 |
 |
 |
| |
HIV/HBV coinfection in pregnancy and response to antiretroviral therapy
|
| |
| |
AIDS 2022 July 29-Aug 2 Montreal
Presenter
Debika Bhattacharya
Authors
D. Bhattacharya * (1), C. Tierney (2), A. Chang (2), D. Moodley (3), V. Govender (3), T. Vhembo (4), N. Mohtashemi (1), H. Ship (5), D. Dula (6), K. George (7), N. Chakhtoura (8), F.K. Matovu (9), M.G. Fowler (10), M. Peters (11), J.S. Currier (1)
Institutions
(1) University of California, Los Angeles David Geffen School of Medicine, Los Angeles, United States, (2) Harvard TH Chan School of Public Health, Boston, United States, (3) University of KwaZulu-Natal, Durban, South Africa, (4) Uz-UCSF Collaborative Research Programme, Harare, Zimbabwe, (5) University of Miami Miller School of Medicine, Miami, United States, (6) Johns Hopkins Research Project, Blantyre, Malawi, (7) FHI 360, Durham, United States, (8) National Institutes of Health, Bethesda, United States, (9) Makerere University'Johns Hopkins University Research Collaboration, Kampala, Uganda, (10) The Johns Hopkins University, Baltimore, United States, (11) University of California San Francisco, San Francisco, United States
program abstract
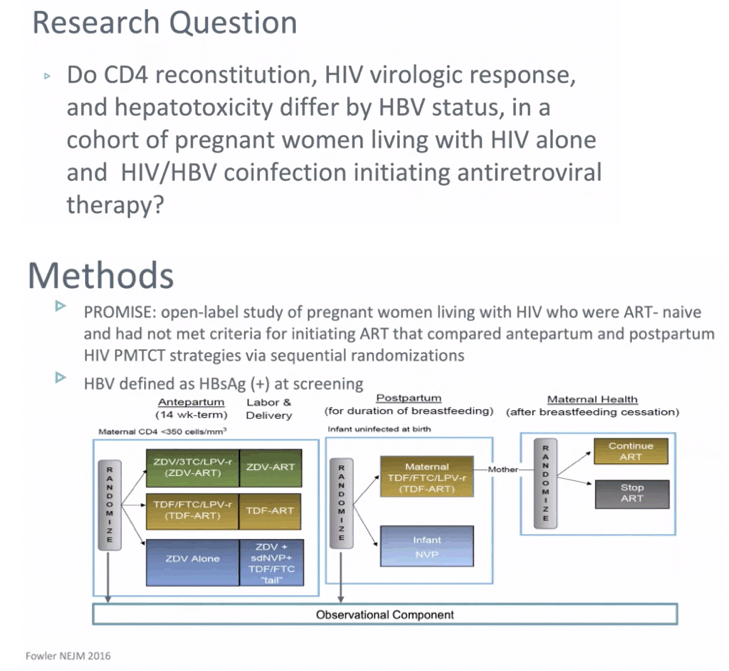
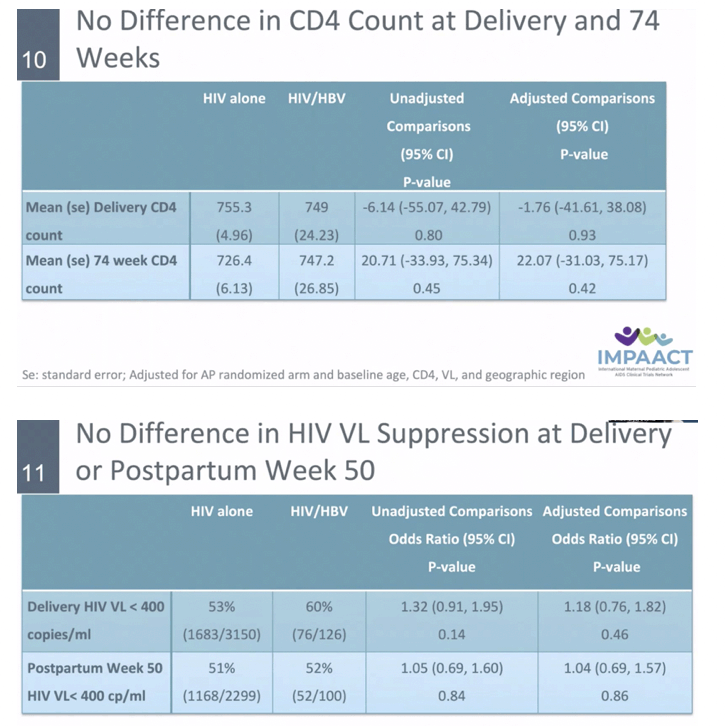
BACKGROUND: Hepatitis B virus (HBV) affects 3-12% of pregnant women with HIV in Africa; however, the impact of HBV infection on HIV outcomes in pregnant women is unclear. We evaluated the association of HIV/HBV coinfection with HIV virologic response, CD4 cell count, and hepatotoxicity in pregnant women in secondary analyses of the IMPAACT PROMISE study.
