| |
Transmitted drug resistance to integrase based first-line
HIV antiretroviral regimens in the Mediterranean Europe
|
| |
| |
Download the PDF here
26 December 2022
Abstract
Objective
To study the prevalence of transmitted drug resistance (TDR) to INSTIs and NRTIs, and of clinically relevant resistance (CRR), in newly-diagnosed people with HIV (PWH) naïve to antiretroviral therapy (ART) in Europe.
Methods
MeditRes HIV is a consortium that includes ART naïve PWH newly diagnosed in France, Greece, Italy, Portugal, and Spain during the years 2018-2021. Reverse transcriptase (RT) and Integrase (INSTI) sequences were provided by participating centers. To evaluate the prevalence of surveillance drug resistance mutations (SDRM) we used the CPR tools from Stanford HIV-website. To evaluate clinically relevant resistance (CRR), defined as any resistance level >= 3, we used the Stanford v.9.1HIVDB Algorithm.
Results
We included 2705 PWH, 72% men, median age of 37 (IQR, 30-48); 43.7% infected by non-B subtypes.
The prevalence of INSTI-SDRMs was 0.30% (T66I, T66A, E92Q, E138T, E138K, Y143R, S147G and R263K, all n = 1), and of NRTI-SDRMs was 5.77% (M184V n = 23, 0.85%; M184I n = 5, 0.18%; K65R/N n = 3, 0.11%; K70E n = 2, 0.07%; L74V/I n = 5, 0.18%; any TAMs n = 118, 4.36%).
INSTI-CRR was 2.33% (0.15% dolutegravir/bictegravir; 2.29% raltegravir/elvitegravir), and 1.74% to first-line NRTIs (0.89% tenofovir/tenofovir alafenamide fumarate; 1.74% abacavir; 1.07% lamivudine/emtricitabine).
Conclusions
We present the most recent data on TDR to integrase based first-line regimens in Europe. Given the low prevalence of CRR to second generation integrase inhibitors and to first-line NRTIs, in the years 2018-2021 it is unlikely that newly diagnosed PWH in MeditRes countries would present with baseline resistance to a first-line regimen based on second generation integrase inhibitors.
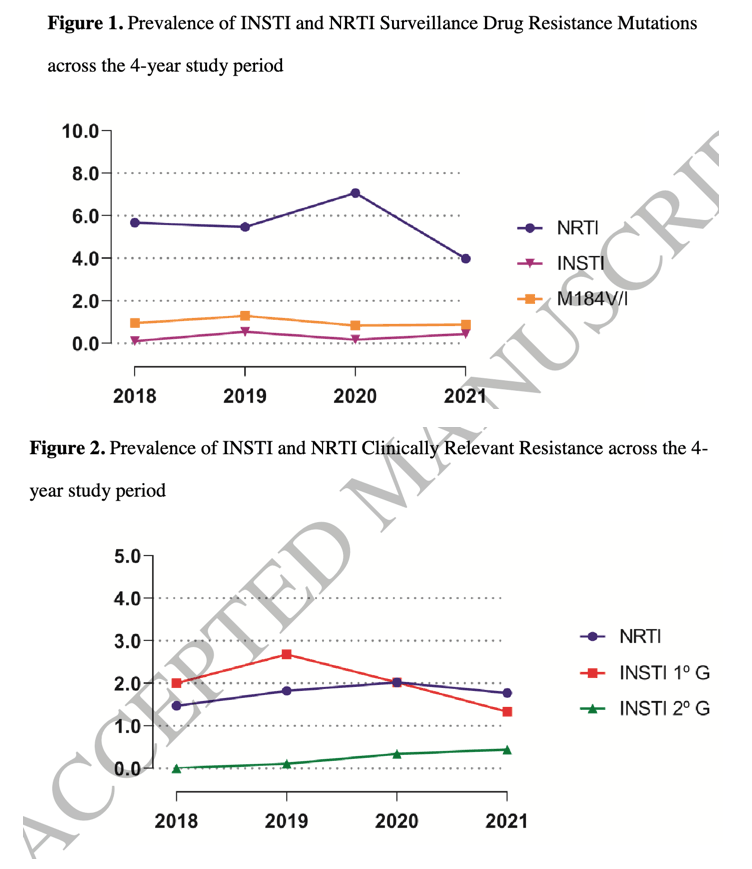
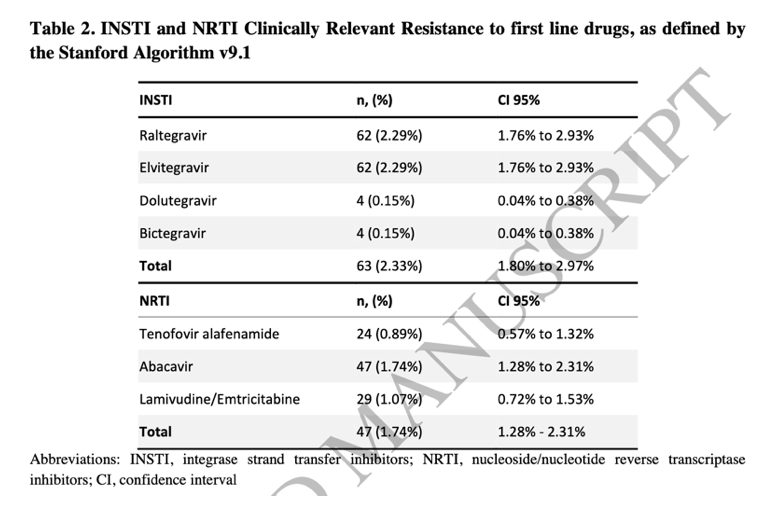
|
|
| |
| |
|
|
|