| |
Effectiveness of Paxlovid in Reducing Severe Coronavirus Disease 2019 and Mortality in High-Risk Patients
|
| |
| |
Download the PDF here
Download the PDF here
Clinical Infectious Diseases 02 June 2022
To minimize confounding by indication, inclusion in this study was limited to patients, who were potentially candidates for Paxlovid treatment, with at least 1 comorbidity or condition associated with high risk for severe COVID-19, as in the EPIC-HR trial, including age ≥60 years, body mass index (BMI) ≥30 kg/m2, diabetes, hypertension, cardiovascular disease, chronic liver disease, chronic lung disease, chronic kidney disease, neurological disease, immunosuppression, and malignancy [12].
our study showed that COVID-19 vaccine is independently associated with a significant decrease in the risk of severe COVID-19 and mortality with an estimated relative risk reduction of 80%.
Despite the shown effectiveness, it should be noted that Paxlovid is not recommended in several medical conditions and has complex drug-drug interactions, due to the ritonavir component of the combination. Therefore, clinicians should carefully review the patient’s concomitant medications and evaluate potential drug-drug interactions. This may limit the use of Paxlovid in high-risk patients, for whom other alternative antivirals might be appropriate [19-21].
Results
Overall, 180 351 eligible patients were included; of these, only 4737 (2.6%) were treated with Paxlovid, and 135 482 (75.1%) had adequate COVID-19 vaccination status. Both Paxlovid and adequate COVID-19 vaccination status were associated with significant decrease in the rate of severe COVID-19 or mortality with adjusted HRs of 0.54 (95% confidence interval [CI], .39-.75) and 0.20 (95% CI, .17-.22), respectively. Paxlovid appears to be more effective in older patients, immunosuppressed patients, and patients with underlying neurological or cardiovascular disease (interaction P < .05 for all). No significant interaction was detected between Paxlovid treatment and COVID-19 vaccination status.
This analysis suggests that Paxlovid appears to be more effective in older patients, patients with cardiovascular disease, patients with neurological disease, and in immunosuppressed patients (P value for interaction <.05 for all). The magnitude of Paxlovid effectiveness appears to be unrelated to COVID-19 vaccination status (P value for interaction = .129) (Figure 3).
In summary, this study suggests that treatment with Paxlovid is associated with prominent reduction of severe COVID-19 and mortality in real life and in the era of Omicron, especially in older patients. This study further confirms that COVID-19 vaccine remains the most effective intervention to prevent disease progression and death among COVID-19 patients.
Effectiveness of Paxlovid
Overall, 942 events occurred in all 180 351 included patients, reflecting a crude incidence rate of 5.6 per 1000 person-months. The crude incidence rate was 3.4 per 1000 person-months in vaccinated patients and 13.4 per 1000 person-months in unvaccinated patients. A total of 39 events occurred in the 4737 patients treated with Paxlovid, reflecting a crude incidence rate of 10.4 per 1000 person-months, whereas 903 events occurred in the 175 614 patients not treated with Paxlovid, reflecting a crude incidence rate of 5.6 per 1000 person-months. The higher crude incidence rate in treated patients is likely attributed to older age and higher frequency of underlying comorbidities (Table 1). Indeed, Paxlovid was independently associated with a significantly decreased risk for the composite of severe COVID-19 or mortality, in a multivariable Cox regression models, with an HR of .54 (95% CI, .39-.75). Adequate COVID-19 vaccination status was also associated with significantly decreased risk for the composite of severe COVID-19 or mortality (HR, 0.20 [95% CI, .17-.22]) (Table 2).
A sensitivity analysis, restricted to patients diagnosed with COVID-19 after mid-January, when the Omicron variant was the prevailing circulating variant, included 146 228 eligible patients, of whom 3761 were treated with Paxlovid. This analysis shows that Paxlovid was associated with greater decrease in the composite of severe COVID-19 and mortality (HR, 0.43 [95% CI, .85-.64]) (Supplementary Table 2).
Age, male sex, low SES, belonging to the Arab population sector, diabetes, cardiovascular disease, chronic lung disease, chronic kidney disease, neurological disease, malignancy, and immunosuppression were retained in the Cox multivariable model and were all associated with increased risk for the composite of severe COVID-19 or mortality. Of them, immunosuppression, neurological disease, and malignancy had the strongest association, with HRs of 6.43 (95% CI, 4.95-8.34), 2.28 (95% CI, 1.95-2.67), and 2.15 (95% CI, 1.54-3.00), respectively. Age was also a strong risk factor with an HR of 2.28 (95% CI, 2.16-2.41) for each 10-year increase (Table 2).
