 |
 |
 |
| |
High prevalence of asymptomatic Omicron carriage and correlation with CD4 T cell count among adults with HIV enrolling in COVPN 3008 Ubuntu clinical trial in sub-Saharan Africa
|
| |
| |
AIDS 2022 Jue 29 - Aug 1 Montreal
Co-auteurs
A. Tapley (1,2), J. Andriesen * (2), L. Fisher (2), Y. Huang (2,3), N. Ketter (2), M. Villaran (2), P. Gilbert (2), J. Hural (2), M. Yacovone (4), L.-G. Bekker (5), L. Corey (2), G. Gray (6), J. Makhema (7), H. Nuwagaba-Biribonwoha (8), T. Samandari (9), P. Elyanu (10), R. Chilengi (11), Z. Chirenje (12), S. Dadabhai (13), N. Mgodi (12), P. Kotze (14), N. Garrett (15), CoVPN 3008 Ubuntu study team (1) University of Washington, Division of Allergy & Infectious Diseases, Department of Medicine, Seattle, United States, (2) Fred Hutchinson Cancer Center, Vaccine and Infectious Disease Division, Seattle, United States, (3) University of Washington, Department of Global Health, Seattle, United States, (4) National Institutes of Health, National Institute of Allergy and Infectious Diseases, Bethesda, United States, (5) Desmond Tutu HIV Centre, University of Cape Town, Cape Town, South Africa, (6) South African Medical Research Council, Pretoria, South Africa, (7) Botswana Harvard AIDS Institute, Gaborone, Botswana, (8) Eswatini Prevent Center, Mbabane, Eswatini, (9) Kisumu CRS/KEMRI-CDC, Kisumu, Kenya, (10) Baylor-Uganda CRS, Kampala, Uganda, (11) Matero CRS, Lusaka, Zambia, (12) University of Zimbabwe Clinical Trials Research Centre, Harare, Zimbabwe, (13) Blantyre CRS, Blantyre, Malawi, (14) Qhakaza Mbokodo Research Clinic, Ladysmith, South Africa, (15) Centre for the AIDS Programme of Research in South Africa, University of KwaZulu'Natal, Durban, South Africa
Abstract
BACKGROUND: The COVID-19 wave driven by the SARS-CoV-2 Omicron variant prompted the need to explore asymptomatic carriage among HIV-immunocompromised adults.
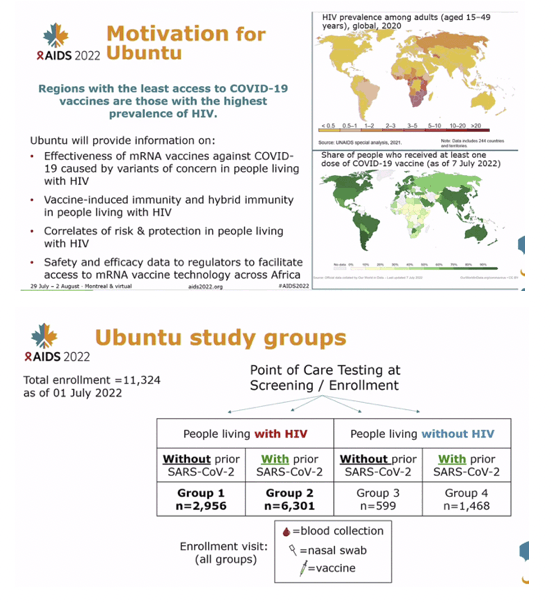
METHODS: In the trial we are assessing COVID-19 mRNA-1273 vaccine efficacy in persons with HIV (PWH) or another COVID-19-associated comorbidity across 7 sub-Saharan African countries. Previously vaccinated persons were excluded. Baseline testing included HIV screening, CD4+ T-cell count and HIV viral load (if HIV+), anti-SARS-CoV-2 antibodies, and nasal swab SARS-CoV-2 reverse-transcriptase polymerase chain reaction (RT-PCR). Participants had to be without COVID-19 signs/symptoms to be vaccinated at enrollment. Here we examine December 2021-April 2022 data to characterize asymptomatic SARS-CoV-2 infections and assessed correlation with CD4 count.
