| |
Booster Waning Benefit vs Omicron UK
Study NEJM
just published
- Covid-19 Vaccine Effectiveness
against the Omicron (B.1.1.529) Variant
|
| |
| |
Waning Benefit
Vaccine effectiveness was calculated after primary immunization with two doses of
BNT162b2 (Pfizer–BioNTech), ChAdOx1 nCoV-19 (AstraZeneca), or mRNA-1273 (Moderna) vaccine and after a booster dose of BNT162b2, ChAdOx1 nCoV-19, or mRNA-1273 in England.
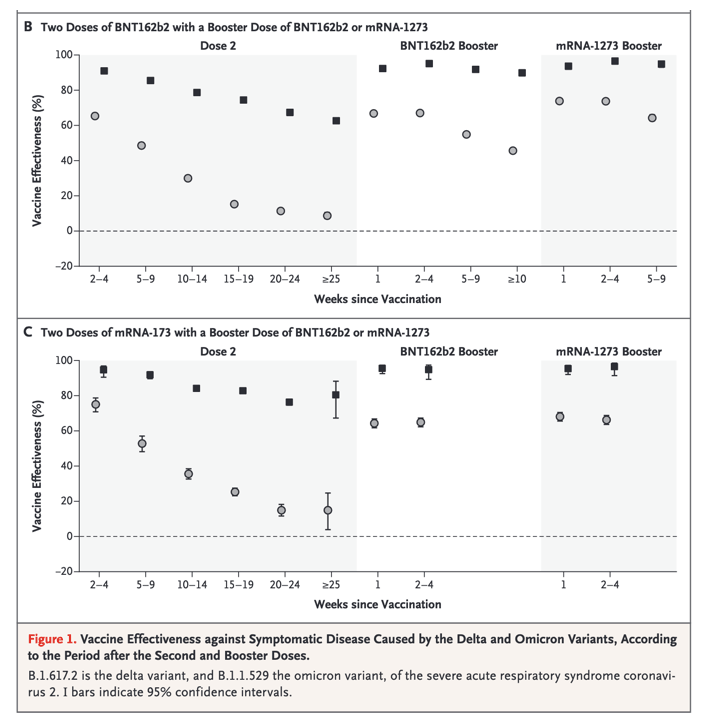
You can see in Table below those who got Pfizer vaccine 7 then booster 10 weeks later 45.7% (declined from 66.9% after 1 week) protection from omicron against symptomatic disease, but with a Moderna booster it was 64% after 5-9 weeks (declined from 74% after 1 week).
Our findings indicate that vaccine effectiveness against symptomatic disease caused by the omicron variant is substantially lower than with the delta variant. After two doses, vaccine effectiveness waned rapidly, with very limited vaccine effects seen from 20 weeks after the second dose of any vaccine. Booster doses resulted in a substantial increase in protection against mild infection; however, waning of protection against symptomatic disease was also seen after booster doses. We are unable to determine protection against severe forms of disease using the test-negative case–control method here owing to the small number of omicron cases resulting in hospitalization so far in our data set and the natural lag between infection and more severe outcomes. Previous experience with the delta variant in the United Kingdom suggested that protection against hospitalization after two doses of vaccine was well maintained.19
The populations that have received different vaccines as a primary course are different. For example, ChAdOx1 nCoV-19 was the main vaccine used early in the program in care homes and among persons in clinical risk groups. Furthermore, mRNA vaccines were the main vaccines used in persons younger than 40 years of age after the reported association between ChAdOx1 nCoV-19 and vaccine-induced thrombotic thrombocytopenia.37 Although adjustments were made for age and clinical risk factors, these age differences may explain some of the differences in the findings for the primary course — for example, the high vaccine effectiveness against omicron 2 to 9 weeks after the second dose of BNT162b2 is likely to be primarily among recently vaccinated young adults and teenagers. Differences are also noted in populations that have received a booster dose as compared with those who have received only two doses, with the former skewed toward older populations with more coexisting conditions. Persons who have not yet received a booster could have missed the boost for reasons that may be associated with exposure risk; for example, booster vaccination may have been delayed owing to an outbreak in a closed setting. The analysis with ChAdOx1 nCoV-19 boosters is particularly likely to be subject to bias because this vaccine was not recommended as a booster in the United Kingdom; therefore, some misclassification may have occurred, and persons who received ChAdOx1 nCoV-19 are likely to have done so because of contraindications to other vaccines.
