| |
Maraviroc-Does it Improve Cognitive Function?
New Research This Week in Mice
|
| |
| |
This week a new paper was published in Nature reporting that in mice Maraviroc “restored middle age memory loss” and the authors suggested Maraviroc potentially “could be used to reverse cognitive deficits caused by HIV”. Here are 5 studies in humans PLWH with cognitive impairment and 4 of the 5 found some improvement; 1 found overall no improvement but an improvement in memory and learning and anther no improvement. Another study in asymptomatic treatment-naives found no improvement compared to Atazanavir although both improved cognition. Perhaps another study is in order but it appears to be a complicated questions to answer. And the study referenced by Scott of adding Maraviroc+dolutegravir to suppressive therapy did not improve neuropsychologist testing or use depression but side effects were more frequent. There appear to be a lot of variables including timing of brain imaging evaluations, confounders line blood brain barrier impairment, if some PLWH have higher levels of ARTs in their brain, viral escape in the CNS. And I included 1 study on NAFLD but many studies report anti-inflammatory properties of Maraviroc. The question is if it does improve cognitive impairment & how much impact there is??? Jules Levin
MVC achieves levels in CSF within the range of IC50 or higher.
[study below] cART intensification with maraviroc in this open-label prospective pilot RCT of virally suppressed (in blood and CSF) HAND participants led to clinically relevant improvements in neurocognition, with a large effect observed after 6 months (d = 0.77) and moderate effect after 12 months (d = 0.55). There were no concomitant changes in 1H-MRS metabolites in FWM and basal ganglia or CSF neuroinflammatory biomarkers.......Conclusive evidence for cART intensification with a high CNS-penetrating antiretroviral drug is still lacking, but our study supports such a strategy. The detection of such effect sizes is encouraging, reinforcing the notion that some antiretrovirals are more beneficial for brain functioning and could even be neuroprotective for HAND, an increasingly important consideration given the effects of HIV on normal ageing processes remain relatively unknown [46].
Maraviroc has anti-inflammatory properties, good antiretroviral efficacy in cells including those of monocyte/macrophage lineage and good CNS penetration [26,27,41], highlighting its clear potential for therapeutic use. However, there is sparse evidence supporting its efficacy in reducing the severity of neurological conditions in HIV infection. To our knowledge, this study, albeit a pilot, provides the most convincing data supporting maraviroc intensification to date.
Maraviroc or Metformin Does Not Lower Liver Fat in People With HIV and NAFLD
Maraviroc, metformin, or both drugs did not lower liver fat percentage in a randomized trial of 90 people with HIV infection and nonalcoholic fatty liver disease (NAFLD) [1]. In fact, people who took maraviroc or metformin longer than 48 weeks ended up with a higher liver fat percentage than people treated for only 48 weeks.
HIV-positive people have a high prevalence of NAFLD, but no medication is licensed to treat this condition. Researchers at University College London and collaborators at other centers suggested that maraviroc (an antiretroviral CCR5 antagonist) and metformin (a diabetes drug) may address NAFLD-maraviroc through its impact on inflammation of the liver and system-wide inflammation, and metformin through its impact on free fatty acid metabolism and insulin sensitization.
Low-Dose Maraviroc, an Antiretroviral Drug, Attenuates the Infiltration of T Cells into the Central Nervous System and Protects the Nigrostriatum in Hemiparkinsonian Monkeys
Neuroprotective maraviroc monotherapy in simian immunodeficiency virus-infected macaques - reduced replicating and latent SIV in the brain
---------------------
New Paper published this week in journal - Remember this is in mice not humans
Maraviroc Restores Middle Age Memory Loss in Mice, "Could Be Used To Reverse Cognitive Deficits Caused by HIV" - (05/26/22)
Silva's lab demonstrated in earlier research that CCR5 expression reduced memory recall. In the current study, Silva and his colleagues discovered a central mechanism underlying mice's ability to link their memories of two different cages. A tiny microscope opened a window into the animals' brains, enabling the scientists to observe neurons firing and creating new memories.
Boosting CCR5 gene expression in the brains of middle-aged mice interfered with memory linking. The animals forgot the connection between the two cages.
When the scientists deleted the CCR5 gene in the animals, the mice were able to link memories that normal mice could not.
"blocking CCR5 with maraviroc ameliorates the memory linking deficits in middle-aged mice. Altogether these results suggest that CCR5 expression has a role in closing the temporal window for memory linking as well as in age-related deficits in memory linking.
