| |
Interindividual Variation in Low-Density
Lipoprotein Cholesterol Level Reduction With
Evolocumab - LDL response at week 4 -
An Analysis of FOURIER Trial Data
|
| |
| |
Download the PDF here
Download the PDF here
Amgen press release
The study was statistically powered around the hard major adverse cardiovascular event (MACE) composite endpoint of first heart attack, stroke or cardiovascular death (key secondary composite endpoint) and found that adding Repatha to optimized statin therapy resulted in a statistically significant 20 percent (p<0.001) reduction in these events. The robust benefit in this objective measure started as early as six months and continued to accrue through the median 2.2 years of the study. In fact, the magnitude of the risk reduction in the hard MACE composite endpoint grew over time, from 16 percent in the first year to 25 percent beyond the first year.
The study also found a statistically significant 15 percent reduction (p<0.001) in the risk of the extended MACE composite (primary) endpoint, which included hospitalization for unstable angina, coronary revascularization, heart attack, stroke or cardiovascular death.
Patients on Repatha experienced a reduction in the risk of heart attack (27 percent, nominal p<0.001), stroke (21 percent, nominal p=0.01) and coronary revascularization (22 percent, nominal p<0.001). Consistent with recent trials of more intensive LDL lowering, there was no observed effect on cardiovascular mortality.1-5 Similarly, there was no observed effect on hospitalization for unstable angina. In an exploratory analysis, the relative risk reduction for fatal and non-fatal heart attack or stroke was 19 percent in the first year (p=0.003) and 33 percent beyond the first year (p<0.00001).
"We now show for the first time in a dedicated outcomes study that decreasing LDL cholesterol with PCSK9 inhibition results in clinically meaningful cardiovascular benefit," said Marc S. Sabatine, M.D., M.P.H., chairman of the TIMI Study Group, the Lewis Dexter, MD, Distinguished Chair in Cardiovascular Medicine at Brigham and Women's Hospital, and Professor of Medicine, Harvard Medical School, Boston. "These benefits were achieved with lowering LDL cholesterol down to a median of 30 mg/dL, which is well below current targets, and the magnitude of risk reduction increased the longer patients were on therapy. These results support the need for long-term, vigorous LDL cholesterol reduction in our patients with cardiovascular disease."
When added to statin therapy, Repatha reduced LDL-C from a median of 92 to 30 mg/dL, a reduction of 59 percent at week 48, which was sustained throughout the trial. At 48 weeks, the LDL-C was reduced to at least 25 mg/dL in 42 percent of patients treated with Repatha, as compared with <0.1 percent in the placebo group (p<0.001). Additionally, treatment with Repatha had favorable effects on other lipid parameters.