Vaccine Effectiveness, Omicron Variant
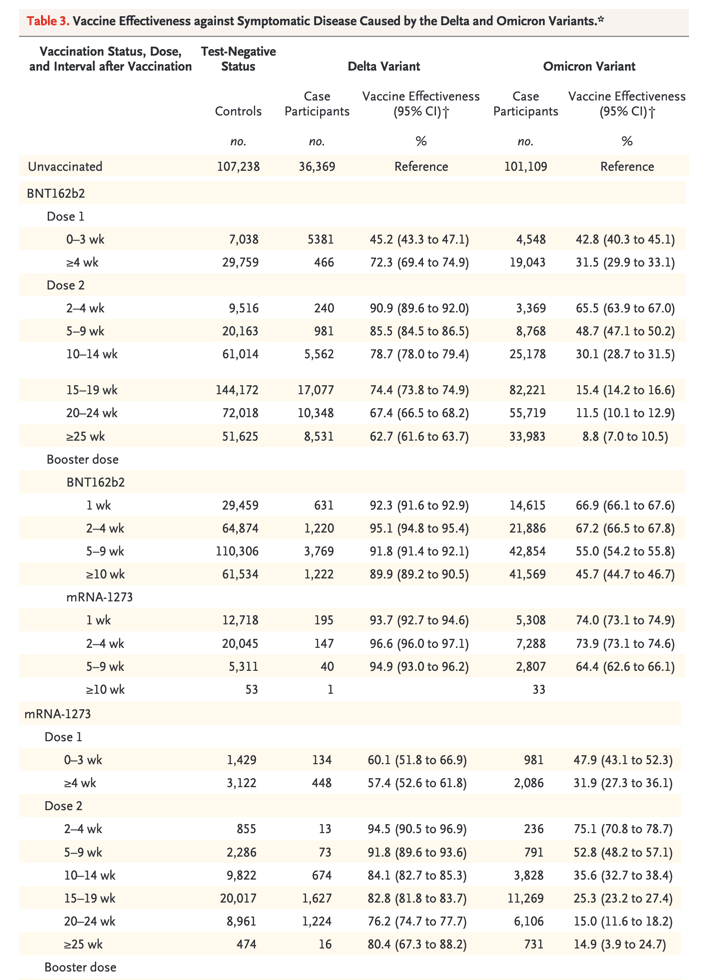
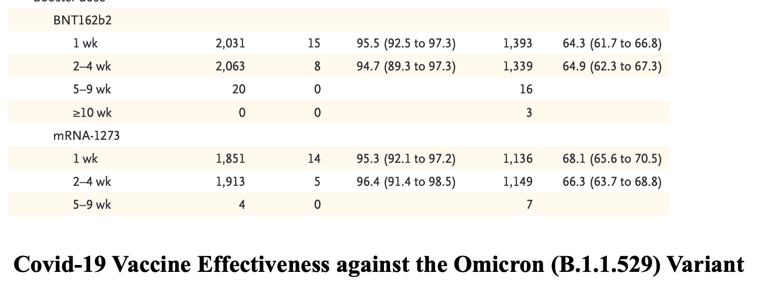
Vaccine effectiveness was lower for the omicron variant than for the delta variant at all intervals after vaccination and for all combinations of primary courses and booster doses investigated. Among those who had received two ChAdOx1 nCoV-19 doses, almost no protective effect of vaccination against symptomatic disease caused by the omicron variant was noted from 20 to 24 weeks after the second dose. Among those who had received two BNT162b2 doses, vaccine effectiveness was 65.5% (95% CI, 63.9 to 67.0) 2 to 4 weeks after the second dose, dropping to 15.4% (95% CI, 14.2 to 16.6) after 15 to 19 weeks and dropping further to 8.8% (95% CI, 7.0 to 10.5) after 25 or more weeks. The vaccine effectiveness of two doses of mRNA-1273 vaccine had a similar reduction over time from 75.1% (95% CI, 70.8 to 78.7) after 2 to 4 weeks to 14.9% (95% CI, 3.9 to 24.7) after 25 or more weeks.
Among persons who received ChAdOx1 nCoV-19 as the primary course, from 2 to 4 weeks after a BNT162b2 booster dose, vaccine effectiveness increased to 62.4% (95% CI, 61.8 to 63.0) before waning to 39.6% (95% CI, 38.0 to 41.1) after 10 or more weeks. The mRNA-1273 booster vaccine increased effectiveness to 70.1% (95% CI, 69.5 to 70.7) after 2 to 4 weeks. This waned to 60.9% (95% CI, 59.7 to 62.1) after 5 to 9 weeks. Vaccine effectiveness was lowest among those who received a ChAdOx1 nCoV-19 primary course with a ChAdOx1 nCoV-19 booster vaccine. Waning efficacy was most notable against the omicron variant, for which the vaccine effectiveness was 46.7% (95% CI, 34.3 to 56.7) at 5 to 9 weeks.