In summary, our findings show that a delayed (12–24 h) increase in CCL5–CCR5 signalling in dCA1 neurons of a given memory ensemble closes the temporal window for memory linking. Activation of CCR5 decreases neuronal excitability, and thus negatively regulates memory allocation. This change in memory allocation decreases the overlap between memory ensembles, and therefore impairs the ability of one memory to trigger the recall of the other, thus closing the temporal window for memory linking (Extended Data Fig. 10).
Notably, our findings also show that an age-related increase in CCL5–CCR5 expression
leads to impairments in memory linking in middle-aged mice that can be reversed with an FDA-approved drug that inhibits this receptor—a result that could have substantial clinical implications. Together, our results provide the first insights into the molecular and cellular mechanisms that close the temporal window for memory linking, thus segregating the memories for events that are temporally distinct.”
--------------------
#1
Maraviroc-Enhanced cART Improves Cognition in Virally-Suppressed HAND: A Pilot Study
#2
RANDOMIZED CONTROLLED TRIAL OF MARAVIROC FOR HIV-ASSOCIATED NEUROCOGNITIVE IMPAIRMENT
Comparison of change from baseline to week 48 in the two arms (table) revealed significant improvement in the MVC arm in the Learning and Memory NPZ domain, but not in the other domains or global NPZ. The group difference in the Learning and Memory domain score did not survive adjustment for multiple comparisons, although the effect size was 0.89.
Conclusion: This preliminary randomized controlled study found no definitive evidence in favor of MVC intensification for HIV-associated NCI. Blood biomarker analyses are pending.
#3
ACTG A5324: A RANDOMIZED TRIAL OF ART INTENSIFICATION FOR COGNITIVE IMPAIRMENT IN PWH
Adding dolutegravir (DTG) with or without maraviroc (MVC) to suppressive antiretroviral therapy (ART) did not significantly improve neuropsychological test performance or ease depression compared with continuing the baseline regimen in ACTG A5324, the InMIND Trial [1]. In this double-blind, placebo-controlled trial enrolling 191 people with cognitive impairment, intensifying the regimen did boost CD4 and CD8 counts, but side effects were more frequent in people adding both dolutegravir and maraviroc to their regimen than in those adding just dolutegravir or those maintaining their initial ART.
$4
Maraviroc Intensification in Virally Suppressed HIV Infected Subjects Leads to Improvement in Neurocognitive Test Performance and Declines in Immune Activation 2013 CROI
#5
Maraviroc-intensified combined antiretroviral therapy improves cognition in virally suppressed HIV-associated neurocognitive disorder
Conclusion: This pilot RCT provides feasibility, tolerability, proof-of-concept and preliminary evidence for efficacy of the CCR5 co-receptor antagonist maraviroc in the management of HAND by improving neurocognitive functioning. This finding, should it be replicated in a larger study, has important implications for the clinical management of HAND by supporting maraviroc intensification as a preferred option to cautious monitoring of participants over time.
Abstract
Objective: To investigate whether intensification of combined antiretroviral therapy (cART) with the CC chemokine receptor type 5 (CCR5) entry inhibitor maraviroc leads to improvement in global neurocognitive functioning in virally suppressed men with HIV-associated neurocognitive disorder (HAND).
Design: Prospective, double observer-blinded, open-label pilot randomized-controlled trial. Participants were randomized to remain on their existing cART regimen (control arm; n = 8) or receive maraviroc-intensification (maraviroc arm; n = 9).
Methods: Participants completed a five-domain neuropsychological battery at baseline, 6- and 12-month visits. Raw scores were transformed into age-corrected z-scores and averaged into a global z-score. Single voxel (1H)-magnetic resonance spectroscopy (MRS) major cerebral metabolite concentrations were collected at baseline and 12 months in the basal ganglia and frontal white matter and quantified using jMRUI. Neuroinflammatory biomarkers cerebrospinal fluid neopterin and β2-microglobulin were also measured.
Results: Fourteen of the 17 participants completed the study: nine maraviroc arm and five control. We found medium to large effect sizes in favour of improved global neurocognitive performance in the maraviroc arm over time {arm*time interaction: P < 0.05; 6 month: [β=-0.10, standard error (SE)= 0.04, 90% confidence interval (90%CI)= -0.18,.03; P < 0.03] yielding a large effect-size d = 0.77 (90%CI = -0.19,1.71); 12 month: [β=-0.01; SE = 0.05; 90%CI = -0.09, 0.06; P < 0.77] yielding a moderate effect-size d = 0.55 (90%CI = -0.47,1.55)}. No treatment-related changes were detected for 1H-MRS metabolites or cerebrospinal fluid biomarkers.
