 |
 |
 |
| |
Arterial remodeling is associated with CD4 T cell count and plasma IL-10 in persons with HIV
|
| |
| |
AIDS 2022 July 29-Aug 2 Montreal

The presentation of coronary atherosclerosis can be gradual, because of progressive flow-limiting stenosis and exertional angina, or dramatic, with plaque rupture and thrombosis causing unstable angina, myocardial infarction, or sudden death. The importance of arterial remodeling, or persistent change in vessel size, has recently become apparent in both situations. Arterial remodeling, not plaque size, has been identified as the primary determinant of lumen size in the presence of stable lesions. Similarly, luminal stenosis in transplant vasculopathy and with restenosis after angioplasty occur mainly because of inward remodeling rather than plaque growth. However, recent evidence also suggests that adequate outward remodeling may be associated with an increased risk of plaque rupture, the underlying cause of acute coronary syndromes and sudden cardiac death.
The term “arterial remodeling” has previously been used to describe any change in vessel wall structure. More recently, however, it has been used specifically to refer to a change in vessel size (or cross-sectional area within the external elastic lamina), and it is on this entity that this review is focused. Inward remodeling denotes a reduction in vessel size. Outward remodeling denotes an increase in vessel size. Various other terms are used in the literature (the Table). When outward remodeling is present but insufficient to prevent luminal stenosis, it is referred to as inadequate outward remodeling.
Remodeling as a Primary Determinant of Lumen Size
It has been known for more than a century1 that blood vessels enlarge to accommodate increasing flow to the organ downstream (eg, during natural growth or in left ventricular hypertrophy). Widespread interest in this phenomenon was stimulated by observations that radial enlargement of vessels (outward remodeling) can compensate for progressive growth of atherosclerotic plaques, thus postponing the development of flow-limiting stenosis.23 These pathological findings were subsequently supported by in vivo intravascular ultrasound (IVUS) studies that revealed just how ubiquitously outward remodeling occurs in the presence of atheroma and how such outward remodeling could hide sizeable plaques from angiographic detection.45 https://www.ahajournals.org/doi/10.1161/01.CIR.102.10.1186
Presenter
Ayoda Werede
Authors
A. Werede * (1), C. Wanjalla (2), S. Nair (3), J. Terry (4), T. Temu (5), B. Shepherd (6), S. Bailin (2), M. Mashayekhi (7), C.L. Gabriel (8), B. Woodward (9), S.A. Mallal (10), J.A. Beckman (11), J. Li (12), J.J. Carr (13), D. Harrison (14), J. Koethe (2)
Institutions
(1) Vanderbilt University Medical Center, Internal Medicine, Nashville, United States, (2) Vanderbilt University Medical Center, Department of Infectious Diseases, Nashville, United States, (3) Vanderbilt University Medical Center, Naschville, United States, (4) Vanderbilt University Medical Center, Nashville, United States, (5) University of Washington, Department of Global Health, Seattle, United States, (6) Vanderbilt University Medical Center, Department of Biostatistics, Nashville, United States, (7) Vanderbilt University Medical Center, Department of Endocrinology, Nashville, United States, (8) Vanderbilt University Medical Center, Department of Gastroenterology, Nashville, United States, (9) Vanderbilt University Medical Center, Tennessee Center for AIDS Research, Nashville, United States, (10) Vanderbilt University Medical Center, Department of Infection Diseases, Nashville, United States, (11) Vanderbilt University Medical Center, Department fo Cardiology, Nashville, United States, (12) Brigham and Women's Hospital, Center for AIDS Research Clinical Core, Boston, United States, (13) Vanderbilt University Medical Center, Department of Radiology, Nashville, United States, (14) Vanderbilt University Medical Center, Division of Clinical Pharmacology, Nashville, United States
program abstract
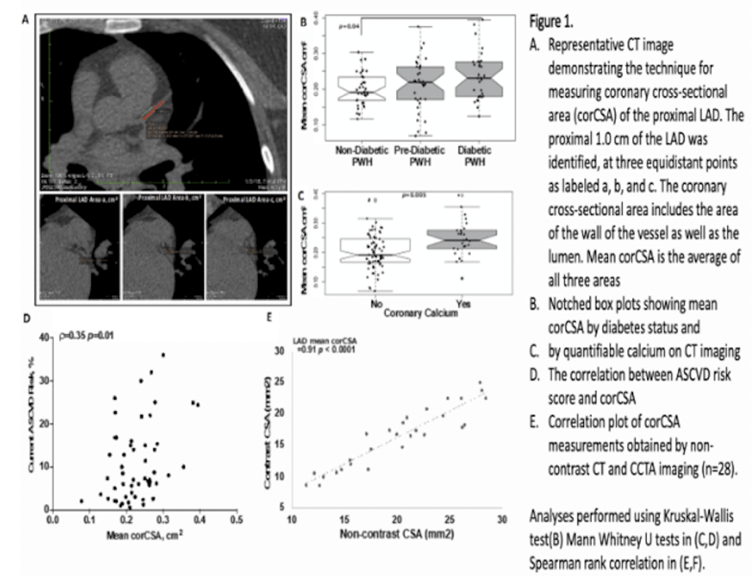
BACKGROUND: Persons with HIV (PWH) have a higher prevalence of non-calcified coronary plaque compared to their HIV-negative counterparts. Although traditional risk factors are important, they do not fully explain the increased risk and presenting phenotype. We assessed coronary cross-sectional area (corCSA) from non-contrast computed tomography (CT) imaging, a measure of arterial remodeling that may have clinical significance among PWH on long-term antiretroviral therapy (ART).