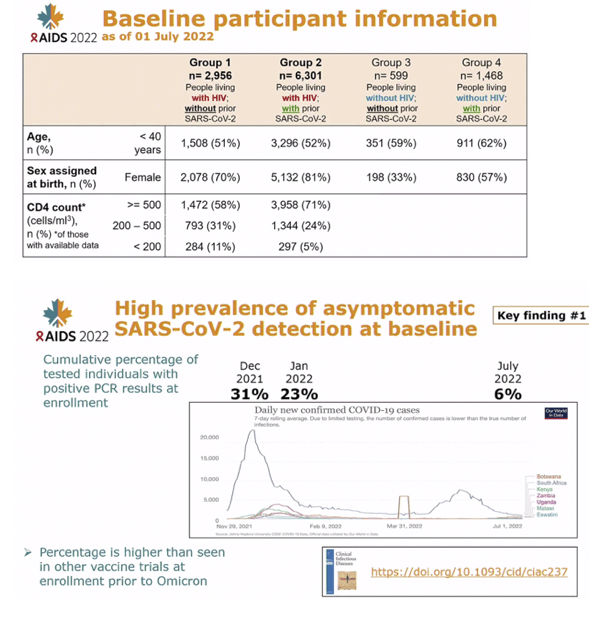
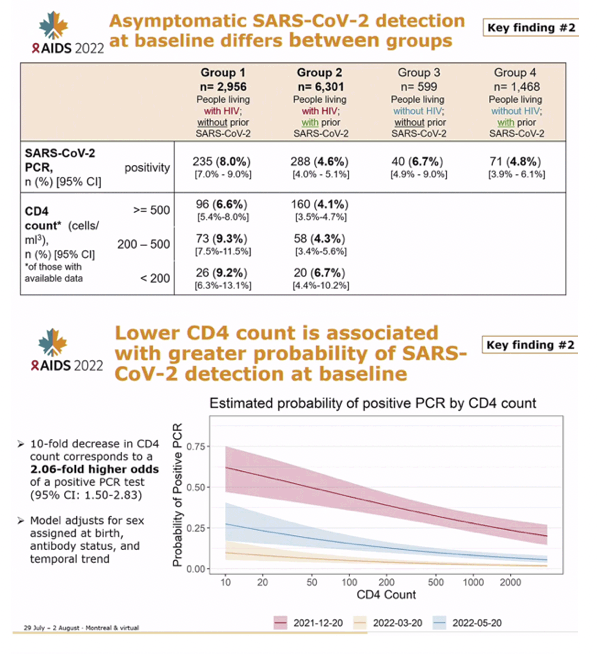
RESULTS: 6397 adults, including 4437 PWH, were enrolled (median age: 38 years; female: 75%). Baseline nasal swab data were available for 5772/6397 (90.2%). 336/5772 (6%) had asymptomatic SARS-CoV-2 infection, more frequent among SARS-CoV-2 seronegative than seropositive participants (9% vs 4%, p<0.001). Infection was detected among 98/1463 (7%) of PWH with a CD4 count<500 cells/mm3 vs 152/2974 (5%) with counts '¥500 cells/mm3 (p=0.037), an association irrespective of SARS-CoV-2 serostatus. A 10-fold CD4 decrease corresponded to 1.72-fold higher odds of PCR positivity, adjusting for serostatus, sex, and non-linear temporal trends (95% confidence interval [CI]:1.09-2.72-fold higher, p=0.019,Figure 1). Over time, the adjusted odds of PCR positivity were highest during the Omicron surge in December 2021 and 44% lower in men than women (95% CI: 17%-62% lower, p=0.004). Gene sequencing on a subset confirmed Omicron.
CONCLUSIONS: Our study of the largest cohort of PWH in a COVID-19 vaccine clinical trial to date reports the asymptomatic SARS-CoV-2 carriage rate was 3-6-fold higher than COVID-19 vaccine trials before Omicron. Additionally, lower CD4 count in PWH strongly correlated with increased odds of SARS-CoV-2 PCR positivity. These data highlight the urgent need for larger studies to better characterize how HIV-associated immunocompromise influences infection acquisition/clearance.



|
| |
|
 |
 |
|
|