Secondary endpoint I
H-MRS analysis with jMRUI did not reveal any treatment effects on metabolites of interest in each brain region (P > 0.30 for all arm*time interaction terms).
Secondary endpoint II
CSF neopterin levels were below detectable range (i.e. <8 nmol/l) in five of 22 (23%) specimens analyzed. No relevant effects were detected for CSF neopterin (arm*time interaction: P= 0.82) or b2-microglobulin levels (arm*time interaction: P ¼ 0.71).
Conclusion: This pilot study provides feasibility, tolerability, proof-of-concept and preliminary evidence for clinically relevant neurocognitive improvement in cART enhancement with maraviroc in virally suppressed HAND patients. Lack of concomitant brain metabolite and biomarker change may be related to complex dynamics of brain repair.
-------------------------
Changes in cerebral function parameters with maraviroc-intensified antiretroviral therapy in treatment naive HIV-positive individuals
Background:
Maraviroc-intensified antiretroviral therapy (ART) may be associated with cognitive benefits.
Methods:
Therapy-naive, cognitively asymptomatic, HIV-positive individuals were randomly allocated on a 1 : 1 basis to standard ART (Arm1: tenofovir-emtricitabine and atazanavir/ritonavir) or maraviroc intensified ART (Arm2: abacavir-lamivudine and darunavir/ritonavir/maraviroc). Over 48 weeks, detailed assessments of cognitive function tests were undertaken and cerebral metabolites measured using proton magnetic resonance spectroscopy. Our primary endpoint was mean change in cognitive function across treatment arms with factors associated with cognitive function changes also assessed.
Results:
Of 60 individuals randomized (30 Arm1 and 30 Arm2), 58 were men and 44 of white ethnicity. Treatment groups had similar disease characteristics including overall mean (SD) baseline CD4+ cell count 428 (209) and 414 (229) cells/μl, Arms1 and 2, respectively. At week 48, plasma HIV RNA was less than 50 copies/ml in 55 of 56 of those completing study procedures. Cognitive function improved over 48 weeks [mean change z-score (SD) 0.16 (0.09) Arm1 and 0.25 (0.08) Arm2, P = 0.96 for differences between study arms]. A greater increase in frontal grey matter N-acetyl aspartate/creatine ratio was observed in Arm1 [ratio change of 0.071 (SD 0.16)] versus Arm2 [change -0.097 (SD 0.18), P = 0.009], although this was not associated with changes in cognitive function (P = 0.17).
Conclusion:
Maraviroc-intensified ART had no demonstrable benefit on cognitive function in individuals initiating ART. Greater improvement in neuronal metabolites (N-acetyl aspartate/creatine) was observed with standard ART. Future work should focus on maraviroc-intensified ART in individuals with cognitive impairment.
----------------------
BBB impairment is a common finding in PLWH affected by neurological or neurocognitive disorders either with or without suppressive cART. Blood–brain barrier impairment (BBBi) may play a crucial role in the pathogenesis of HAND. In this regard, up to 22% of asymptomatic HIV-positive subjects, 50% of patients suffering from AIDS and up to 100% of patients with HIV-associated dementia (HAD) showed an increased BBB permeability [4]BBBi was observed in 35.4% naïve and in 22.7% treated participants; the use of integrase inhibitors was associated with a lower prevalence (18.3 vs. 30.9%, p = 0.050. In this study cART therapy resulted as an independent protective factor of BBBi. At multivariate binary logistic regression (including age and sex) nadir CD4 cell count (p = 0.034), presence of central nervous system (CNS) opportunistic infections (p = 0.024) and cerebrospinal fluid (CSF) HIV RNA (p = 0.002) in naïve participants and male sex (p = 0.021), a history of CNS opportunistic infections (p = 0.001) and CSF HIV RNA (p = 0.034) in treated patients were independently associated with BBBi. We have also identified female sex and cART therapy as independent protective factors for BBBi. Indeed, BBB plays a critical role in CNS physiology and in neurological disorders, such as neurodegenerative disorders. We observed higher levels of HIV RNA and neopterin in the CSF of study participants with BBBi, highlighting the significant role of inflammation in the pathogenesis of BBBi. Aside from virological control, female sex was found to be protective: further studies are needed to confirm this finding and to understand the long–term consequences of an impaired BBB in PLWH. BBBi has been observed despite cART and it has been associated with neuronal damage
biomarkers, such as increased levels of total tau and phosphorylated tau.