https://www.amgen.com/newsroom/press-releases/2017/03/landmark-outcomes-study-shows-that-repatha-evolocumab-decreases-ldlc-to-unprecedented-low-levels-and-reduces-risk-of-cardiovascular-events-with-no-new-safety-issues
-----------------------------------
JAMA Cardiol. 2019
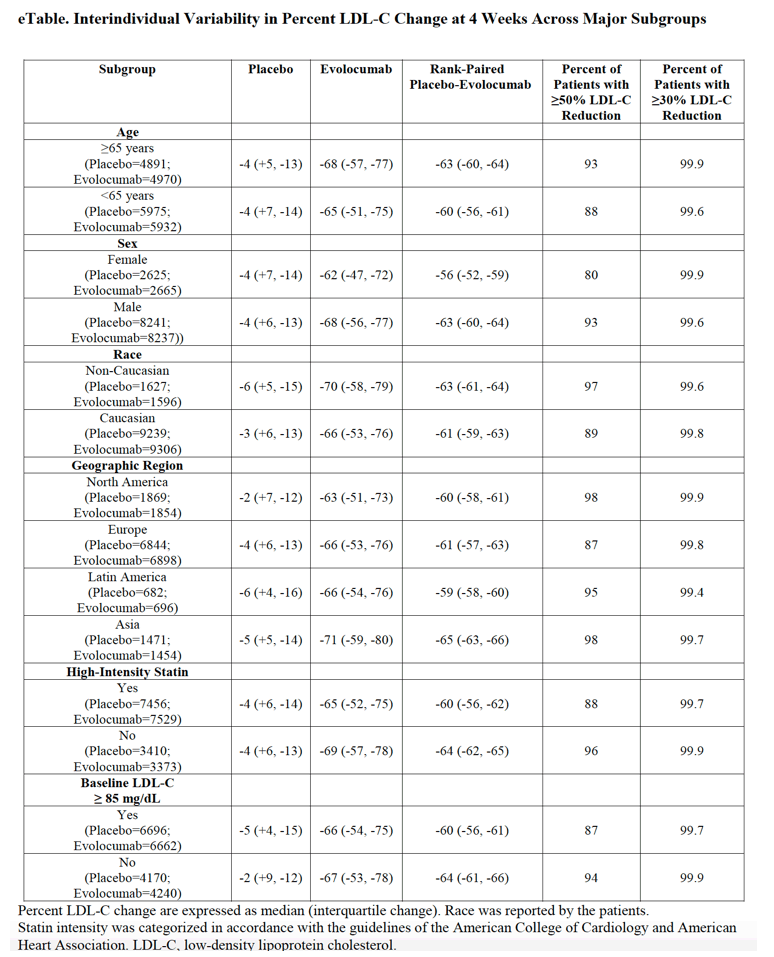
In this secondary analysis of a large randomized clinical trial, the addition of evolocumab to statin therapy lowered LDL-C levels by 50% or greater in more than 90% of patients and by 30% or greater in more than 99% of patients. These findings support the consistency of robust LDL-C level reduction with evolocumab.
At week 4, the median percent reduction in LDL-C levels from baseline was 66% (interquartile range, 54%-76%; median [interquartile range] baseline value, 90 [79-105] mg/dL; postchange value, 31 [21-44] mg/dL) with evolocumab. During the first year, a total of 10 325 of 10902 patients in the evolocumab group (94.7%) had a reduction 50% or greater in LDL-C levels, 10 669 of 10 902 (97.9%) had a reduction 30% or more, and 10 849 of 10 902 (99.5%) had any reduction in LDL-C levels. Fifty-three patients (0.5%) had no apparent reduction in LDL-C levels. In the placebo arm, the median LDL-C reduction was 4% (interquartile range, 6% increase to 13% reduction; baseline median [IQR] value, 90 [79-106] mg/dL; postchange value, 87 [74-103] mg/dL) at 4 weeks.
At the first 2 or 3 study drug administrations, patients were supervised in the administration of the study drug. After the week 4 visit, patients administered the study drug on their own. Levels of LDL-C were measured on the day of first administration of the study drug and at week 4, week 12, week 24, and every 6 months thereafter.
Discussion
In this secondary analysis of a large randomized clinical trial, the addition of evolocumab to statin therapy lowered LDL-C levels by 50% or greater in more than 90% of patients and by 30% or greater in more than 99% of patients. These findings support the consistency of robust LDL-C level reduction with evolocumab.
The concerns for large interindividual variability in LDL-C level reduction response with PCSK9 inhibitors were first raised in clinical trials with bococizumab, a humanized but not fully human monoclonal antibody targeting PCSK9.6 In a pooled analysis of the SPIRE trials (the Evaluation of Bococizumab [PF-04950615; RN316] in Reducing the Occurrence of Major Cardiovascular Events in High Risk Subjects [SPIRE-1] and the Evaluation of Bococizumab [PF-04950615; RN316] in Reducing the Occurrence of Major Cardiovascular Events in High Risk Subjects [SPIRE-2]), waterfall plots showed a large interindividual variation in the LDL-C level reduction with bococizumab, even among patients in whom antidrug antibodies were not detected.6 In addition, although rare, neutralizing antidrug antibodies have also been noted for alirocumab.7 Neutralizing antidrug antibodies have not been seen with evolocumab,4 to our knowledge.