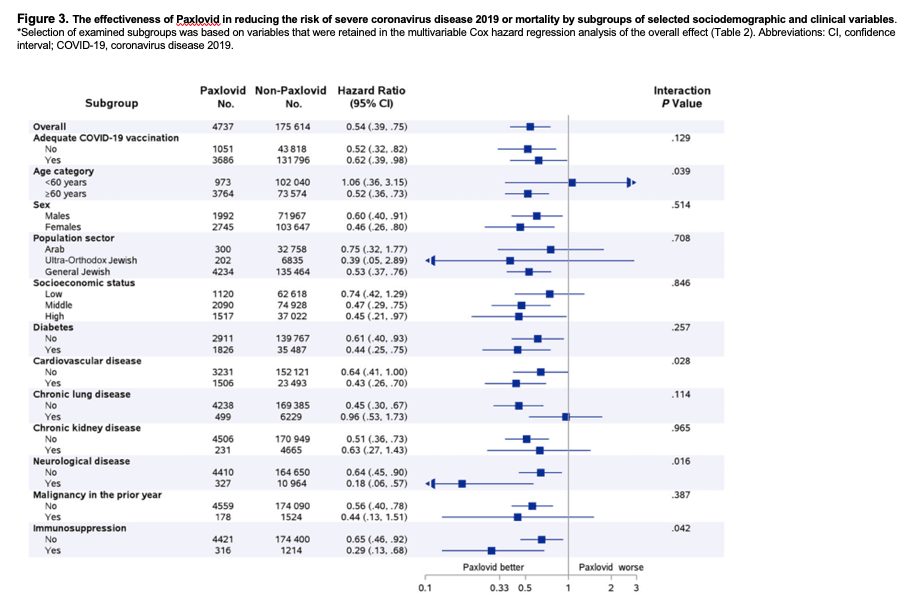
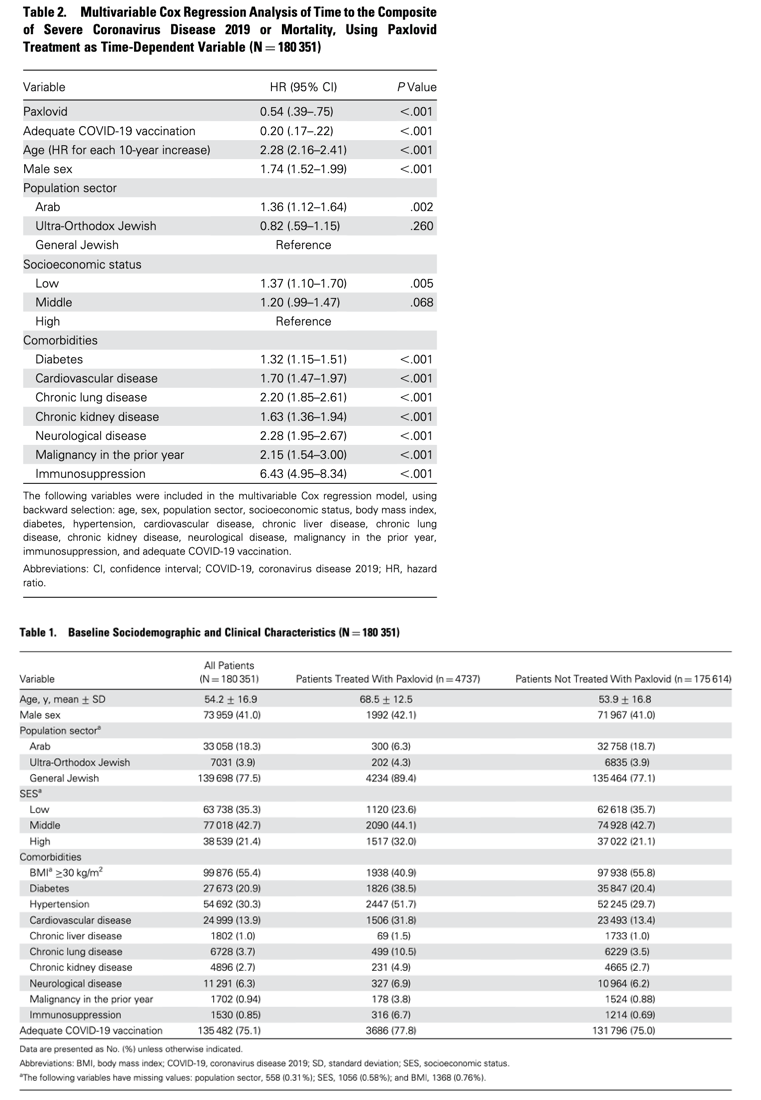
------------------------------------------------
Safety and Tolerability of Paxlovid (Nirmatrelvir/Ritonavir) in High-risk Patients
Clinical Infectious Diseases 23 July 2022
to the editor-We read with great interest the analysis by Saliba and colleagues on the real-world experience with nirmatrelvir/ritonavir (Paxlovid) in reducing severe coronavirus disease 2019 (COVID-19) and mortality in high-risk patients [1]. This is the largest retrospective cohort study to date that demonstrated Paxlovid treatment and vaccination status were associated with significant decrease in the rate of severe COVID-19 or mortality with adjusted hazard ratio (HR) 0.54 (95% confidence interval, 0.39-0.75) and 0.2 (95% confidence interval, 0.17-0.22), respectively. Timely real-world data are important to evaluate the effectiveness of Paxlovid in populations outside of the clinical trial, but the authors were unable to assess medication tolerability and safety.
