| |
Fatty Liver in HIV: higher prevalence,
progresses faster, ALT/AST Predict
|
| |
| |
Clinical significance of elevated liver
transaminases in HIV-infected patient
Prevalence and risk factors of nonalcoholic
fatty liver disease in HIV-monoinfection
Hepatic steatosis progresses faster in HIV
mono-infected than HIV/HCV co-infected patients
and is associated with liver fibrosis
Fatty liver disease in HIV: common,
underappreciated, and understudied
3 studies follow this editorial comment: Clinical Significance of Elevated ALT, AST; Hepatic Steatosis Progresses Faster in HIV+ than HCV/HIV; Prevalence & Risk Factors of NAFLD in HIV Mono-infection.

In HIV-negative populations, NAFLD and NASH are associated with increased cardiovascular and renal disease as well as all-cause mortality. Their findings are remarkable for several reasons. First, the analysis identifies a NAFLD prevalence in HIV-monoinfected patients of 35%, higher than prevalence estimates for the general population in North America, Europe, or worldwide. Although awaiting additional data from trials, clinical assessment for fatty liver disease can be integrated into HIV care in most settings. Patients with metabolic risk factors, especially those with elevated aminotransferase levels, should be evaluated and managed following guidelines for the general population [17,18]. BMI is especially concerning given the rising prevalence of overweight and obesity in HIV-positive patients. weight gain was more strongly associated with increased risk of incident diabetes mellitus in HIV-positive vs. HIV-negative individuals (10 vs. 6% increased risk per 5-lb gain).. Perhaps the most important take-home message is that persistently elevated aminotransferase levels in HIV-infected patients should not be ignored or attributed to minor ART-related toxicities. They may be the first clue to significant liver disorders that can have major clinical consequences.
As the introduction of combination antiretroviral therapy (ART), HIV-infected individuals have experienced reduced morbidity from opportunistic infections and malignancies, and an increased life expectancy. With longer life, the prevalence of comorbid conditions in HIV-infected adults has increased.
Liver disease is the highest non-AIDS cause of death in HIV-infected patients, in part due to the high rates of coinfection with viral hepatitis [1,2]. Recent reports suggest a decline in liver-related deaths and, with the increasing availability of effective antiviral therapy for hepatitis B and C, further reductions in liver-related morbidity and mortality are expected. Consequently, nonalcoholic fatty liver disease (NAFLD), and the necroinflammatory, progressive form of NAFLD, nonalcoholic steatohepatitis (NASH), have emerged as increasingly frequent causes of clinically significant liver disease in HIV-infected persons.
In HIV-negative populations, NAFLD and NASH are associated with increased cardiovascular and renal disease as well as all-cause mortality [3,4]. NASH can progress to end-stage liver disease and is associated with an increased risk of hepatocellular carcinoma, even in the absence of cirrhosis [5,6]. Despite these significant clinical risks, the natural history of NAFLD and NASH in HIV-infection remains poorly understood. Further, the safety and efficacy of available preventive and therapeutic strategies for NAFLD and NASH are unstudied in HIV.