Interindividual variation in LDL-C level reduction with statin use has been observed.1,2 These variations have been attributed to demographic, phenotypic, and genetic factors.8,9
We10 and others2 have shown that in patients receiving a high-intensity statin only approximately 45% of patients achieve an LDL-C reduction of 50% or greater. In comparison, almost all patients receiving evolocumab achieved a 50% or greater LDL-C level reduction. Because many payors require subsequent laboratory testing to demonstrate an optimal reduction in LDL-C with PCSK9 inhibitors, our findings should reassure clinicians and payors that nearly all patients have a robust LDL-C level reduction and such testing is largely unnecessary.
In general practice, nonadherence is likely the most common cause of considerable variation in LDL-C level lowering with a drug.11,12 In this study, we excluded patients who acknowledged alteration in background lipid-lowering therapy and those who did not receive or missed the study drug. However, inclusion of patients who did alter background lipid-lowering therapy but did not acknowledge it may explain the suboptimal response in LDL-C level reduction observed in a small subset of patients allocated to receive evolocumab. Other potential contributors might include changes in dietary intake, lifestyle modifications, concomitant use of medications that influence lipid metabolism, unappreciated errors in study drug assignment (eg, a wrong kit provided) or administration (eg, improper injection technique), mistakes in laboratory sample labeling or handling, or problems with assays. These factors may also explain the significant changes in LDL-C level (increases or decreases of at least 25%) that were noted in 14% of patients in the placebo arm.
In contrast with what was observed for bococizumab, development of neutralizing antidrug antibodies were not seen in any patient in the FOURIER trial or in follow-up through 5 years in the Open Label Study of Long Term Evaluation Against LDL-C Trial (OSLER) and Open Label Study of Long Term Evaluation Against LDL-C Trial-2 (OSLER-2).13 However, other theoretical possibilities exist, such as more rapid clearance of evolocumab or diminished efficacy owing to drug target alterations (eg, rare mutations in the LDL receptor). In the present study, only 53 patients (0.5%) in the evolocumab group had no reduction in LDL-C level at any time within the first year. The biological basis of suboptimal response to PCSK9 inhibition in these patients warrants further investigation.
Limitations
Potential limitations should be acknowledged. First, we calculated the placebo-adjusted difference by subtracting the rank-ordered percentage of LDL-C change within the placebo arm from the corresponding value within the evolocumab arm. Although the baseline characteristics of the 2 groups were comparable, the potential for unmeasured imbalances remains. Second, this analysis was limited to selected population enrolled in a clinical trial; thus, our findings may not be generalizable to the population taking evolocumab. Third, we excluded patients with admitted noncompliance with the study drug but could not exclude patients with unacknowledged noncompliance or unappreciated technical issues in study drug administration. These issues would have no influence on the placebo arm data but potentially a large influence on the evolocumab arm data. The exclusion of patients with noncompliance with the study drug was necessary but does represent a postrandomization variable; however, the baseline characteristics of the patients in the 2 treatment arms used in this analysis remained very similar.
Conclusions
Evolocumab reduced LDL-C levels by 50% or greater in more than 90% of patients and by 30% or greater in more than 99% of patients. These findings provide reassurance that LDL-C level can be robustly reduced with evolocumab without concern for considerable individual-level variability in response.