As of 14 May 2022, Assistant Secretary for Preparedness and Response estimated 668 954 Paxlovid courses were given in the United States [2]. In the randomized clinical trial, EPIC-HR (Evaluation of Protease Inhibition for Covid-19 in High-Risk Patient), Paxlovid appeared to be well tolerated but real-world data are lacking [3]. At the University of Washington Medicine, we developed a standardized process to prioritize high-risk patients meeting National Institutes of Health Tier 1 and 2 criteria and assessed patient tolerability and safety through telephonic postprescription outreach when supplies were initially limited. Patients who were prescribed Paxlovid were interviewed over the phone using a standardized questionnaire to assess adverse effects and adherence within 1 week of starting therapy.
Between January 5 and 21, 2022, 50 patients were prescribed Paxlovid (Table 1). The median age was 48 years, with 64% of females and 20% of Latino/Hispanic ethnicity. The majority (82%) had immunocompromising conditions with a median of 5 comorbid conditions and 8.5 concomitant medications. Sixteen (32%) patients required chronic medication adjustment while taking Paxlovid. All patients were contacted within 1 week of starting Paxlovid, 19 (38%) and 21 (42%) patients were interviewed during therapy and after completion of therapy, respectively, whereas 9 (18%) patients were lost to follow-up and 1 (2%) patient did not start Paxlovid. Among the 40 respondents, 34 (85%) had experienced at least 1 adverse effect. Twelve (30%) patients reported ≥ 3 adverse effects and all were ≥ 5 concomitant chronic medications. The most frequently reported adverse effect was dysgeusia (57.5%). Most patients reported the onset of 1 to 2 hours after taking Paxlovid and usually subsided within 24 hours after discontinuation of therapy.
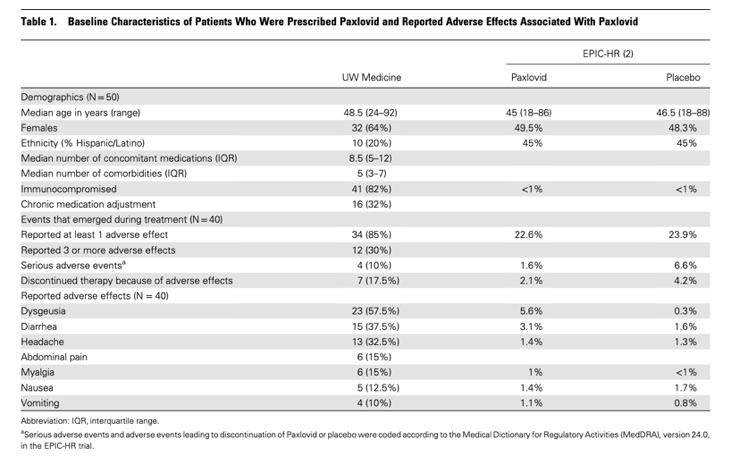
We reported a high incidence of adverse effects with 85% of patients reporting at least 1 side effect while taking Paxlovid leading to 17.5% of patients discontinuing therapy prematurely compared with 2% observed in the EPIC-HR trial. Most notably, dysgeusia was reported in 57.5% of patients compared to 5.6% in clinical trial [3]. We used this information to better prepare our patients for anticipated side effects and provided mitigation strategies based on our evaluation. To the best of our knowledge, this is the first report to date evaluating the tolerability and safety of Paxlovid through telephonic postprescription outreach in the real-world setting. This underscores the importance of close monitoring and postmarketing surveillance with medications that are still under investigation.
|
|
| |
| |
|
|
|