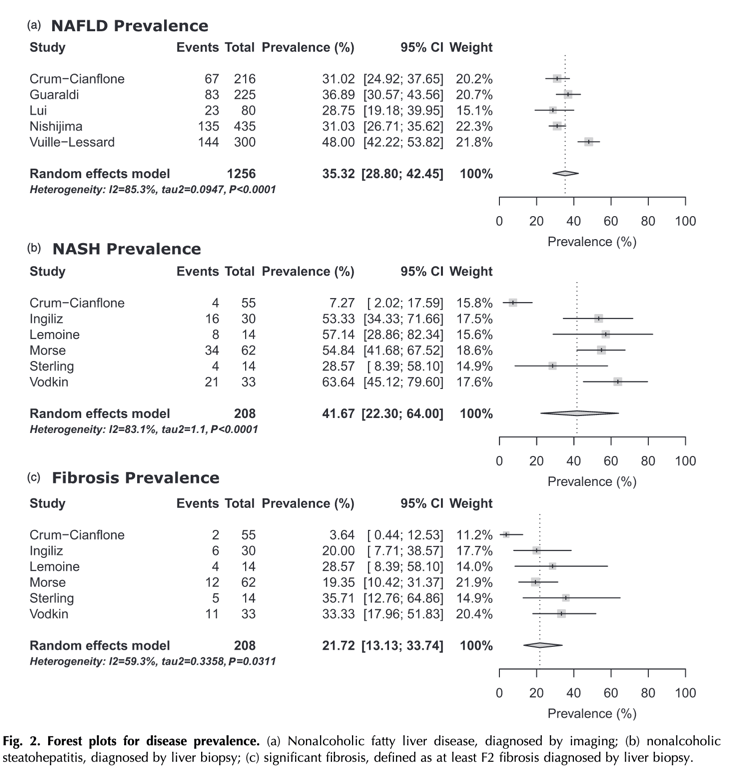
In this issue of AIDS, Maurice et al.[7] report a systematic review and meta-analysis of NAFLD, NASH, and hepatic fibrosis in HIV-monoinfected adults. Their findings are remarkable for several reasons. First, the analysis identifies a NAFLD prevalence in HIV-monoinfected patients of 35%, higher than prevalence estimates for the general population in North America, Europe, or worldwide [8,9]. In HIV-infected patients undergoing liver biopsy for further evaluation of elevated aminotransferases, NASH was seen is 45% and significant fibrosis in 22%. In similar studies in HIV-negative populations, NASH prevalence is 12-30% and significant fibrosis in 7% [10]. Though a number of factors can influence these rates, including race and ethnicity, and the prevalence of obesity and diabetes, taken together, these findings confirm that HIV-infected patients are at high risk for NASH, fibrosis, and related complications.
Second, the meta-analysis confirms that metabolic factors, notably elevated BMI, insulin resistance, and dyslipidemia, confer the greatest increased risk of NAFLD in HIV infection, similar to the general population. CD4+ cell count was the only HIV-specific variable associated with NAFLD in the analysis, suggesting a role for immune recovery. Notably, age, time since HIV diagnosis, and duration of ART were not associated with increased risk.
The association with BMI is especially concerning given the rising prevalence of overweight and obesity in HIV-positive patients [11]. In one large US outpatient cohort, nearly half were overweight or obese at the time of ART initiation and 20% increased to a higher BMI category within the subsequent 2 years [12]. In a US Veterans' Affairs cohort, HIV-positive veterans gained significantly more weight 1 year after initiation of ART compared with age-matched and sex-matched HIV-negative controls [13].
Not surprisingly, these weight changes translate to increased risk of metabolic complications. In the Veterans' Affairs cohort, weight gain was more strongly associated with increased risk of incident diabetes mellitus in HIV-positive vs. HIV-negative individuals (10 vs. 6% increased risk per 5-lb gain). In the multicohort Data Collection on Adverse Events of Anti-HIV Drugs study, weight gain in the first year after initiation of ART was associated with an increased risk of cardiovascular disease and diabetes [14]. Coupled with HIV-related inflammation and immune activation, the high prevalence of obesity places HIV-infected patients at increased risk for metabolic complications including NAFLD and NASH and highlights the need for more aggressive management of obesity and fatty liver in the aging HIV-infected population.
Perhaps most remarkable, this analysis, limited to studies including imaging or histologic confirmation of NAFLD and NASH, identified only 10 studies suitable for inclusion, and the quality of the studies, graded by the NIH Quality Assessment Tool, was moderate to poor, due to the retrospective, cross-sectional study designs and lack of longitudinal follow-up. More than 20 years after the introduction of combination ART and recognition of the complex adverse effects of ART and the associated immune recovery, assessment of fatty liver disease in HIV-positive patients remains woefully understudied. Consequently, one of the more vexing questions in HIV and NAFLD remains unanswered: what is the contribution of specific antiretroviral classes and agents to liver disease risk? One study included in the meta-analysis identified an association between steatosis with NRTIs, whereas four others did not. Integrating noninvasive assessments of liver disease into trials of ART initiation or switch studies could help define the incidence and prevalence of NAFLD and potential contribution of ART to disease progression.
