 |
 |
 |
| |
INSTIs Plus PIs-But Not INSTIs Alone-Tied to Rising Body Mass Index in Children
|
| |
| |
AIDS 2022, July 29-August 2, Montreal
Mark Mascolini
Use of integrase inhibitors (INSTIs) could not be linked to increasing body mass index (BMI) percentile in a retrospective study of 197 children and teens in Canada [1]. But taking an INSTI with a protease inhibitor (PI) was associated with climbing BMI percentile in this 2015-2019 study.
Several studies tie INSTIs to weight gain in adults with HIV. Although guidelines recommend this potent antiretroviral class as first-line therapy for adults and children, little work has addressed the impact of INSTIs on weight in children.
To address that question, researchers involved in the 7-center Canadian EPIC4 Study conducted this substudy. Children and teens up to 19 years were eligible for the substudy if they had perinatally acquired HIV and received treatment in Canada. EPIC4 investigators used spline regression analysis to figure change in BMI percentile compared with baseline.
The researchers matched 113 INSTI-treated children to 84 no-INSTI controls by age and sex. The study group had a median age of 13 years (range 0.4 to 18.5) when starting an INSTI, 53% were female, 75% had normal body mass index (BMI) at baseline, 20% were overweight or obese, and 5% were underweight. Among the 113 children starting an INSTI, 29% took dolutegravir, 38% raltegravir, and 33% elvitegravir.
At baseline viral load could be detected in 39% of the INSTI group and 11% of the no-INSTI group. After 2 years of therapy 11.5% in the INSTI group and 17.8% in the no-INSTI group had a detectable viral load. Median CD4 count stood at 694 in the INSTI cohort at baseline and at 772 in the no-INSTI control group. After 2 years of treatment median CD4 counts were 772 with INSTIs and 780 with a non-INSTI combination.
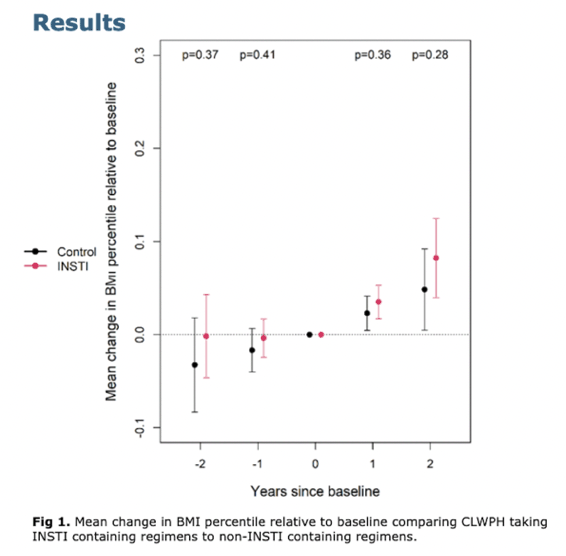
Compared with the no-INSTI control group, average change in BMI percentile relative to baseline did not differ significantly in the INSTI group in the 2 years before baseline (P = 0.37 at year -2 and P = 0.41 at year -1) or in the 2 years after baseline (P = 0.36 at 1 year and P = 0.28 at 2 years). BMI percentile rose gradually after baseline in both groups. Regression analysis showed no between-group difference in BMI percentile (P = 0.276).
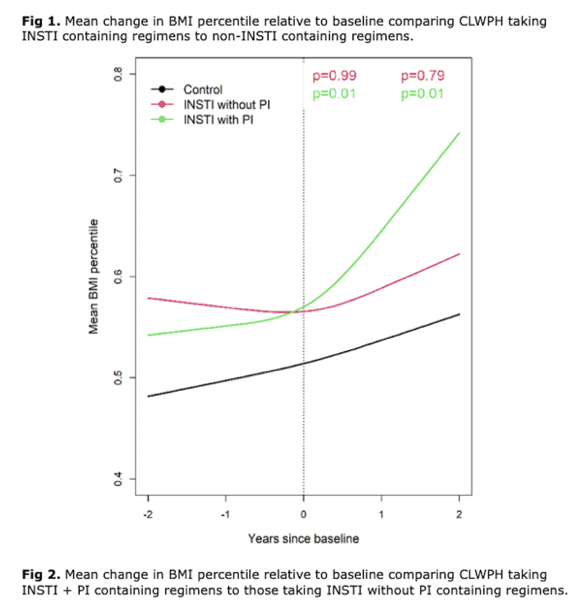
But when the researchers split the INSTI group into 27 children also taking a PI and 86 not taking a PI, they charted a steep rise in BMI percentile change from baseline in year 1 with an INSTI + PI (P = 0.01) and at year 2 with an INSTI + PI (P = 0.01). But there was no such change in BMI percentile in the INSTI-only group.
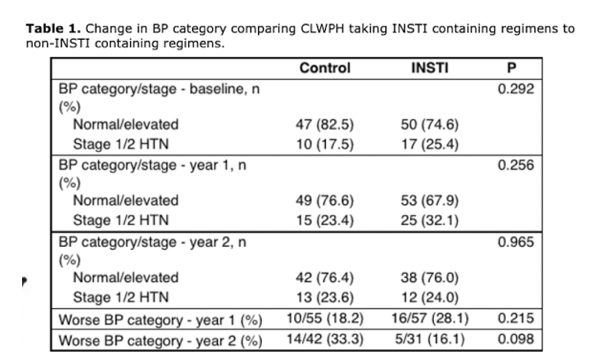
The overall INSTI group did not differ from the no-INSTI control group in normal versus elevated blood pressure, stage 1 or 2 hypertension, or proportion in a worse blood pressure category at treatment year 1 or 2.
The researchers cautioned that their analysis lacks information on medication adherence, sexual maturity of participants, fat distribution, and confounding medications. But with those limitations in mind they concluded that combining a PI with an INSTI is associated with increasing BMI percentile, whereas an INSTI combination not including a PI does not affect BMI percentile over 2 years.
Reference
1. Baba K, Lee T, Kakkar F, et al. Effects of integrase strand transfer inhibitors (INSTIs) on body mass index and blood pressure in children living with HIV. AIDS 2022, July 29-August 2, Montreal. Abstract EPB219.




|
| |
|
 |
 |
|
|