----------------------------------
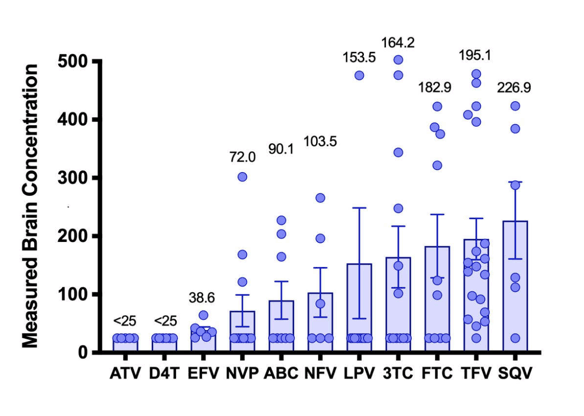
In this project, ART drug concentrations were measured in brain tissue that was collected at autopsy from adults who died with HIV disease..
The
association between higher drug concentrations in the brain and worse neurocognitive performance may indicate ART neurotoxicity. The direction of the relationship with neurocognitive performance, however, indicates that this may come at a cost: higher drug concentrations in brain were associated with worse neurocognitive performance. This is consistent with recent concerns about ART neurotoxicity. CONCLUSIONS: The manuscript reports ART drug concentrations from human brain tissue collected at autopsy, finding that they exceed published ART drug concentrations in CSF and that higher concentrations are associated with viral suppression in blood, longer duration of HIV infection and its treatment, and worse global neurocognitive performance. Larger studies are needed in which modern ART drugs are quantified in CSF, brain tissue, and blood to validate these pilot findings and to better understand the mechanisms of different ART distribution patterns into the CNS.
Maraviroc Restores Middle Age Memory Loss in Mice, "Could Be Used To Reverse Cognitive Deficits Caused by HIV" - (05/26/22)
----------------------------------
Maraviroc-intensified combined antiretroviral therapy improves cognition in virally suppressed HIV-associated neurocognitive disorder
Conclusion: This pilot RCT provides feasibility, tolerability, proof-of-concept and preliminary evidence for efficacy of the CCR5 co-receptor antagonist maraviroc in the management of HAND by improving neurocognitive functioning. This finding, should it be replicated in a larger study, has important implications for the clinical management of HAND by supporting maraviroc intensification as a preferred option to cautious monitoring of participants over time.
Abstract
Objective: To investigate whether intensification of combined antiretroviral therapy (cART) with the CC chemokine receptor type 5 (CCR5) entry inhibitor maraviroc leads to improvement in global neurocognitive functioning in virally suppressed men with HIV-associated neurocognitive disorder (HAND).
Design: Prospective, double observer-blinded, open-label pilot randomized-controlled trial. Participants were randomized to remain on their existing cART regimen (control arm; n = 8) or receive maraviroc-intensification (maraviroc arm; n = 9).
Methods: Participants completed a five-domain neuropsychological battery at baseline, 6- and 12-month visits. Raw scores were transformed into age-corrected z-scores and averaged into a global z-score. Single voxel (1H)-magnetic resonance spectroscopy (MRS) major cerebral metabolite concentrations were collected at baseline and 12 months in the basal ganglia and frontal white matter and quantified using jMRUI. Neuroinflammatory biomarkers cerebrospinal fluid neopterin and β2-microglobulin were also measured.
Results: Fourteen of the 17 participants completed the study: nine maraviroc arm and five control. We found medium to large effect sizes in favour of improved global neurocognitive performance in the maraviroc arm over time {arm*time interaction: P < 0.05; 6 month: [β=-0.10, standard error (SE)= 0.04, 90% confidence interval (90%CI)= -0.18,.03; P < 0.03] yielding a large effect-size d = 0.77 (90%CI = -0.19,1.71); 12 month: [β=-0.01; SE = 0.05; 90%CI = -0.09, 0.06; P < 0.77] yielding a moderate effect-size d = 0.55 (90%CI = -0.47,1.55)}. No treatment-related changes were detected for 1H-MRS metabolites or cerebrospinal fluid biomarkers.
Secondary endpoint I
H-MRS analysis with jMRUI did not reveal any treatment effects on metabolites of interest in each brain region (P > 0.30 for all arm*time interaction terms).