As Maurice et al.[7] discuss, although NAFLD and NASH are common in HIV-infected adults, the absence of long-term studies limits our understanding of how disease progression compares with that in HIV-negative patients. This meta-analysis serves as a call to action for further study of NAFLD and NASH in HIV. Including assessments of NAFLD and NASH in HIV clinical trials can better define the epidemiology and natural history in HIV, a critical first step to reducing risk and improving outcomes in our patients. Longitudinal studies ideally with histologic assessments are needed to characterize the natural history of NAFLD in HIV, especially as NASH may occur with more fulminant features and at a lower BMI in HIV-infected patients [15,16].
Although awaiting additional data from trials, clinical assessment for fatty liver disease can be integrated into HIV care in most settings. Patients with metabolic risk factors, especially those with elevated aminotransferase levels, should be evaluated and managed following guidelines for the general population [17,18]. Perhaps the most important take-home message is that persistently elevated aminotransferase levels in HIV-infected patients should not be ignored or attributed to minor ART-related toxicities. They may be the first clue to significant liver disorders that can have major clinical consequences.

With the aging of the HIV-infected population following the successful implementation of ART in Western countries, liver-related conditions are now a major comorbidity in this setting.
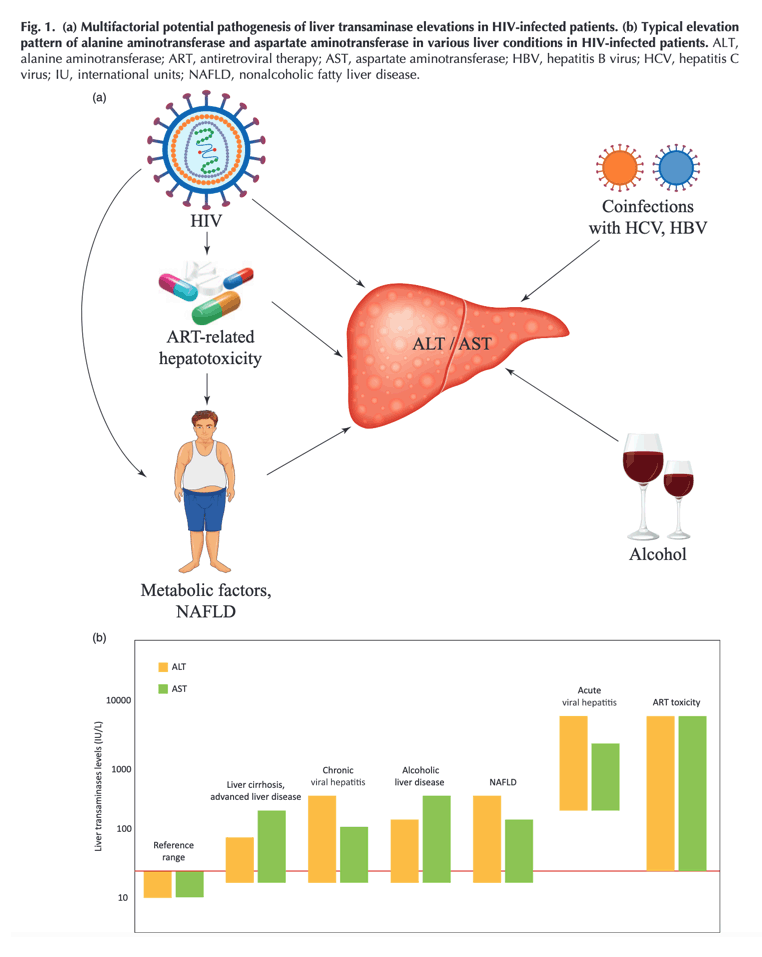
Elevated liver transaminases are particularly common in HIV-infected patients, occurring in 20-93% of patients on antiretroviral therapy (ART), even in the absence of viral hepatitis coinfection [16,17]. Causes of elevated transaminases in HIV-infected adults include coinfection with hepatitis viruses A, B, C, D, E, alcohol abuse and ART toxicity. A possible contribution to chronic liver injury by HIV infection itself or the associated immunodeficiency has also been hypothesized [18,19]. Furthermore, in HIV mono-infected patients transaminase elevations may suggest the presence of an emerging and highly prevalent liver disease, namely nonalcoholic fatty liver disease (NAFLD), with associated hepatic fibrosis and cirrhosis [20-23]. In the general population, elevated ALT is associated with liver-related mortality [12,24]. Likewise, in HIV-infected patients elevated liver transaminases are independently associated with all-cause and liver-related mortality [25,26]. Patients with transaminase elevations require additional visits for laboratory and clinical assessments and often undergo interruptions and changes in ART [27].
