| |
NYC DOH COVID PrEP, Treatment -
Evusheld for pre-exposure prophylaxis
|
| |
| |

Download the PDF here
April 4, 2022
Dear Colleagues,
COVID-19 cases have begun to increase in NYC. Protect your patients by ensuring they are up to date with their COVID-19 vaccines, encouraging them to get tested regularly with at-home or provider-based tests, making sure at risk patients are aware of and can access treatment, and offering eligible patients Evusheld for pre-exposure prophylaxis.
Evusheld is a combination of two long-acting monoclonal antibodies (tixagevimab and cilgavimab) administered by intramuscular injection. In clinical trials, Evusheld reduced the risk of symptomatic COVID-19 by 83% over 6 months. Evusheld should only be given to patients who are not currently infected and have not had a recent known exposure to someone with COVID-19. Evusheld is authorized for people ages 12 or older who weigh at least 88 pounds (44 kg) and:
SARS-CoV-2 Variants and Evusheld
The Omicron variant currently accounts for nearly all COVID-19 cases in NYC. Due to decreased neutralization activity of Evusheld against Omicron subvariants BA.1 and BA.1.1 (BA.1+R346K), the initial dosage has been increased to 600 mg (300 mg of tixagevimab and 300 mg of cilgavimab). Evusheld is expected to retain activity against the BA.2 Omicron subvariant.
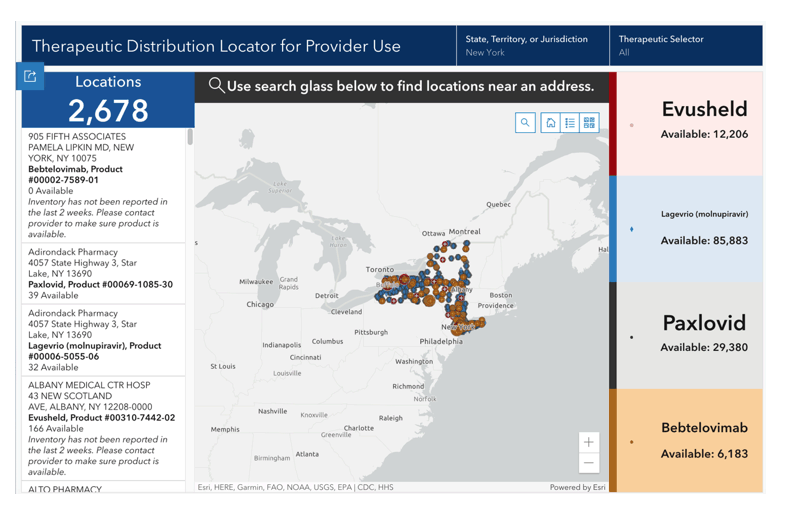
COVID-19 Therapeutics Locator
The national map below displays public locations that have received shipments of U.S. Government-procured COVID-19 therapeutics under U.S. Food and Drug Administration (FDA) Emergency Use Authorization (EUA) authority. The long-acting antibody combination, Evusheld; the monoclonal antibody treatment, bebtelovimab; as well as the oral antiviral therapies, Paxlovid and Lagevrio (molnupiravir) are products authorized by the FDA for either prevention (Evusheld) or treatment (Paxlovid, bebtelovimab, and Lagevrio (molnupiravir)) of COVID-19.
Sotrovimab distribution was paused due to the majority prevalence of the BA.2 omicron subvariant on March 25, 2022 following FDA's revised EUA limiting its use.
As of January 24, 2022, allocations of bamlanivimab/etesevimab and REGEN-COV have been paused following FDA's revised EUA's for both products limiting their use due to the omicron variant.
These therapies require a prescription by a licensed and authorized provider. The therapeutics locator is intended for provider use. Patients should not contact locations directly unless instructed to do so by their healthcare provider.
Additional resources and information related to COVID-19 Therapeutics currently distributed by the federal government can be found on the ASPR COVID-19 Therapeutics page. For questions regarding this site, contact 1-800-232-0233 (TTY 888-720-7489).
Find Locations
Search by therapy and by zip code to find potential locations.
Contact Us 1-800-232-0233 (TTY 888-720-7489)
|
|
| |
| |
|
|
|