Secondary endpoint II
CSF neopterin levels were below detectable range (i.e. <8 nmol/l) in five of 22 (23%) specimens analyzed. No relevant effects were detected for CSF neopterin (arm*time interaction: P= 0.82) or b2-microglobulin levels (arm*time interaction: P ¼ 0.71).
Conclusion: This pilot study provides feasibility, tolerability, proof-of-concept and preliminary evidence for clinically relevant neurocognitive improvement in cART enhancement with maraviroc in virally suppressed HAND patients. Lack of concomitant brain metabolite and biomarker change may be related to complex dynamics of brain repair.
cART intensification with maraviroc in this open-label prospective pilot RCT of virally suppressed (in blood and CSF) HAND participants led to clinically relevant improvements in neurocognition, with a large effect observed after 6 months (d = 0.77) and moderate effect after 12 months (d = 0.55). There were no concomitant changes in 1H-MRS metabolites in FWM and basal ganglia or CSF neuroinflammatory biomarkers.......Conclusive evidence for cART intensification with a high CNS-penetrating antiretroviral drug is still lacking, but our study supports such a strategy. The detection of such effect sizes is encouraging, reinforcing the notion that some antiretrovirals are more beneficial for brain functioning and could even be neuroprotective for HAND, an increasingly important consideration given the effects of HIV on normal ageing processes remain relatively unknown [46].
Maraviroc has anti-inflammatory properties, good antiretroviral efficacy in cells including those of monocyte/macrophage lineage and good CNS penetration [26,27,41], highlighting its clear potential for therapeutic use. However, there is sparse evidence supporting its efficacy in reducing the severity of neurological conditions in HIV infection. To our knowledge, this study, albeit a pilot, provides the most convincing data supporting maraviroc intensification to date.
By employing a randomized-controlled design, longer follow-up period, and optimal neuropsychological methods for longitudinal research, this study supports and extends recent observations of improved neurocognition in a small sample (n = 6) of HIV-infected participants with some degree of cognitive impairment who underwent maraviroc intensification for 24 weeks [28]. That study also reported partial reversal of monocyte-mediated pathological changes previously associated with neurocognitive impairment, namely reducing the proportion of circling intermediate and nonclassical CD16-expressing monocytes, CD14+ HIV DNA monocyte burden and pro-inflammatory biomarker sCD163 levels in plasma [47-50]. However, the authors did not directly examine whether changes observed in plasma also occurred in the CNS.
Figure 3 displays mean change in global neurocognitive functioning in the study arms at each time-point. There were no baseline differences between the two arms (P < 0.71). Medium to large effect sizes were observed favouring improved global neurocognitive functioning in maraviroc arm over control arm over time [arm*time interaction: P < 0.05; 6 months: β = -0.10, standard error (SE) = 0.04, 90% CI = -0.18, 0.03; P < 0.03; 12 months: β = -0.01, SE = 0.05, 90% CI = -0.09, 0.06; P < 0.77] yielding a large between-groups effect-size at 6 months: (d = 0.77; 90% CI = -0.19,1.71) and medium effect-size at 12 months (d = 0.55; 90% CI:-0.47, 1.55). CVs comparing baseline to follow-up visits did not differ between study arms (Table 2).
Demographic and clinical information for the sample are presented in Table 1. Overall, the sample primarily comprised middle-aged (M = 55 years) White males with average education level (M = 12.1 years) and premorbid intelligence (FSIQ: M = 103). Nadir CD4+ cell count was less than 200 cells/μl in eight of 14 (57%) participants.
Current CD4+ cell count was above 200 cells/μl in all participants with eight of 14 (57%) CD4+ cell count below 500 cells/μl at baseline and seven of 13 (54%) at 12 months. No significant differences were found between study arms on any of these variables, although trends were observed for higher baseline CD4+ cell counts in controls (P < 0.06), whereas maraviroc participants were almost exclusively MND (89%) compared with a larger spread of HAND severity in controls (P < 0.09).
Two maraviroc and one control had confirmed CXCR4-tropic virus in blood; one control had confirmed CCR5-tropic virus. HIV-tropism status was unavailable for other participants as their HIV RNA fell below the lower limit of assay detection and could not be amplified by PCR for V3-loop DNA. All participants had undetectable HIV RNA (<50 cpml) at baseline per inclusion criteria. However, at 12 months one maraviroc had detectable plasma and CSF HIV RNA whereas one control had mildly elevated plasma HIV RNA with undetectable CSF HIV RNA. Both had CXCR4-tropic virus and the maraviroc participant self-reported antiretroviral nonadherence in the week preceding the study visit.
|
|
| |
| |
|
|
|