Elevation of liver transaminases is common in patients infected with the HIV. Although this is usually an incidental finding during regular work-up, HIV-infected patients with transaminase elevations require additional visits for laboratory studies and clinical assessments, and often undergo interruptions and changes in antiretroviral therapy (ART). Alanine aminotransferase is present primarily in the liver, thus being a surrogate marker of hepatocellular injury. Aspartate aminotransferase is present in the liver and other organs, namely cardiac and skeletal muscle, kidney and brain. Serum levels of both liver transaminases predict liver-related mortality. Moreover, serum fibrosis biomarkers based on alanine aminotransferase and aspartate aminotransferase predict all-cause mortality. In a busy clinical setting, a diagnostic approach to elevated liver transaminases could be complicated given the frequency and nonspecificity of this finding. Indeed, HIV-infected individuals present multiple risk factors for liver damage and chronic elevation of transaminases, including coinfection with hepatitis B and C viruses, alcohol abuse, hepatotoxicity due to ART, HIV itself and frequent metabolic comorbidities leading to nonalcoholic fatty liver disease.

Surprisingly, HIV mono-infected individuals had significantly higher rates of HS progression than patients with HIV/HCV co-infection. Longitudinal studies assessing incidence rates of HS in the context of HIV infection are scarce, particularly for HIV mono-infected patients. In a study of 146 HIV/HCV co-infected patients with paired liver biopsies, progression to HS was observed in 40% of the patients over a median follow-up period of 3.3 (2.0-5.2) years, which is in line with our data.[8] However, no prospective study assessed dynamics of HS in HIV mono-infected patients. The mechanistic explanation underlying our finding is likely multifactorial. HIV mono-infected patients had different clinical characteristics at baseline compared to HIV/HCV co-infected cases, particularly a higher frequency of metabolic dysfunction and a higher BMI, which was the strongest predictor of HS progression. Moreover, HIV/HCV co-infected patients had a different lipid profile than HIV mono-infected patients, including lower levels of total HDL and LDL cholesterol. The term "lipid paradox" has recently been coined for HIV/HCV co-infected patients, that is, despite the upregulation in HIV/HCV co-infection of proproteins which generally lead to higher serum LDL, LDL levels remain low in these patients. [31] This is likely due to the profound chronic inflammatory state characterizing HIV/HCV co-infection, which may affect the ability of the liver to synthesize lipoproteins. Finally, given that liver fibrosis progresses faster in HIV/HCV co-infected patients, a slower HS progression could be partly explained by fat loss due to higher rates of advanced fibrosis.[32] On multivariable analysis, BMI and HIV mono-infection were the only independent predictors of HS progression. The influence of BMI in HS dynamics measured by CAP has already been suggested by Macias and colleagues.[21] However, the follow-up period was shorter than in our study and most of the included patients (74%) were HIV/HCV co-infected, so a direct comparison with HIV mono-infected individuals was not carried out.