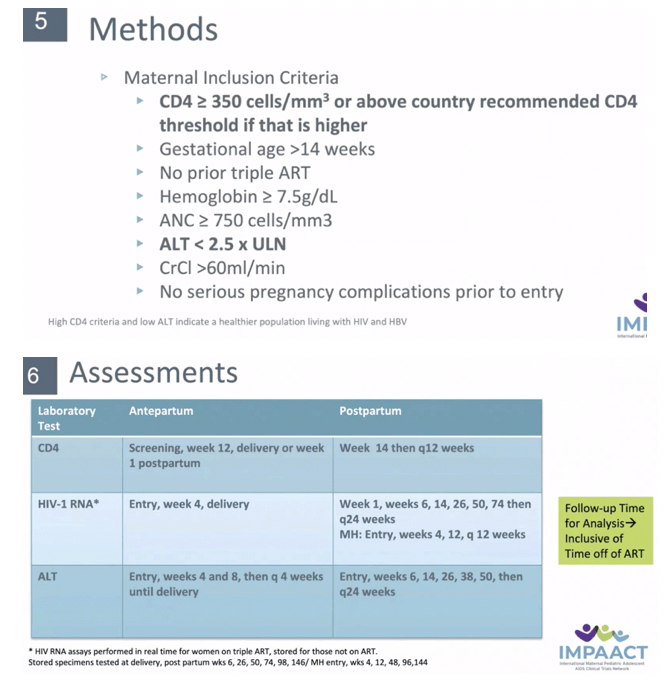
METHODS: The PROMISE study enrolled pregnant women living with HIV who were ART-naive and had not met criteria for initiating ART. Women at '¥14 weeks gestation were randomized to either: Arm A: ZDV alone, Arm B: 3TC+ZDV+LPV/r, or Arm C: FTC+TDF+LPV/r. HBV was defined as HBsAg(+). We compared women with HIV alone to HIV/HBV with outcomes of HIV viral load, CD4, and ALT elevation at delivery and through 74 weeks postpartum using Fisher's exact, t-tests. Time to ALT used the log-rank test. Analyses also compared women with HIV alone to HIV/HBV who were also HBeAg(+).
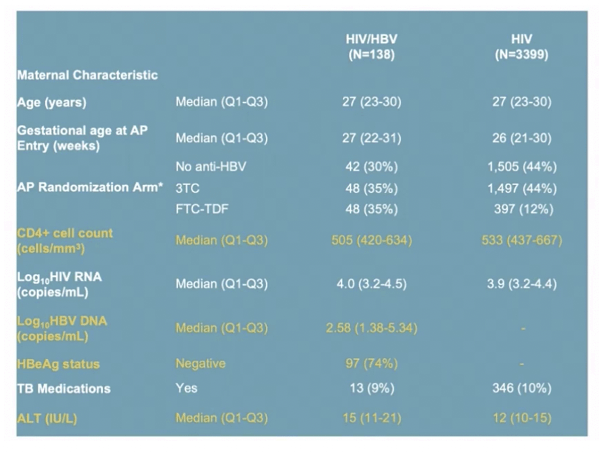
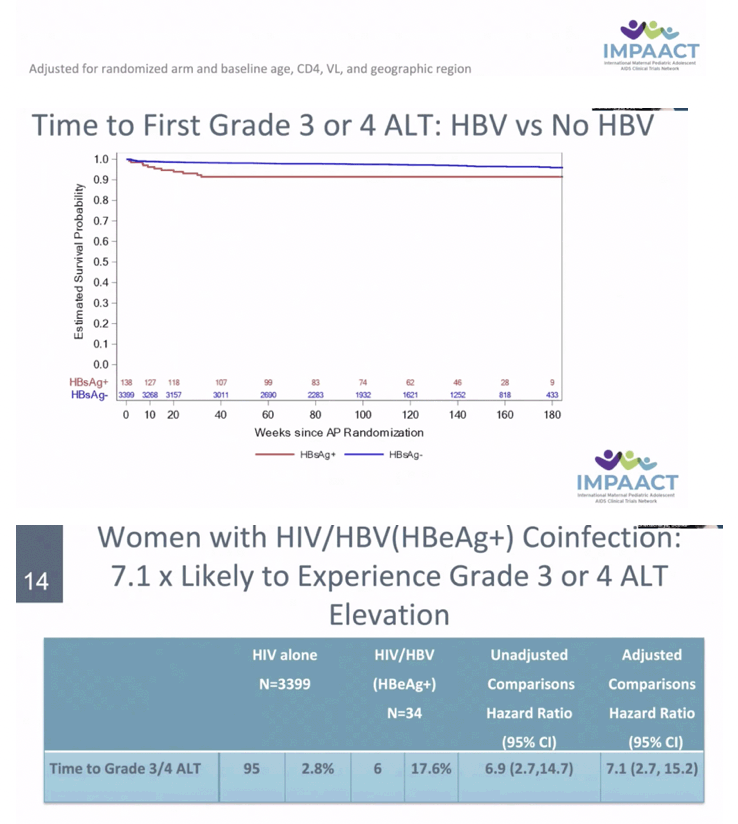
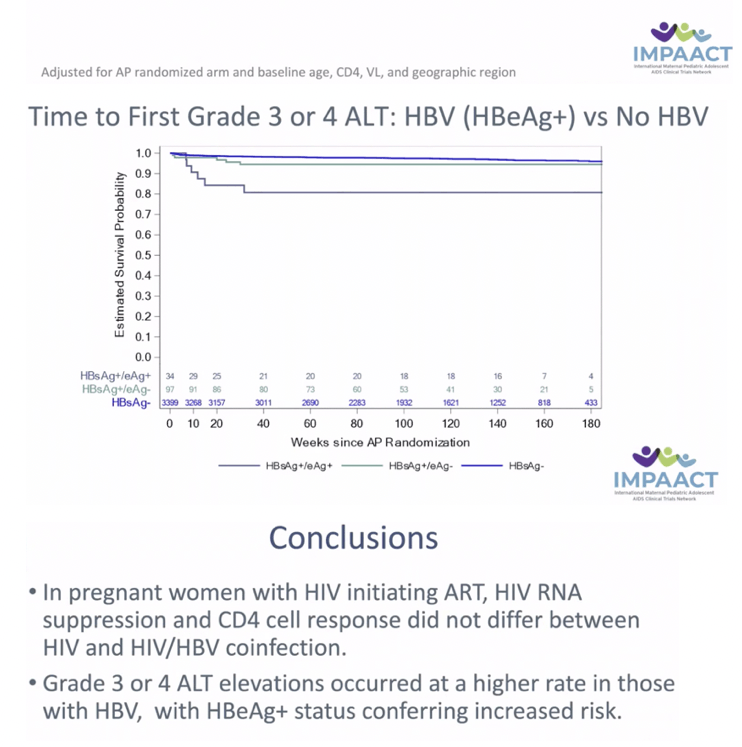
RESULTS: Among 3537 women analyzed, 138 had HBV. Median age, CD4, and HIV VL were 27 years, 505 cells/mm3 and 4.0 Log10 copies/mL, respectively. Thirty-four women (26%) with HBV were HBeAg(+). Median ALT at baseline was 15 and 12 IU/ml amongst HBV-infected and uninfected. There was no statistically significant difference by HBV/HIV co-infection status in HIV VL suppression or CD4 at delivery, at one year postpartum (PP) (primary) or 74 weeks PP. Those with HIV/HBV had more frequent grade 3/4 ALT events (8% vs 3%) overall. Numerically more grade 3/4 ALT events occurred in HIV/HBV coinfected women in Arms B and C (10.4%, 10.4%) vs Arm A (2.4%) but this did not reach statistical significance. Women with HIV/HBV also had earlier first grade 3/4 ALT elevation (hazard ratio (HR) 3.08, 95% CI: 1.56, 5.50). Compared to those with HIV alone, HBV infected women with HBeAg(+) had earlier Grade 3/4 ALT elevation (HR 6.93, 95% CI: 2.70, 14.51).
CONCLUSIONS: In pregnant women with HIV initiating ART, HIV RNA suppression and CD4 cell response did not differ between HIV and HIV/HBV coinfection. However, grade 3 or 4 ALT elevations occurred at a higher rate in those with HBV, with HBeAg+ status conferring increased risk.





|
| |
|
 |
 |
|
|