| |
Association of Direct-Acting Antiviral Therapy With Liver and Nonliver
Complications and Long-term Mortality in Patients With Chronic Hepatitis C
|
| |
| |
Dec 12 2022
Download the PDF here
• In the present study, DAA treatment (vs no treatment) was associated with a large and significant reduction (57%) in all-cause mortality, regardless of age, sex, race and ethnicity, comorbidity, and the presence of alcohol use, cirrhosis, or HCC.
• DAA treatment was independently associated with a 27% lower risk of developing HCC.
• DAA treatment was independently associated with a 64% reduction in the risk of developing liver decompensation (aHR, 0.36.
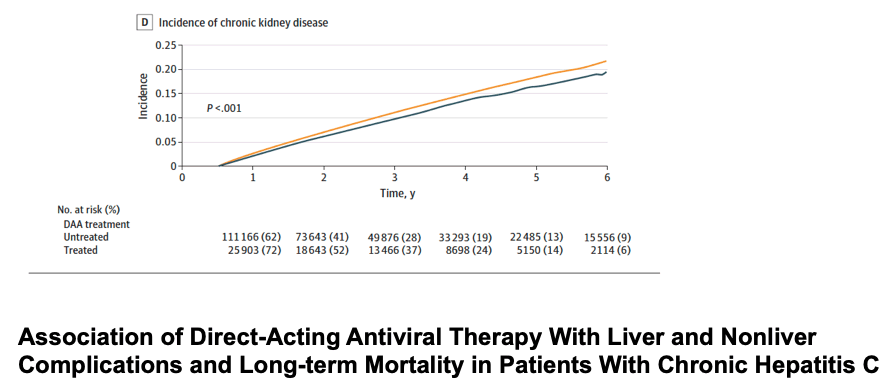
• DAA treatment was associated with a 26% lower risk of developing diabetes (aHR, 0.74; 95% CI, 0.70-0.77; P < .001), a 19% lower risk of CKD (aHR, 0.81; 95% CI, 0.78-0.85; P < .001), a 10% lower risk of CVD (aHR, 0.90; 95% CI, 0.86-0.94; P < .001). There was also an 11% lower risk of nonliver cancers (aHR, 0.89; 95% CI, 0.85-0.94; P < .001) after adjusting for potential confounders despite the lack of a significant difference in the crude incidence of nonliver cancers between treated and untreated patients.
