| |
Efficacy and Safety of Further Lowering of Low-Density
Lipoprotein Cholesterol in Patients Starting With Very Low Levels
A Meta-analysis
|
| |
| |
Download the PDF here
Download the PDF here
These data suggest further lowering of LDL-C thresholds for initiating more intensive therapy, or simply targeting LDL-C at least as low as approximately 0.5 mmol/L or 20 mg/dL, would further reduce cardiovascular risk.
Extending observations made with statins, we found consistent clinical benefit from further LDL-C lowering in patient populations starting as low as a median of 1.6 mmol/L (63 mg/dL) and achieving levels as low as a median of 0.5 mmol/L (21 mg/dL). Specifically, when examining 11 492 major vascular events, there was a 21% relative risk reduction per 1-mmol/L (38.7-mg/dL) reduction in LDL-C through this range. This relative risk reduction is virtually the same as the 22% reduction seen in the overall CTTC analysis in which the starting LDL-C was nearly twice as high.3 Moreover, these data parallel observational data showing progressively greater coronary atherosclerotic plaque regression and progressively lower adjusted risk of major vascular events with progressively LDL-C levels down to less than 0.2 mmol/L (7 mg/dL).9,10 Furthermore, there was no evidence of an increased incidence of adverse events with lowering LDL-C to such levels. These levels are considerably lower than the targets or thresholds for additional nonstatin LDL-C–lowering therapy in current cholesterol guidelines that, for high-risk patients, range from 1.8 mmol/L to 2.6 mmol/L (70 mg/dL to 100 mg/dL).11-13
For example, in patients starting with an LDL-C level of 2.6 mmol/L (100 mg/dL), a 60% decrease in LDL-C (what a PCSK9 inhibitor typically achieves) should lower LDL-C by 1.6 mmol/L (60 mg/dL), reduce the relative risk of major vascular events by 31%, and, assuming a 5-year major vascular event rate of 25% in a secondary prevention population, yield an absolute risk reduction of 7.8%. If the same patients started with an LDL-C level of 1.8 mmol/L (70 mg/dL), LDL-C should be lowered by 1.1 mmol/L (42 mg/dL) and the relative risk of major vascular events by 23%, which would yield an absolute risk reduction of 5.8%.
The clinical benefit per millimoles per liter reduction in LDL-C was virtually identical for statins, ezetimibe, PCSK9 inhibition, and CETP inhibition, despite these drugs having different effects on other risk markers such as high-density lipoprotein cholesterol, lipoprotein(a), and high-sensitivity C-reactive protein. This observation reinforces the notion that the reduction in LDL-C (or more broadly, atherogenic apolipoprotein B–containing particles) is the primary driver of clinical benefit.
press release: The study was statistically powered around the hard major adverse cardiovascular event (MACE) composite endpoint of first heart attack, stroke or cardiovascular death (key secondary composite endpoint) and found that adding Repatha to optimized statin therapy resulted in a statistically significant 20 percent (p<0.001) reduction in these events. The robust benefit in this objective measure started as early as six months and continued to accrue through the median 2.2 years of the study. In fact, the magnitude of the risk reduction in the hard MACE composite endpoint grew over time, from 16 percent in the first year to 25 percent beyond the first year.
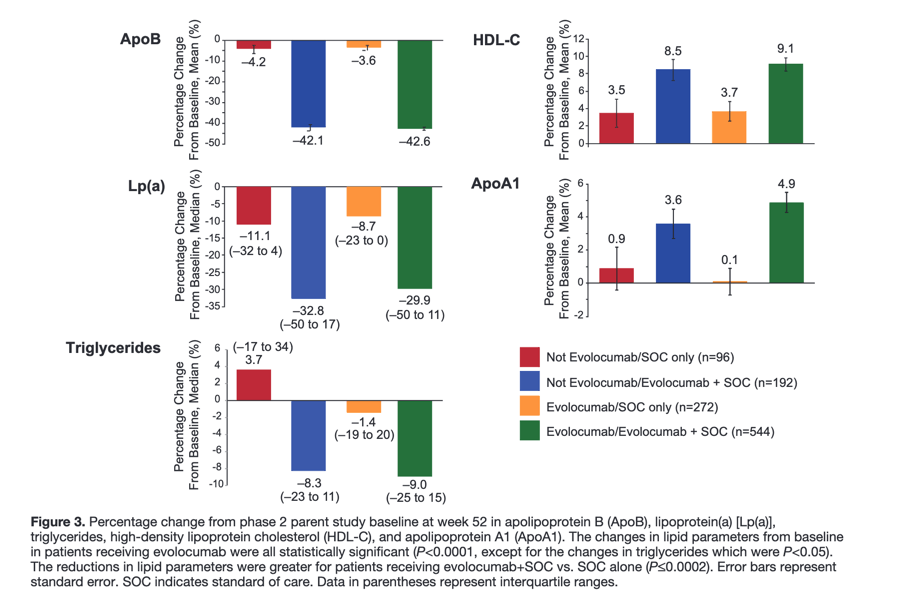
When added to statin therapy, Repatha reduced LDL-C from a median of 92 to 30 mg/dL, a reduction of 59 percent at week 48, which was sustained throughout the trial. At 48 weeks, the LDL-C was reduced to at least 25 mg/dL in 42 percent of patients treated with Repatha, as compared with <0.1 percent in the placebo group (p<0.001). Additionally, treatment with Repatha had favorable effects on other lipid parameters.
There were no significant changes in C-reactive protein, mean levels being 1.7 mg/L at baseline and 1.4 mg/L in both evolocumab and placebo groups at 48 weeks. This finding suggests that evolocumab lacks an anti-inflammatory action as opposed to statins that consistently decrease C-reactive levels reflecting their anti-inflammatory effect [7]. The lack of effect of evolocumab on C-reactive protein appears to be a class effect because a meta-analysis of 16 trials of various PCSK9 monoclonal antibodies, including evolocumab, also showed no effect on C-reactive protein [8].
