| |
Bulevirtide monotherapy for 48 weeks in patients with HDV-related
compensated cirrhosis and clinically significant portal hypertension
|
| |
| |
Dec 2022
Introduction
Download the PDF here
Chronic hepatitis delta (CHD) is a severe form of chronic viral hepatitis that affects approximately 12-72 million patients worldwide.
Until now, the only therapeutic approach available was the off-label use of a 48-week course of pegylated-interferon-α (PegIFNα), an antiviral strategy characterized by limited off-therapy responses and an unfavorable safety profile.
In 2020, the European Medicines Agency provided conditional approval for bulevirtide (BLV) at the dose of 2 mg/day subcutaneously for the treatment of compensated CHD. BLV is a first in class entry inhibitor of HBV; it is a lipopeptide that mimics the Na+-taurocholate co-transporting polypeptide (NTCP) receptor binding domain, blocking HDV/HBV entry exclusively in liver cells and thereby preventing NTCP-mediated intrahepatic HDV spreading.
In phase II trials and in week 48 analysis of the phase III study, BLV treatment significantly reduced HDV RNA levels and improved alanine aminotransferase (ALT) levels, although these benefits were lost in most patients after drug withdrawal.
When combined with PegIFNα, a synergistic effect on both HDV viremia and HBsAg decline was demonstrated.
Virological response was mirrored by a significant decline of intrahepatic HDV RNA levels and of the number of HDV antigen (HDAg)-positive cells in a subset of patients treated for 48 weeks.
Preliminary real-life studies not only confirmed the effectiveness and safety of this treatment strategy but also suggested that treatment extension may further improve both virological and biochemical responses.
However, to date, the safety and effectiveness of BLV 2 mg/day in patients with compensated cirrhosis and clinically significant portal hypertension (CSPH) are poorly known, as these patients have been scarcely represented in clinical trials of BLV and there are only a few case reports in the literature.
These difficult-to-manage patients face a substantial risk of developing decompensated liver disease and hepatocellular carcinoma (HCC) within a short period of time. Moreover, treatment with PegIFNα is contraindicated due to the severity of the underlying liver disease.
Our study aimed to explore the safety and effectiveness of BLV 2 mg/day monotherapy in patients with HDV-related compensated cirrhosis and CSPH with or without HCC.
Highlights
• 48 weeks of bulevirtide 2 mg/day monotherapy was safe in patients with compensated cirrhosis and clinically significant portal hypertension.
• Virological, biochemical, and combined responses were achieved in 78%, 83% and 67% of patients, respectively.
• BLV treatment led to a significant improvement in most biochemical variables and an increase in liver function parameters.
• Treatment was safe and well tolerated, an asymptomatic increase of bile acids was observed.
Background & Aims
Bulevirtide (BLV) has recently been conditionally approved for the treatment of chronic hepatitis delta (CHD) in Europe, but its effectiveness and safety in patients with compensated cirrhosis and clinically significant portal hypertension (CSPH) are unknown.
Methods
Consecutive patients with HDV-related compensated cirrhosis and CSPH who started BLV 2 mg/day were enrolled in this single-center study. Clinical/virological characteristics were collected at baseline, weeks 4, 8 and every 8 weeks thereafter. HDV RNA was quantified by Robogene 2.0 (lower limit of detection 6 IU/ml).
Results
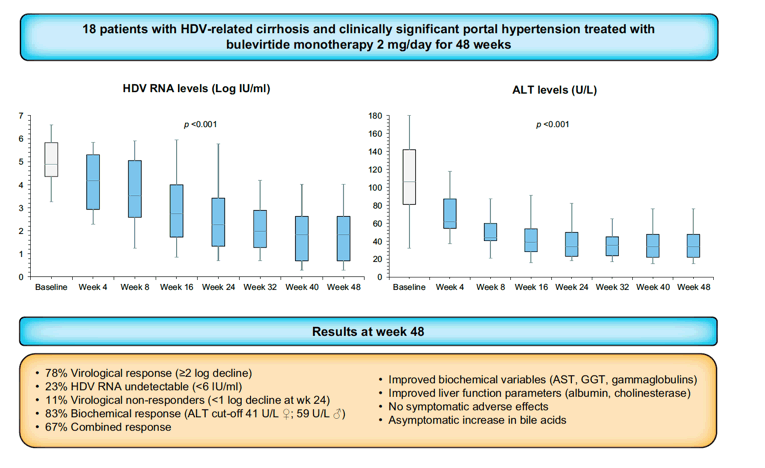
Eighteen Caucasian patients with compensated cirrhosis and CSPH under nucleos(t)ide analogue treatment were enrolled: median (IQR) age was 48 (29-77) years, and 67% were male. Median (IQR) platelet count was 70 (37-227) x103/μl, liver stiffness measurement (LSM) 16.4 (7.8-57.8) kPa, alanine aminotransferase (ALT) 106 (32-222) U/L, HBsAg 3.7 (2.5-4.3) log IU/ml, HDV RNA 4.9 (3.3-6.6) log IU/ml. During 48 weeks of BLV monotherapy, HDV RNA declined by 3.1 (0.2-4.3) log IU/ml (p <0.001 vs. baseline), becoming undetectable in 5 patients (23%). A virological response was observed in 14 (78%) patients while a non-response was observed in 2 (11%). ALT decreased to 35 (15-86) U/L (p <0.001 vs. baseline), normalizing in 83% of patients. A combined response was observed in 67% of patients. Aspartate aminotransferase and gamma-glutamyltransferase levels significantly improved. Concerning liver function parameters, albumin values significantly increased and bilirubin remained stable. LSM significantly improved in patients with virological response, while platelet count was unchanged. None of the patients developed decompensating events or hepatocellular carcinoma. BLV was well tolerated, no patient discontinued treatment and the increase in bile acids was fully asymptomatic.
Conclusions
A 48-week course of BLV 2 mg/day monotherapy is safe and effective even for difficult-to treat patients with HDV-related compensated cirrhosis and CSPH.
Lay summary
Hepatitis delta virus (HDV) is associated with the most severe form of viral hepatitis. A new treatment for HDV called bulevirtide has recently received conditional approval for patients with chronic HDV infection. However, its safety and effectiveness in patients with more advanced liver disease is not known. Herein, we show that it is safe and effective in patients with HDV-related cirrhosis and clinically significant portal hypertension.
|
|
| |
| |
|
|
|