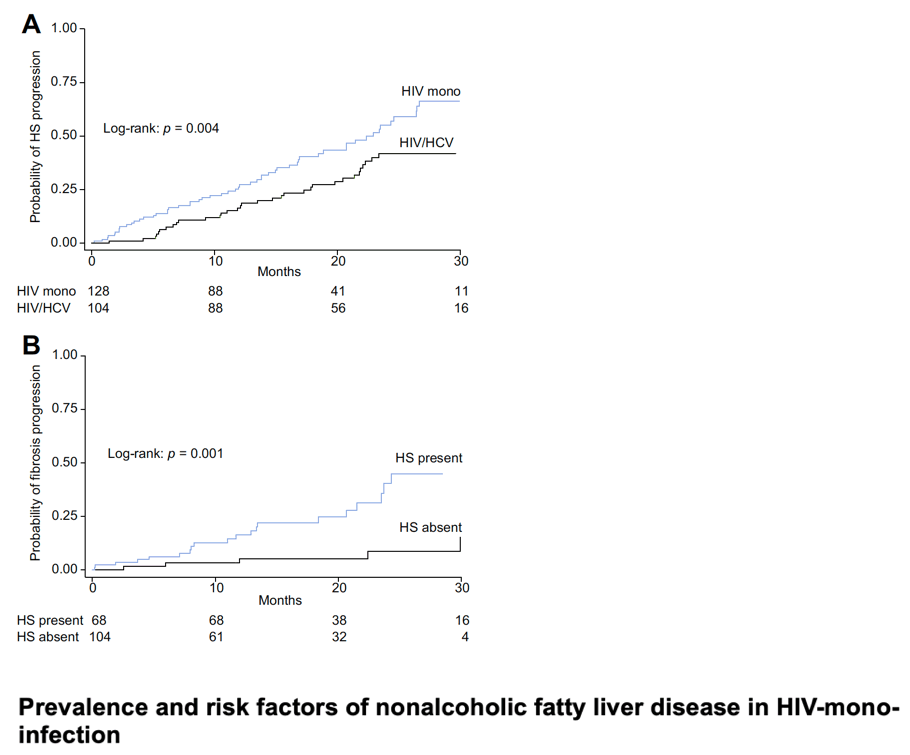
NAFLD is common in HIV-monoinfected patients. Metabolic disorders are key risk factors of NAFLD independent of HIV parameters and predict its complications. The strong association between obesity and liver fibrosis in HIV-monoinfected patients supports the adipocentric concept of liver fibrogenesis in this population. This needs to be confirmed by additional studies. Our systematic review underlines the need for additional data on NAFLD in HIV infection as well as a better standardized assessment and management of the disease.
Based on radiological criteria, our review found a prevalence of NAFLD in HIV-monoinfected of 35%. Prevalence estimates in the general population vary significantly with diagnostic modality and study population. A recent systematic review has reported a worldwide prevalence of NAFLD diagnosed by imaging at 25%. Stratified by region, the prevalence in Asia, Europe and the United States America was 27, 24 and 24%, respectively [4]. However, the figure increases up to 70% in diabetic patients and over 90% among obese patients [25].
BMI (MD 2.92 95% CI 2.14-3.70, P < 0.00001), waist circumference [MD 8.05 (5.46-10.64) P < 0.00001], type 2 diabetes [OR 1.61 (1.09-2.39) P = 0.02], hypertension [OR 1.75 (1.27-2.41) P = 0.0006], high triglycerides [MD 61.52 (24.31-98.74) P = 0.001], high total cholesterol [MD 6.19 (0.93-11.45) P = 0.02], low HDL cholesterol [MD -4.21 (-6.82 to -1.59) P = 0.002], high LDL cholesterol [MD 5.80 (2.01-9.58) P = 0.003], high fasting glucose [MD 0.43 (0.18-0.68) P = 0.0007], high alanine aminotransferase [MD 15.98 (8.04-23.92) P < 0.0001], high aspartate aminotransferase (AST) [MD 5.27 (2.66-7.88) P < 0.0001] and high CD4+ T-cell count [MD 54.83 (11.55-98.11) P = 0.0]) were associated with NAFLD. Age, suppressed HIV viral load, duration of HIV infection, duration of HAART and CD4+ nadir were not associated with NAFLD (Fig. 3). There were insufficient data to meta-analyse the diagnosis of dyslipidaemia (based on the use of lipid-lowering drugs).
BMI (MD 1.38 95% CI 0.04-2.71 P = 0.04), fasting glucose [MD 0.80 (0.47-1.13) P < 0.00001] and AST level [MD 13.00 (4.34-21.65) P = 0.003] were associated with fibrosis, whereas male sex [OR 0.27 (0.15-0.51) P < 0.0001] and black ethnicity [OR 0.18 (0.08-0.44) P = 0.0002] were protective factors. CD4+ cell count (NASH) and duration of HIV infection (fibrosis) were the only HIV-specific variables with data for meta-analysis, neither of which were associated with the outcome of interest (Fig. 4). There was significant statistical heterogeneity for age (I2 = 85% P = 0.0002).
