| |
Interferon [lambda] therapy shows striking results against COVID-19
|
| |
| |
Download the PDF here
Download the PDF here
Given early, one shot sliced hospitalization risk by half in large trial
'Other scientists also note that the interferon response isn't vulnerable to evolution of new, resistant SARS-CoV-2 variants, unlike monoclonal antibodies, vaccine-induced immunity, or, perhaps, antiviral pills such as Paxlovid. "This is a host-targeting drug versus a virus-targeting drug ... so resistance is really less of an issue," says Jordan Feld'
5 May 2022 Science
From the earliest days of the pandemic, scientists have hoped that interferons, a family of potent proteins that are the body's own first line of defense against viruses, could become weapons against SARS-CoV-2. Because the virus effectively blunts the interferon response, researchers thought providing extra interferons could counter it. But for 2 years, interferons have disappointed in trials in hospitalized patients.
Now, a strikingly positive result from a large trial of nonhospitalized, high-risk people in Brazil has revived hopes. In a study of more than 1900 people, those who received a single shot of a drug called peginterferon lambda within 7 days of developing symptoms of COVID-19 were half as likely to be hospitalized or to endure lengthy emergency room visits as those who received placebo. The effect, which the trial's sponsor, Eiger BioPharmaceuticals, reported in a press release, was seen across many SARS-CoV-2 variants, including Omicron.
Eiger said today it plans to apply for an emergency use authorization for the shot from the U.S. Food and Drug Administration by 30 June. It plans to make full data from the trial available at that time.
"If what they said in the press release is true, it is a very good result," says Ivan Zanoni, an immunologist at Harvard Medical School and Boston Children's Hospital. But he is reserving judgment until a paper details the results, in part because a much smaller trial in younger outpatients with early, uncomplicated SARS-CoV-2 infection found the Eiger injection did not reduce symptom duration or the time it took people to clear the virus. The scientists who led that trial agree. "Until we see a peer-reviewed publication, I am cautious re[garding] press release[s] from companies," Upinder Singh, an infectious diseases physician at the Stanford University School of Medicine, said in an email.
The caution may also reflect the discouraging results from trials of other kinds of interferons. Large trials sponsored by the National Institutes of Health, the World Health Organization, and the company Synairgen all treated hospitalized patients, and all failed.
The current trial was set up to catch patients early. That's because interferons act in the earliest hours and days after viral infection, kicking off a cascade of other proteins that attack the virus at every stage of its life cycle. Located at 12 sites in Brazil, the trial targeted nonhospitalized patients who were older than 50 and/or were at higher risk of severe COVID-19 because they had conditions including diabetes, obesity, high blood pressure, and lung disease. Eighty-four percent of participants were vaccinated. They received a single injection under the skin of placebo or peginterferon lambda, a drug Eiger was already developing to fight hepatitis D.
The company says 25 of 916 patients (2.7%) in the treatment arm were hospitalized or spent more than 6 hours in an emergency room, compared with 57 of 1020 patients (5.6%) who received placebo. Eiger also reported that only one person in the treatment group died, compared with four in the placebo group, although the number of deaths was too small to be statistically meaningful.
"We believe we have a study that is highly generalizable to the current COVID environment in the U.S. and globally," says Eiger CEO David Cory. He says that whereas the current leading antiviral, Pfizer's Paxlovid, is given as a series of pills over 5 days, a single shallow injection of interferon-similar to those people with type 1 diabetes routinely self-administer-"has the potential to be a one-and-done therapy, especially for high-risk patients."
Based on the press release, the results are "quite impressive," says Andreas Wack, an immunologist at the Francis Crick Institute, who has studied the role of lambda interferons in COVID-19. "I'm very hopeful that this may go somewhere."
"From a basic science perspective, this is what was expected to happen," Zanoni says.
Lambda interferons are type 3 interferons, which have receptors mainly on epithelial surfaces, such as those lining the respiratory tract. The better known, type 1 interferons act on every cell in the body, increasing the likelihood of off-target effects. They also promote inflammation more than type 3 interferons-a decided risk in a disease that, later in its course, can tip patients into hyperinflammatory states.
In mice inoculated with SARS-CoV-2, inhaled lambda interferon limited viral infection throughout the respiratory tract without causing excessive inflammation, a team based at Washington University School of Medicine in St. Louis reported in Cell Reports on 15 April. And when the same team engineered mice to lack a receptor specific to lambda interferon-1 (IFNL-1)-the protein in the Eiger product-their viral loads soared compared to mice with intact receptors.
Last year, Zanoni, and colleagues analyzed lung fluid and throat and nose samples from COVID-19 patients. IFNL1 appeared to be associated with the most protective responses, keeping the virus corralled in the upper airways. "I was happy to see [Eiger's apparently successful interferon] was lambda 1 because that would have been our prediction," Zanoni says.