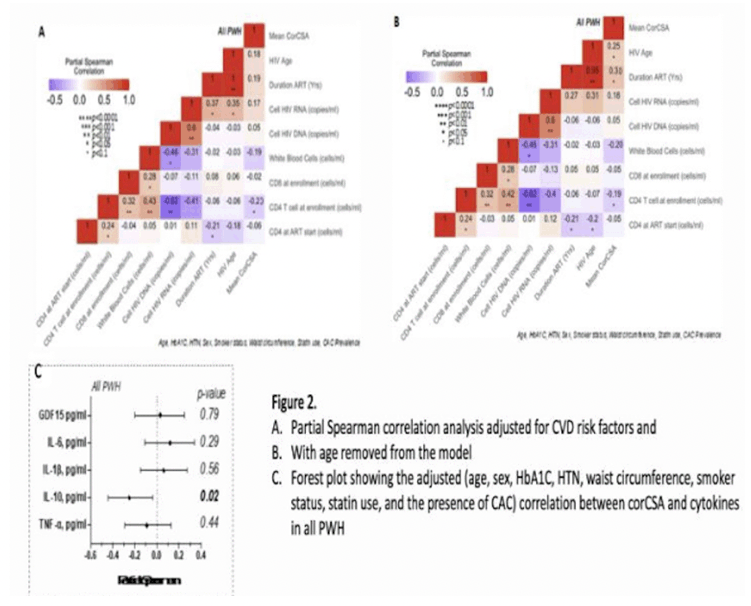
METHODS: We assessed 105 PWH with sustained virologic suppression and a spectrum of cardiometabolic health (37 non-diabetics, 36 pre-diabetics, and 32 diabetics). Participants underwent electrocardiogram gated non-contrast CT scans of the chest to measure the mean corCSA of the proximal left anterior descending artery. Partial Spearman rank correlation adjusted for CVD risk factors (age, sex, smoker status, hypertension, hemoglobin A1C, body mass index, and statin use) was used to assess relationships of corCSA with anthropometric measurements, HIV-related factors, and plasma cytokines (IL-10, IL-6 and IL-1I²).
RESULTS: Mean corCSA was similar between the three metabolic groups but higher in individuals with detectable coronary calcium on CT imaging (p=0.08). In adjusted models we observed an inverse correlation between corCSA and CD4 T-cell count (Ï=-0.22, p<0.05) and plasma IL-10 (Ï=-0.25, p=0.03) but not IL-6 (Ï=0.13, p=0.26) or IL-1I² (Ï=0.07, p=0.58). We also observed a positive correlation between corCSA with duration of ART (Ï=0.33, p<0.001), but this was only significant if not adjusted for age.
CONCLUSIONS: A larger external coronary artery diameter on non-contrast CT imaging, a combined measure of coronary lumen diameter and wall thickness, was observed in PWH with lower circulating CD4 T cell counts and anti-inflammatory IL-10. Future studies will determine the contribution of atherosclerosis and arterial remodeling to corCSA in PWH.


|
| |
|
 |
 |
|
|