Taken together, these results warrant three comments. First, imaging studies suggest that NAFLD is common in HIV-monoinfected patients but limited histological data without long-term follow-up on the incidence of NASH/fibrosis in the post-HAART era mean we still do not know what the rate is of fibrosis progression as compared with HIV-negative patients with NAFLD. It is possible that although metabolic factors predominate in disease pathogenesis, HIV infection and/or drug exposure could potentiate the effects in the liver such that disease progresses with milder features of the metabolic syndrome, which may explain why some studies show NAFLD occurring in HIV-infected patients with lower BMI [28]. However, to address this question longitudinal studies with good-quality histological data are needed.
Second, patients with HIV infection and features of the metabolic syndrome require a full diagnostic workup for NAFLD. The prevalence of NAFLD is rising rapidly in the Western world in parallel with increasing obesity and the metabolic syndrome and has become a prominent cause of liver-related morbidity and requirement for liver transplantation [29]. Both NAFLD and HIV also confer increased risk of cardiovascular disease [30]. Therefore, the management of dyslipidaemia, hypertension, diabetes mellitus and obesity in a multi-disciplinary setting is therefore an essential aspect of HIV care and is already an integral service offered to patients in many centres. This needs to be combined with effective liver assessment and risk stratification with noninvasive tools such as transient elastography and/or biochemical markers (e.g. Enhanced Liver Fibrosis were and NAFLD fibrosis score) [31], to identify patients who need more dedicated input from a specialist hepatologist. However, the performance of these tools has been poorly validated in HIV monoinfected patients, and the disparity between estimates of fibrosis in the studies reporting both FIB-4 and transient elastography data highlights this point.
Third, we are entering a new era in the management of NAFLD. The mainstay of treating the disease has long been based on life style modification including weight loss and physical activity [32]. However, only a small proportion of patients successfully achieve this. Significantly, multiple new treatments are emerging for advanced disease [33]. Yet HIV infection is invariably an exclusion factor for patients enrolled in these trials. The data from this review show how therapeutic options are at least as important in patients with HIV, and it is essential such trials do not exclude HIV patients.
Abstract
Objective:
To identify the prevalence and risk factors of nonalcoholic fatty liver disease (NAFLD), nonalcoholic steatohepatitis (NASH) and fibrosis in HIV-monoinfected patients.
Design:
Systematic review and meta-analysis.
Methods:
We searched Medline and Embase and included studies that enrolled HIV-monoinfected patients with NAFLD defined by imaging and/or liver histology. Data on prevalence and risk factors for NAFLD, NASH and fibrosis were collected for meta-analysis using random effects models.
Results:
Ten studies were included from the United States of America (n = 4), Canada (n = 1), France (n = 2), Italy (n = 1), Japan (n = 1) and China (n = 1). The prevalence of NAFLD (Imaging studies), NASH and fibrosis (biopsied populations) were 35% [95% confidence interval (CI) 29-42], 42% (95% CI 22-64) and 22% (95% CI 13-34), respectively.
Meta-analysis of risk factors showed that high BMI, waist circumference, type 2 diabetes, hypertension, triglycerides and high CD4+ cell count were associated with NAFLD, whereas HIV viral load, duration of HIV infection, duration of antiretroviral therapy and CD4+ cell count nadir were not.
Patients with high BMI [mean difference (MD) 1.38, 95% CI 0.04-2.71 P = 0.04], fasting glucose (MD 0.80, 95% CI 0.47-1.13 P < 0.00001) and AST level (MD 13.00, 95% CI 4.34-21.65 P = 0.003) were at increased risk of significant liver fibrosis.
Conclusion:
NAFLD is frequently observed in HIV-monoinfected patients, and NASH is a common cause of unexplained abnormal liver function in patients selected for liver biopsy. Metabolic disorders are key risk factors independently of HIV parameters. Future trials on pharmacological interventions in NASH with fibrosis should include patients with HIV.
|
|
| |
| |
|
|
|