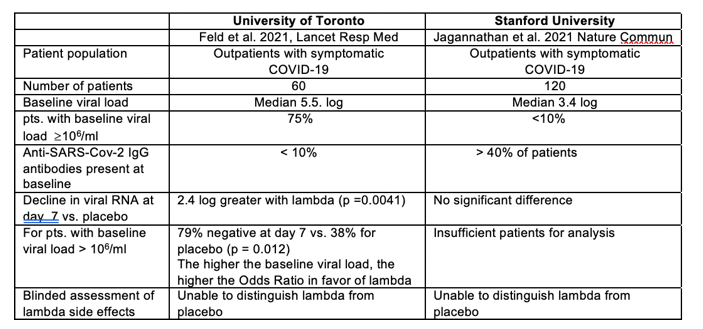
Other scientists also note that the interferon response isn't vulnerable to evolution of new, resistant SARS-CoV-2 variants, unlike monoclonal antibodies, vaccine-induced immunity, or, perhaps, antiviral pills such as Paxlovid. "This is a host-targeting drug versus a virus-targeting drug ... so resistance is really less of an issue," says Jordan Feld, a hepatologist at the University of Toronto. He ran a smaller trial of the Eiger drug in early-stage outpatients and found that a single injection sped clearance of the virus. (Feld has received consulting fees from Eiger.)
Eleanor Fish, an immunologist at the University of Toronto who is an investigator on two unrelated trials of type 1 interferons wonders whether a small company can make enough product to make a difference. "The results are good. My question is: Do they have the capacity to actually make this available?" (The company says it expects to have 300,000 doses ready by the end of this year.)
Feld, who treats patients at Toronto General Hospital, says if the data hold up, the Eiger drug's all-purpose antiviral qualities could make it useful for future respiratory disease pandemics. "While you are waiting for the very specific targeted therapy ... this is one to think about early because it's very likely to have activity against most ----viruses."
--------------------------
Peginterferon Lambda-1a for treatment of outpatients with uncomplicated COVID-19: a randomized placebo-controlled trial
Abstract
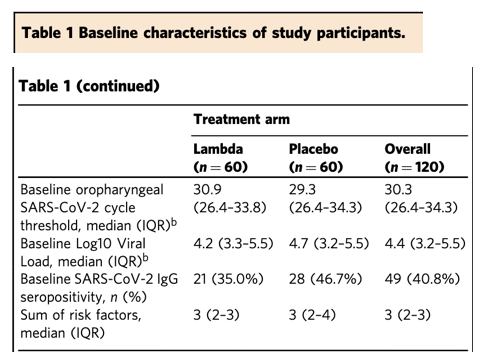
Type III interferons have been touted as promising therapeutics in outpatients with coronavirus disease 2019 (COVID-19). We conducted a randomized, single-blind, placebo-controlled trial (NCT04331899) in 120 outpatients with mild to moderate COVID-19 to determine whether a single, 180 mcg subcutaneous dose of Peginterferon Lambda-1a (Lambda) within 72 hours of diagnosis could shorten the duration of viral shedding (primary endpoint) or symptoms (secondary endpoint). In both the 60 patients receiving Lambda and 60 receiving placebo, the median time to cessation of viral shedding was 7 days (hazard ratio [HR] = 0.81; 95% confidence interval [CI] 0.56 to 1.19). Symptoms resolved in 8 and 9 days in Lambda and placebo, respectively, and symptom duration did not differ significantly between groups (HR 0.94; 95% CI 0.64 to 1.39). Both Lambda and placebo were well-tolerated, though liver transaminase elevations were more common in the Lambda vs. placebo arm (15/60 vs 5/60; p = 0.027). In this study, a single dose of subcutaneous Peginterferon Lambda-1a neither shortened the duration of SARS-CoV-2 viral shedding nor improved symptoms in outpatients with uncomplicated COVID-19.
Liver transaminase elevations were more common in the Lambda vs. placebo arm (15 vs. 5; p = 0.027). Furthermore, we observed significant elevations in alanine transaminase levels from day 0 to day 5 among individuals randomized to Lambda, but not among individuals randomized to placebo (Supplementary Fig. 7). However, there were no associated symptoms and lab abnorm alities were not sustained.
-----------------------------------
Peginterferon lambda for the treatment of outpatients with COVID-19: a phase 2, placebo-controlled randomised trial
February 05, 2021
Between May 18, and Sept 4, 2020, we recruited 30 patients per group. The decline in SARS-CoV-2 RNA was greater in those treated with peginterferon lambda than placebo from day 3 onwards, with a difference of 2⋅42 log copies per mL at day 7 (p=0⋅0041). By day 7, 24 (80%) participants in the peginterferon lambda group had an undetectable viral load, compared with 19 (63%) in the placebo group (p=0⋅15). After controlling for baseline viral load, patients in the peginterferon lambda group were more likely to have undetectable virus by day 7 than were those in the placebo group (odds ratio [OR] 4⋅12 [95% CI 1⋅15-16⋅73; p=0⋅029). Of those with baseline viral load above 106 copies per mL, 15 (79%) of 19 patients in the peginterferon lambda group had undetectable virus on day 7, compared with six (38%) of 16 in the placebo group (OR 6⋅25 [95% CI 1⋅49-31⋅06]; p=0⋅012).
Peginterferon lambda was well tolerated, and adverse events were similar between groups with mild and transient aminotransferase, concentration increases more frequently observed in the peginterferon lambda group. Two individuals met the threshold of grade 3 increase, one in each group, and no other grade 3 or 4 laboratory adverse events were reported.
Interpretation
Peginterferon lambda accelerated viral decline in outpatients with COVID-19, increasing the proportion of patients with viral clearance by day 7, particularly in those with high baseline viral load. Peginterferon lambda has potential to prevent clinical deterioration and shorten duration of viral shedding.

|
|
| |
| |
|
|
|