Among persons who received BNT162b2 as the primary course, from 2 to 4 weeks after a BNT162b2 booster dose, vaccine effectiveness increased to 67.2% (95% CI, 66.5 to 67.8) before declining to 45.7% (95% CI, 44.7 to 46.7) after 10 or more weeks. The mRNA-1273 booster increased vaccine effectiveness to 73.9% (95% CI, 73.1 to 74.6) after 2 to 4 weeks before decreasing to 64.4% (95% CI, 62.6 to 66.1) after 5 to 9 weeks. After an mRNA-1273 primary course, vaccine effectiveness increased to 64.9% (95% CI, 62.3 to 67.3) at 2 to 4 weeks after a BNT162b2 booster and 66.3% (95% CI, 63.7 to 68.8) at 2 to 4 weeks after an mRNA-1273 booster.
Neutralizing antibodies correlate with protection against reinfection and vaccine effectiveness against infection; therefore, reduced vaccine effectiveness against the omicron variant is anticipated on the basis of these early laboratory findings.6-8
Waning of protection has been observed with time since vaccination, especially with the delta variant, which is able to at least partially evade natural and vaccine-induced immunity.20 However, third (booster) doses provide a rapid and substantial increase in protection against both mild and severe disease.19,21-25
NEJM March 2, 2022
Abstract
Background
A rapid increase in coronavirus disease 2019 (Covid-19) cases due to the omicron (B.1.1.529) variant of severe acute respiratory syndrome coronavirus 2 in highly vaccinated populations has aroused concerns about the effectiveness of current vaccines.
Methods
We used a test-negative case–control design to estimate vaccine effectiveness against symptomatic disease caused by the omicron and delta (B.1.617.2) variants in England. Vaccine effectiveness was calculated after primary immunization with two doses of BNT162b2 (Pfizer–BioNTech), ChAdOx1 nCoV-19 (AstraZeneca), or mRNA-1273 (Moderna) vaccine and after a booster dose of BNT162b2, ChAdOx1 nCoV-19, or mRNA-1273.
Results
Between November 27, 2021, and January 12, 2022, a total of 886,774 eligible persons infected with the omicron variant, 204,154 eligible persons infected with the delta variant, and 1,572,621 eligible test-negative controls were identified.
At all time points investigated and for all combinations of primary course and booster vaccines, vaccine effectiveness against symptomatic disease was higher for the delta variant than for the omicron variant.
No effect against the omicron variant was noted from 20 weeks after two ChAdOx1 nCoV-19 doses, whereas vaccine effectiveness after two BNT162b2 doses was 65.5% (95% confidence interval [CI], 63.9 to 67.0) at 2 to 4 weeks, dropping to 8.8% (95% CI, 7.0 to 10.5) at 25 or more weeks.
Among ChAdOx1 nCoV-19 primary course recipients, vaccine effectiveness increased to 62.4% (95% CI, 61.8 to 63.0) at 2 to 4 weeks after a BNT162b2 booster before decreasing to 39.6% (95% CI, 38.0 to 41.1) at 10 or more weeks.
Among BNT162b2 primary course recipients, vaccine effectiveness increased to 67.2% (95% CI, 66.5 to 67.8) at 2 to 4 weeks after a BNT162b2 booster before declining to 45.7% (95% CI, 44.7 to 46.7) at 10 or more weeks.
Vaccine effectiveness after a ChAdOx1 nCoV-19 primary course increased to 70.1% (95% CI, 69.5 to 70.7) at 2 to 4 weeks after an mRNA-1273 booster and decreased to 60.9% (95% CI, 59.7 to 62.1) at 5 to 9 weeks.
After a BNT162b2 primary course, the mRNA-1273 booster increased vaccine effectiveness to 73.9% (95% CI, 73.1 to 74.6) at 2 to 4 weeks; vaccine effectiveness fell to 64.4% (95% CI, 62.6 to 66.1) at 5 to 9 weeks.
Conclusions
Primary immunization with two doses of ChAdOx1 nCoV-19 or BNT162b2 vaccine provided limited protection against symptomatic disease caused by the omicron variant. A BNT162b2 or mRNA-1273 booster after either the ChAdOx1 nCoV-19 or BNT162b2 primary course substantially increased protection, but that protection waned over time. (Funded by the U.K. Health Security Agency.)
|
|
| |
| |
|
|
|