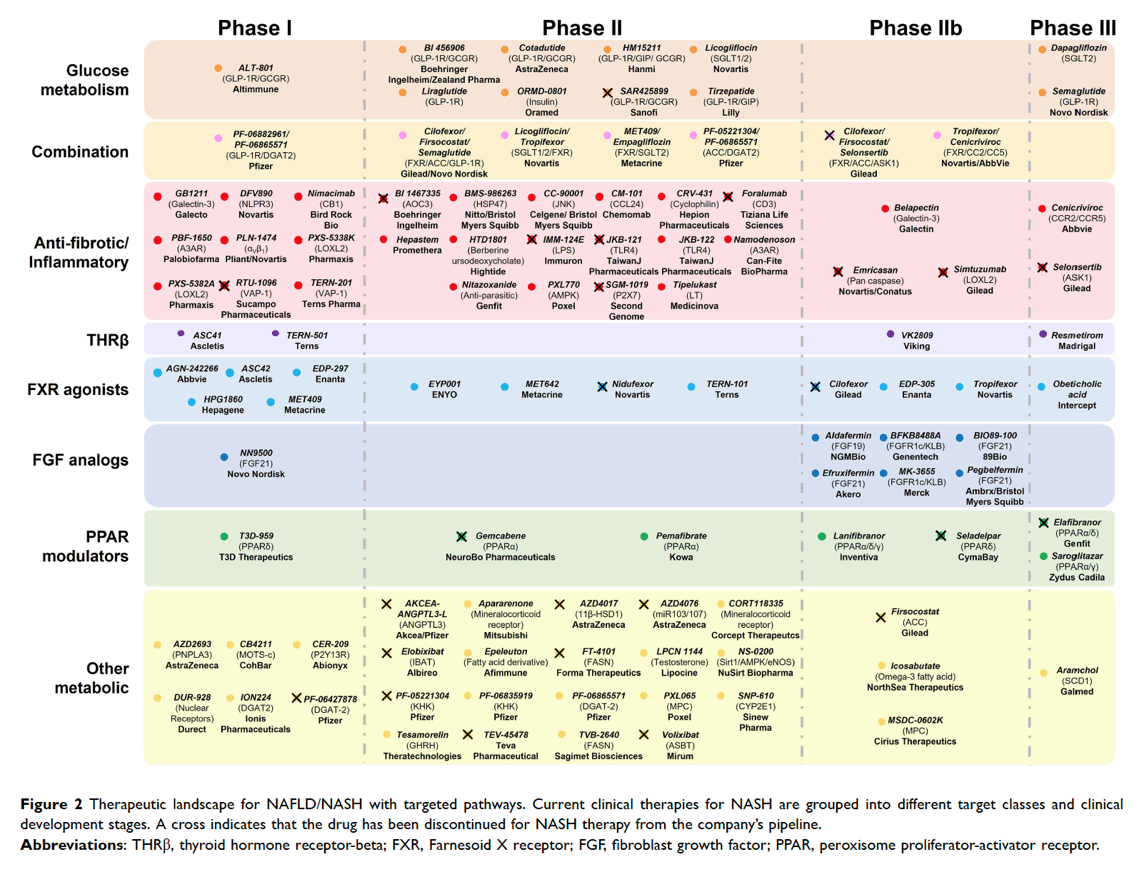
Journey of drugs in NASH
For the approval of a drug for a disease, it needs to undergo preclinical trials, followed by Phase 1 safety studies, which are supported by Phase 2 studies. Phase 2 studies are dose-finding trials and aid in evaluating efficacy of a drug. Once a drug is found to be effective, it undergoes Phase 3 studies that compare the safety and efficacy of the drug with existing therapies. Then, the drug is placed for review before an appropriate drug review committee that may/may not approve the drug.12 Post-approval, there are phase 4 trials, a strategy which is also known collectively as ‘post-marketing surveillance’. The objective of the phase IV studies is to check the drug’s performance in real-life scenarios, to study the long-term risks and benefits of the drug, and to discover any rare side effects. Interestingly, for each phase of the trial, there are recommended endpoints to be achieved before proceeding to the next stage of a trial in adult patients, such as those with NASH.13 However, there are no clear endpoints for pediatric NASH.13 For NASH, the endpoint for a phase 3 trial is NASH resolution (defined as disappearance of ballooning and disappearance or persistence of minimal, lobular inflammation that does not qualify for the diagnosis of NASH), with or without a reduction in fibrosis stage by one point.13
There are certain major hindrances to conducting a clinically relevant trial in NASH.14,15
Some of the limiting factors are:
1. Biopsies are required to define participants and are needed to establish efficacy, as well.
2. Many trials have high screen fail rate, due to stricter inclusion criteria.
3. There is often a high placebo response rate in the control group. The reasons for this are unclear but may be due to behavioral changes in the control group, resulting in weight loss.
4. Most of the studies use two primary endpoints.
o 4a. Improvement in fibrosis, with no worsening of NASH
o 4b. NASH resolution without worsening of fibrosis
5. Challenges in developing clinical endpoints, due to the lack of specific symptoms of NASH.
6. Difficulties in managing confounders, such as recording alcohol intake before and during the study, and the lack of uniformity in diet and physical activity.
Lifestyle changes for the treatment of NASH
The most effective and proven therapy for NASH is weight loss. Analysis of data from eight randomized control trials has shown that >5% weight loss leads to resolution hepatic steatosis, and ≥7% improves the inflammatory score of NAS (NAFLD activity score).26 But, reportedly, only 50% of patients can achieve a weight loss of 7%.27 Lastly, a weight loss of ≥10% results in the resolution of early fibrosis, in approximately 45% of subjects.27
Physical activity of ≥150 m/week is associated with improvement in liver enzymes, irrespective of the weight loss. More than 8,000 steps per day is associated with a reduction in all-cause mortality; however, the number of steps required for reduction in NAS score/liver enzymes has not been evaluated.28
Dietary interventions
A triple hit behavioral phenotype exists, which involves i) sedentary behavior, ii) low physical activity, and iii) poor diet, all of which are well known to be associated with poor cardio-metabolic health, NAFLD, and overall mortality.29 Along with physical activity, a healthy diet is recommended for NAFLD patients.24 Refined sugar and sugar-sweetened beverages are a common source of empty calories.29 These fructose-rich diets increase the hepatic synthesis of triglycerides. Hence, sugar-sweetened beverages should be avoided. Mediterranean diet is primarily plant-based (whole grains, legumes, fruit, vegetables), low in carbohydrates (limited simple sugars and refined carbohydrates), and rich in monounsaturated (mostly olive oil) and omega-3 fats, and which incorporates limited red meat, ad low-fat dairy products, which has been shown to improve the hepatic steatosis and insulin resistance and is the recommended diet for NAFLD.30,31 Recent studies have shown that 1 year of a hypocaloric (750 Kcal/day) diet combined with 200 m of exercise per week has a dose relationship with weight loss and leads to improvement in NASH, including fibrosis.27 The combination of a hypocaloric diet (reduction by 500-1,000 kcal/day) and moderate-intensity exercise would provide the best likelihood of sustaining weight loss over time.24 Currently, the time-tested diet and exercise therapy remains the most effective and cost saving intervention for management of NAFLD.
| |
| |
| |
|
|
|