Addition of the PCSK9 inhibitor evolocumab to statin therapy produced greater low-density lipoprotein cholesterol lowering and atheroma regression. The LDL-C level of less than 50 mg/dl is considered low while a level of less than 20 mg/dl is considered extremely low. Intensive lipid lowering treatment has been found to halt the progression of atherosclerosis as compared to moderate lipid lowering treatment. It also regresses the atheroma plaque volume as reported by the REVERSAL (Reversal of Atherosclerosis with Aggressive Lipid Lowering) trial and ASTEROID (A Study to Evaluate the Effect of Rosuvastatin on Intravascular Ultrasound-Derived Coronary Atheroma Burden) trial. SATURN (Study of Coronary Atheroma by Intravascular Ultrasound: Effect of Rosuvastatin Versus Atorvastatin) trial also supports this [15]. An LDL level below 2.5 mmol/l can cause an atherosclerotic plaque to regress. Similarly, GLAGOV trial reported that patients who received evolocumab on a baseline treatment with statins demonstrated plaque regression in a larger number of patients as compared to placebo (64.3% versus 47.3%) after 76 weeks of therapy [16]. In a retrospective analysis, coronary calcium score was reduced with the aggressive lowering of LDL [17]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5937425/#B16
Individuals carrying PCSK9 loss of function mutations not only have naturally low LDL‐C levels but also have reduced CVD risk.31, 33 The Dallas Heart Study followed 12 887 individuals for 15 years, including carriers of specific PCSK9 loss of function mutations that in this study were associated with absolute LDL‐C levels of approximately 2.5 mmol/L (100 mg/dL). These PCSK9 loss of function mutation carriers exhibited a low incidence of CHD. Compared with non‐carriers, the CHD risk reduction was 88% and 47% in Black and White PCSK9 loss of function mutation carriers, respectively. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5215677/
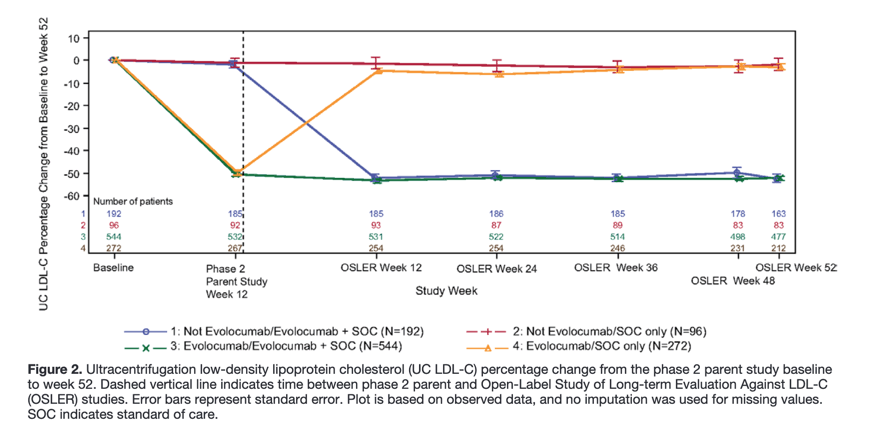
A total of 411 patients (99.5% of whom were receiving evolocumab) achieved LDL-C <1.3 mmol/L (50 mg/dL), and 98 patients (all of whom were receiving evolocumab) achieved LDL-C <0.65 mmol/L (25 mg/dL), whereas 682 patients (47.4% of whom were receiving evolocumab) had LDL-C levels ≥1.3 mmol/L (50 mg/dL). Achievement of ultracentrifugation LDL-C levels of <1.3 mmol/L (50 mg/dL) or <0.65 mmol/L (25 mg/dL) at any OSLER visit occurred in 55.6% and 13.3% of evolocumab+SOC patients, respectively.
Patients not taking evolocumab in the phase 2 parent study had large initial LDL-C reductions determined at 12 weeks after starting evolocumab treatment in OSLER (51.8% [SE, 1.6%] reduction from the parent study baseline; P<0.0001 versus baseline), with reductions maintained over the 52-week study period (52.3% [SE, 1.8%] at week 52; P<0.0001 versus baseline; Figure 2 and Table II in the online-only Data Supplement).
For patients who discontinued evolocumab after random assignment to SOC in OSLER, mean reductions in calculated LDL-C from baseline went from 53.1% [SE, 1.2%] at the start of OSLER to 17.9% [SE, 1.2%] at 4 weeks and then to near baseline levels (5.8% [SE, 1.2%]) within 12 weeks without a rebound effect.
Among the evolocumab+SOC group patients, of those who achieved LDL-C <1.3 mmol/L (50 mg/dL), 76.0% used statins at baseline, and of those who achieved LDL-C <0.65 mmol/L (25 mg/dL), 86.7% took statins. Statin use was 51.1% in patients with LDL-C levels ≥1.3 mmol/L (50 mg/dL). Among SOC patients, 1 of the 2 patients who reached LDL-C <1.3 mmol/L (50 mg/dL) and 57.9% of those with LDL-C ≥1.3 mmol/L (50 mg/dL) were taking statins at baseline.
Evolocumab reduced LDL-C on average by ≈50% beyond the reduction achieved with optimal SOC in various hypercholesterolemic patient populations. The reductions in lipid levels achieved after 12 weeks were stable over the course of the study without evidence of attenuation. Conversely, discontinuation of evolocumab led to a fairly rapid return to baseline levels, although importantly, without a rebound phenomenon.
|
|
| |
| |
|
|
|