Results
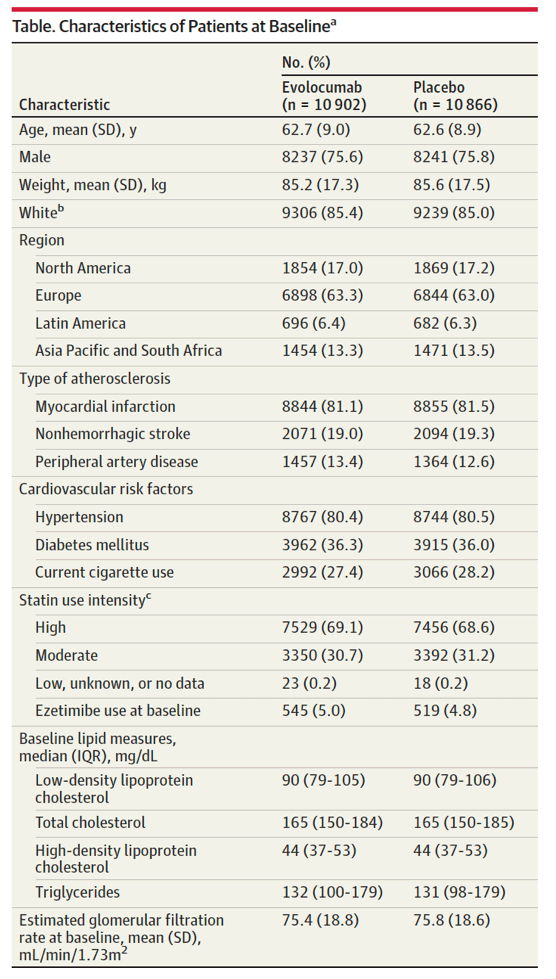
A total of 21 768 patients were included in this analysis. The baseline characteristics of the patients in the 2 treatment groups were comparable and are shown in the Table.
In the evolocumab group, the median percentage of reduction in LDL-C levels from baseline to week 4 was 66% (interquartile range [IQR], 54%-76%; median [interquartile range] baseline value, 90 [79-105] mg/dL; postchange value, 31 [21-44] mg/dL) at 4 weeks (Figure 1A). Of the 10 902 patients in the evolocumab group, 10 700 (98.1%) demonstrated at least some reduction in LDL-C levels, 10 124 (92.9%) had a reduction of 30% or greater, and 8744 (80.2%) had a reduction of 50% or greater. Among the 2158 patients (19.8%) who did not have at least a 50% reduction in LDL-C level at 4 weeks, 2125 (98.5%) had LDL-C levels measured within the first year, and of those, 1581 (74.4%) had a reduction of 50% or greater. Thus, during the first year in the evolocumab group, 10 325 of 10 902 participants (94.7%) had a reduction of 50% or greater in LDL-C level, 10 669 (97.9%) had a reduction of 30% or greater, and 10 849 patients (99.5%) had at least some reduction in LDL-C levels. Fifty-three patients (0.5%) had no apparent reduction in LDL-C level within the first year.
In the placebo group, the median percentage of reduction in LDL-C level from baseline was 4% (IQR, 13% reduction to 6% increase; baseline median [IQR] value, 90 [79-106] mg/dL; postchange value, 87 [74-103] mg/dL) at 4 weeks (Figure 1B). Despite the exclusion of patients who acknowledged altering their background lipid-lowering therapy use, 531 of 10 866 patients (4.9%) showed an increase of 25% or more in LDL-C levels and 985 of 10 866 patients (9.1%) showed a decrease of 25% or more in LDL-C levels.
In the placebo-adjusted analysis, the median percentage of reduction in LDL-C levels with evolocumab was 61% (IQR, 58%-63%) at 4 weeks (Figure 2). Evolocumab reduced the level of LDL-C by 50% or greater in 9839 of 10 866 patients (90.5%) and by 30% or more in 10 846 of 10 866 patients (99.8%), adjusted for changes in the placebo group. This pattern of LDL-C level reduction was consistent across all major subgroups (eTable in the Supplement). A sensitivity analysis that included the 3524 additional patients with high baseline variability in LDL-C levels also showed consistent results with a median percentage of reduction in LDL-C levels of 60% (IQR, 57%-62%) with evolocumab, with 30% or more reduction in 12 601 of 12 627 patients (99.8%).
|
|
| |
| |
|
|
|