To our knowledge, this is the largest clinical study to date that comprehensively evaluates overall mortality and liver as well as nonliver outcomes associated with DAA treatment for patients with CHC. Of note, a significant reduction in mortality was found, even for patients without cirrhosis, a population previously considered to receive less benefit from an HCV cure than patients with cirrhosis. In these study findings, DAA treatment was associated with a reduced risk of both liver and extrahepatic outcomes, ie, HCC, liver decompensation, diabetes, CKD, CVD, nonliver cancer, and ultimately, overall survival. Because HCV treatment with a DAA regimen is well tolerated for nearly all patients, we believe these findings provide further support for universal HCV treatment coverage for all patients affected by HCV.
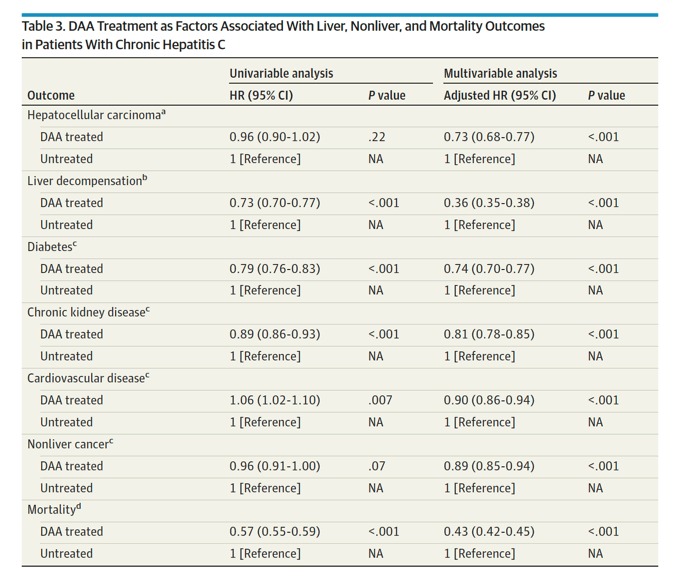
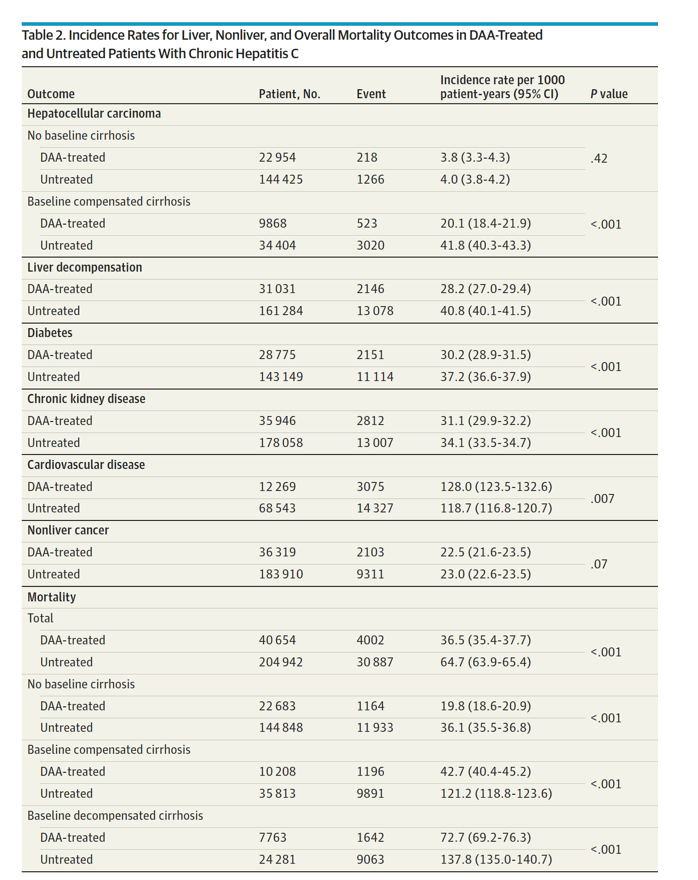
In multivariable regression analysis, DAA treatment was independently associated with a significant decrease in the risk of liver (adjusted hazard ratio [aHR] for HCC, 0.73; decompensation, 0.36),nonliver (aHR for diabetes, 0.74; chronic kidney disease, 0.81; cardiovascular disease, 0.90; nonliver cancer, 0.89), and mortality outcomes (aHR, 0.43).
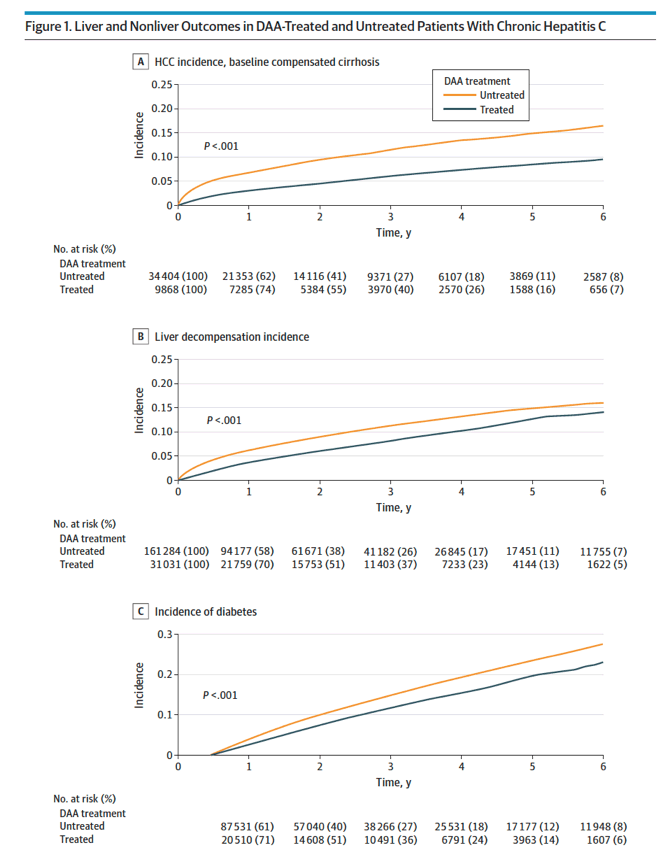
HCC incidence for patients with cirrhosis at baseline was significantly lower (20.1 [95% CI, 18.4-21.9] vs 41.8 [95% CI, 40.3-43.3]; P < .001; Figure 1A and Table 2).
