 |
 |
 |
| |
A phase IV open-label evaluation of safety and tolerability of coformulated bictegravir/emtricitabine/tenofovir alafenamide for post-exposure prophylaxis following potential exposure to HIV-1
|
| |
| |
AIDS 2022 July 29-Aug 2 Montreal
Download the PDF here
Presenter
Lijun Sun
Authors
A. Liu (1), R. Xin (2), H. Zhang (1), L. Dai (1), R.E. Wu (3), X. Wang (1), A. Li (1), H. Wei (1), J. Li (1), Y. Shao (1), Y. Gao (1), Z. Wang (1), J. Ye (1), G. A bu dou re xi ti (4), Z. Li (1), L. Sun * (1)
Institutions
(1) Beijing Youan Hospital, Capital Medical University, Beijing, China, (2) Beijing Center for Disease Prevention and Control, Beijing, China, (3) Colgate University, Hamilton, United States, (4) The eighth Affiliated Hospital of Xinjiang Medical University, Urumqi, China
program abstract
BACKGROUND: Single-tablet regimen provides a convenient once-daily regimen for the prevention of HIV infection. Here, we investigated the adherence and safety of co-formulated bictegravir/emtricitabine/tenofovir alafenamide (BIC/FTC/TAF) as a 3-drug, single-tablet regimen for post-exposure prophylaxis (PEP) in China.
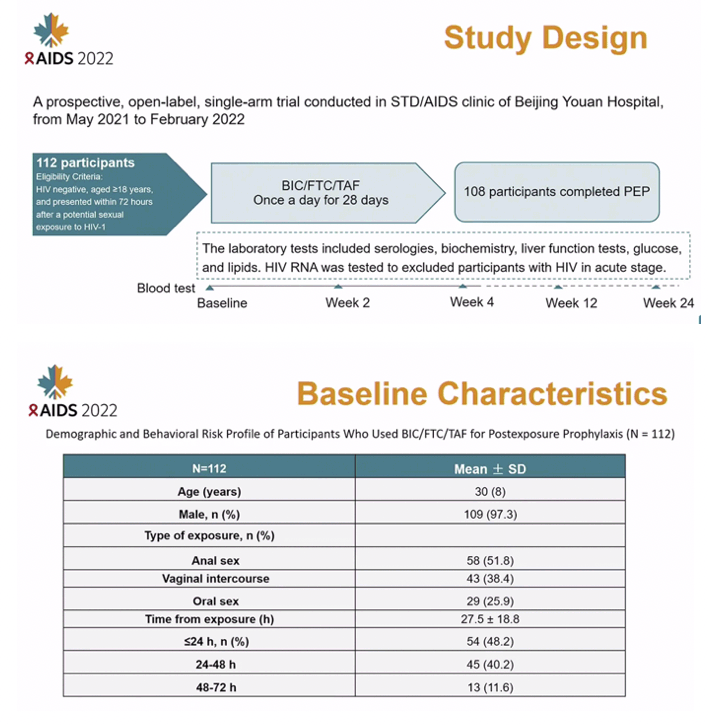
METHODS: This was a prospective, open-label, single-arm trial conducted in an STD/AIDS clinic of a tertiary hospital in Beijing, from May 2021 to February 2022. Adults requiring PEP were prescribed BIC/FTC/TAF one pill once a day for 28 days. Clinical and laboratory data were collected and analyzed at baseline, weeks 2, 4, 8, 12, and 24.
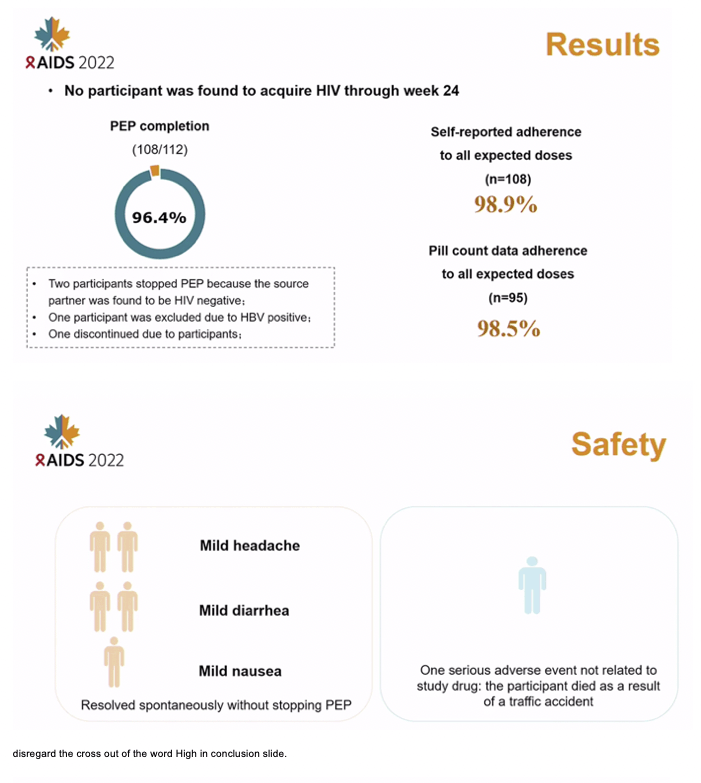
RESULTS: Of 112 participants enrolled in the study, 109 (97.3%) were male and the mean age was 30A±8 years. PEP completion was 96.4% (95% CI: 91.1-99.0). Two participants stopped PEP after 2 days because the source partner was identified as HIV uninfected. One participant was excluded due to hepatitis B virus infection according to the exclusion criteria. One discontinued due to the participant's decision. No participant acquired HIV through week 24. Adherence was 98.9% (standard deviation [SD], 3.3) by self-reporting and 98.5% (SD, 3.5) by pill count. Only 5 participants experienced mild clinical adverse events attributed to study drug (including headache, diarrhea, and nausea) and 4 participants had elevated serum creatinine (grade 1).
CONCLUSIONS: A once daily, single-tablet regimen of BIC/FTC/TAF used as PEP was safe and well-tolerated with a high rate of completion and adherence. BIC/FTC/TAF may be a good option for PEP.



|
| |
|
 |
 |
|
|