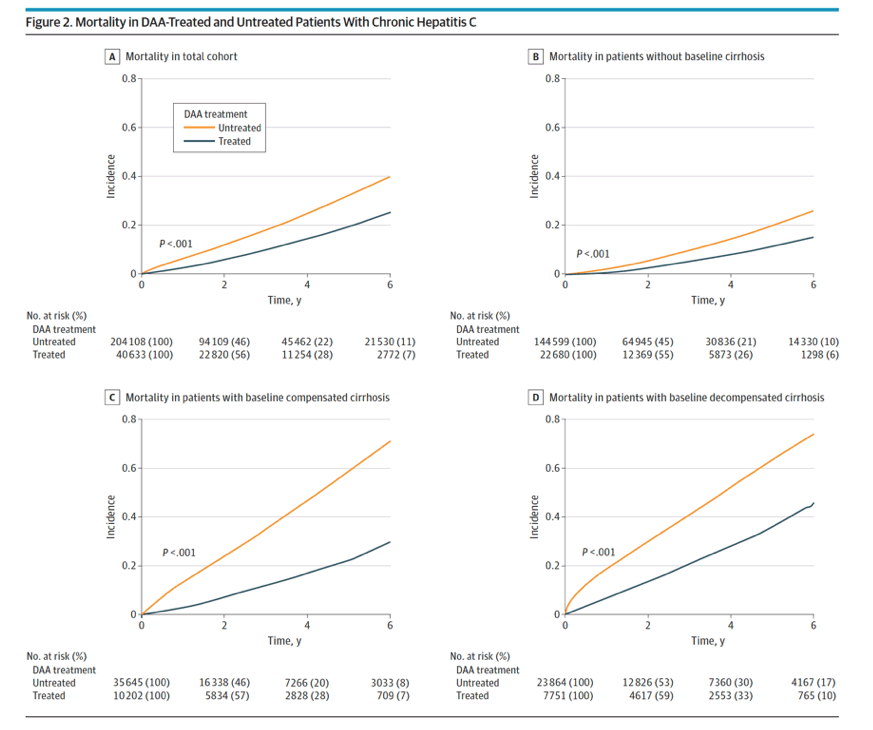
For the total study cohort, the mortality rate per 1000 person-years was twice higher among untreated compared with the DAA-treated patients (64.7 vs 36.5; P < .001; Figure 2A and Table 2).
This was a retrospective cohort study of adults (≥18 years) with HCV infection, using data from the Optum Clinformatics Data Mart (CDM) (Optum Inc) database provided by the Center for Population Health Sciences of Stanford University
Since 2014, major improvements have been made in therapies for hepatitis C virus (HCV), most notably direct-acting antiviral (DAA) agents. Short-course all-oral DAA treatment can achieve a virologic cure for almost all treated patients, regardless of age, cirrhosis status, prior treatment failure history, and presence of comorbidities such as CKD, making CHC treatment tolerable and applicable to all populations with CHC, including historically difficult-to-treat populations.8Some studies have highlighted the association of HCV elimination by DAAs with the risk of HCC and other liver-related complications.9,10 In addition, HCV elimination by DAAs has been associated with significant reductions in the risk of certain nonliver diseases such as diabetes.11-13 However, the effects of DAAs on most nonliver comorbidities have not been well documented. In addition, data for long-term outcomes after DAA treatment are limited. Large-scale data obtained from clinical practices without the selection bias of tertiary care center data are more generalizable and can better inform future public health and policy planning.
Therefore, using a large nationwide database of insured patients with CHC monitored in routine clinical practice across the US, we investigated and compared the risk of liver-related and nonliver complications as well as overall mortality between DAA-treated and untreated patients with CHC.
JAMA Intern Med. Published online December 12, 2022
Eiichi Ogawa, MD, PhD; Nicholas Chien, MD; Leslie Kam, MD; Yee Hui Yeo, MD; Fanpu Ji, MD, PhD;
Daniel Q. Huang, MD; Ramsey Cheung, MD; Mindie H. Nguyen, MD, MAS
Key Points
Question What is the risk of mortality and liver and nonliver complications for patients with chronic hepatitis C (CHC) who are being treated with direct-acting antivirals (DAAs)?
Findings This cohort study of 245 596 adults with CHC found that DAA treatment (vs no treatment) was independently associated with a lower risk of mortality and liver (ie, hepatocellular carcinoma and decompensation) and nonliver (ie, diabetes, chronic kidney disease, cardiovascular disease, and nonliver cancer) outcomes.
Meaning These findings support the need for continued efforts to promote hepatitis C screening for diagnosis and treatment of CHC before onset of complications to prevent liver and nonliver complications and to lower all-cause mortality.
Abstract
Importance Chronic hepatitis C (CHC) and its complications are associated with high rates of morbidity and mortality. However, large-scale data analysis of the long-term liver and nonliver effects of direct-acting antiviral (DAA) treatment has been limited.
Objective To assess the association of hepatitis C virus elimination through DAA treatment with the risk of liver and nonliver morbidity and mortality during long-term follow-up among a large nationwide cohort of insured patients with CHC in the US.
Design, Setting, and Participants This was a retrospective cohort study of 245 596 adult patients with CHC using data from the Optum Clinformatics Data Mart database, 2010 to 2021. Of the total cohort, 40 654 patients had received 1 or more prescriptions for DAA medication (without interferon), and 204 942 patients were untreated.
Exposure Treatment with a DAA.
Main Outcomes and Measures Incidence of hepatocellular carcinoma (HCC), liver decompensation, relevant nonliver events (nonliver cancer, diabetes, chronic kidney disease, cardiovascular disease), and overall mortality.
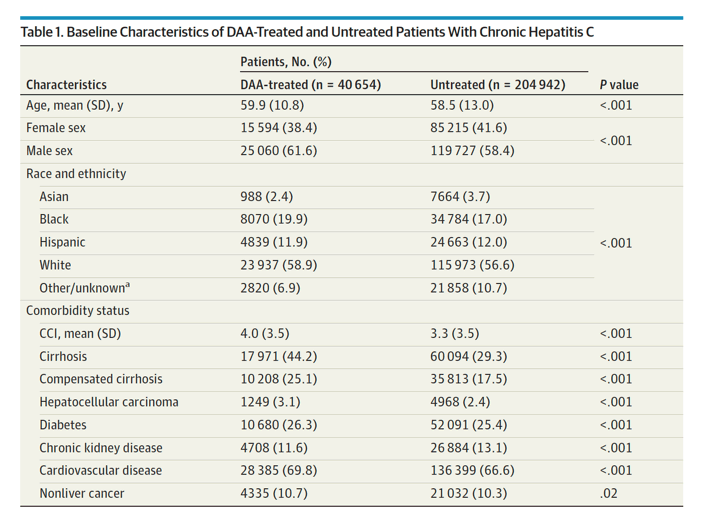
Results The DAA-treated cohort (vs untreated) were older (mean [SD] age, 59.9 [10.8] vs 58.5 [13.0] years; P < .001); more likely to be male (25 060 [62%] vs 119 727 [58%] men; P < .001) and White (23 937 [59%] vs 115 973 [57%]; P < .001) individuals; and more likely to have diabetes (10 680 [26%] vs 52 091 [25%]; P < .001) or cirrhosis (17 971 [44%] vs 60 094 [29%]; P < .001). Comparing DAA-treated with untreated patients, the incidence (per 1000 person-years) of liver outcomes (eg, decompensation, 28.2 [95% CI, 27.0-29.4] vs 40.8 [95% CI, 40.1-41.5]; P < .001, and HCC in compensated cirrhosis, 20.1 [95% CI, 18.4-21.9] vs 41.8 [95% CI, 40.3-43.3]; P < .001) and nonliver outcomes (eg, diabetes, 30.2 [95% CI, 35.4-37.7] vs 37.2 [95% CI, 36.6-37.9]; P < .001; and chronic kidney disease, 31.1 [95% CI, 29.9-32.2] vs 34.1 [95% CI, 33.5-34.7]; P < .001) were significantly lower in treated patients. The all-cause mortality rates per 1000 person-years were also significantly lower in DAA-treated compared with untreated patients (mortality, 36.5 [95% CI, 35.4-37.7] vs 64.7 [95% CI, 63.9-65.4]; P < .001). In multivariable regression analysis, DAA treatment was independently associated with a significant decrease in the risk of liver (adjusted hazard ratio [aHR] for HCC, 0.73; decompensation, 0.36), nonliver (aHR for diabetes, 0.74; chronic kidney disease, 0.81; cardiovascular disease, 0.90; nonliver cancer, 0.89), and mortality outcomes (aHR, 0.43).
Conclusions and Relevance The findings of this retrospective cohort study indicate that DAA treatment for insured patients with CHC was associated with improved liver- and nonliver outcomes, and ultimately, with long-term overall survival.
| |
| |
| |
|
|
|