 |
 |
 |
| |
Some "Excess Risk" of Delayed ART in START Trial Persists for Years
|
| |
| |
IDWeek 2022, October 19-23, 2022, Washington, DC
Mark Mascolini
Higher risk of AIDS and serious non-AIDS conditions or death seen in the delayed treatment arm of the START trial ebbs after antiretroviral therapy (ART) begins in the delayed group, according to an updated analysis of this landmark study [1]. "But some excess risk remains," says START's chief statistician, Abdel Babiker of University College London.
The 4684-person 3-year START trial famously found a 57% lower risk of AIDS, a serious non-AIDS disease, or death in people who began ART with a CD4 count above 500 rather than waiting for an AIDS condition to arise or for the CD4 count to drift down to 350 (hazard ratio [HR] 0.43, 95% confidence interval [CI] 0.30 to 0.62, P < 0.001). [2]. Babiker and colleagues undertook this new analysis to assess the long-term impact of delayed HIV control before ART begins, today mainly because of late diagnosis or poor antiretroviral access.
When the randomized phase of START ended because of the clearcut advantage for immediate ART, researchers recommended that people in the delayed-ART arm begin therapy, and follow-up continued until December 31, 2021. The key research question in this continuation phase was "can the excess risk resulting from deferral of ART be eliminated once treatment is started and viral load suppressed or does it persist?"
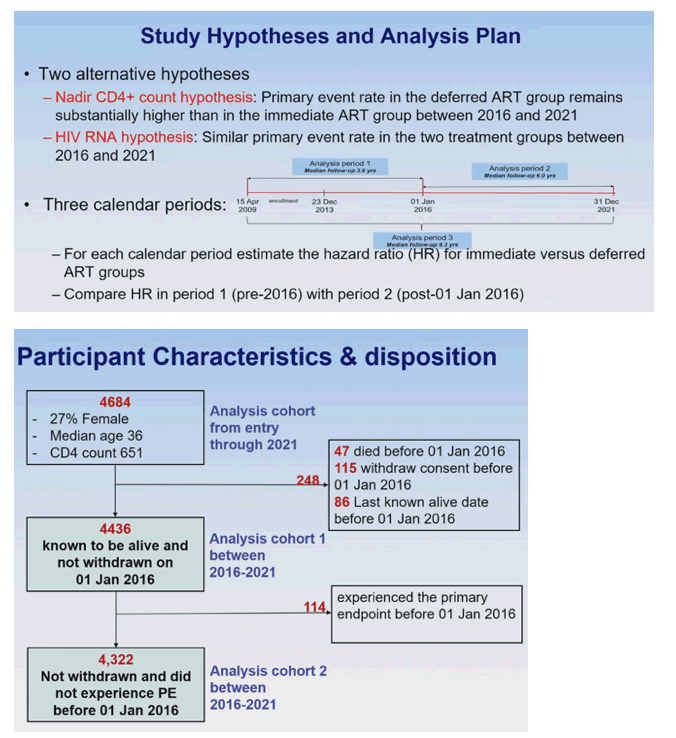
The START group broke study follow-up into three periods: Period 1. April 2009 to January 2016 (median follow-up 3.6 years); Period 2. January 2016 to December 2021 (median follow-up 6.0 years); Period 3. April 2009 to December 2021 (median follow-up 9.3 years). For each period they aimed to estimate the hazard ratio for immediate versus delayed ART, then to compare the hazard ratio for Period 1 to the hazard ratio for period 2.
The investigators considered two hypotheses. The nadir CD4 count hypothesis posited that the "primary event rate in the deferred ART group remains substantially higher than in the immediate ART group between 2016 and 2021." The HIV RNA hypothesis held that there would be a "similar primary event rate in the two treatment groups between 2016 and 2021."
At study entry, the 4684-person cohort had a median age of 36 and a median CD4 count of 651. About one quarter (27%) were women. On January 1, 2016, the study cohort included 4436 people known to be alive and not withdrawn from the study. Before January 1, 2016, 114 of these 4436 people experienced a primary endpoint (AIDS, a serious non-AIDS illness, or death), leaving 4322 people who did not reach a primary endpoint and had not withdrawn before January 1, 2016.
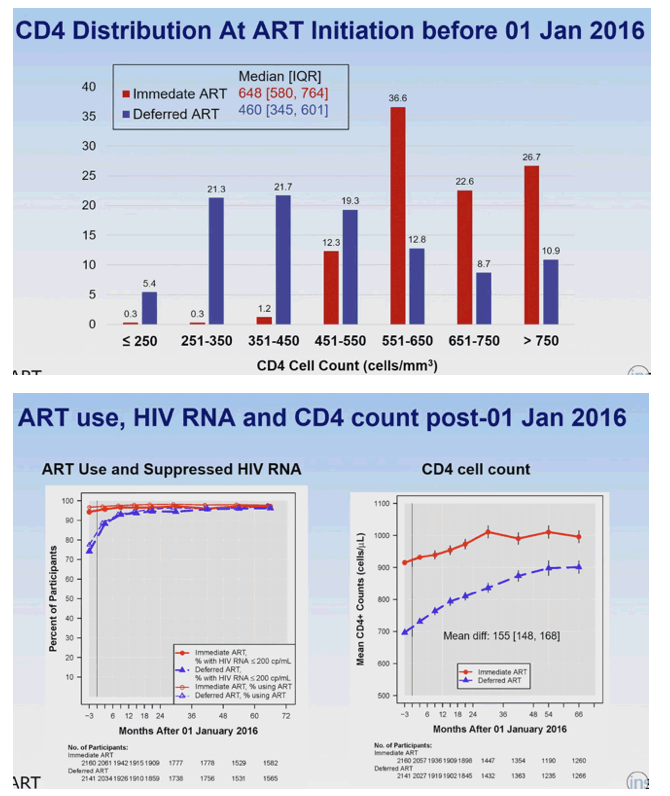
The immediate ART group started ART at a median CD4 count of 648 (interquartile range [IQR] 580 to 764), whereas the deferred group started at a median CD4 count of 460 (IQR 345 to 601). A large majority of the immediate ART group started therapy at a CD4 count of 551 to 650 (36.6%), 651 to 750 (22.6%), or above 750 (26.7%). Respective proportions in the deferred ART who started treatment at those CD4 counts were 12.8%, 8.7%, and 10.9%.
Before 2016 the immediate ART group spent 94.5% of the time taking ART, compared with 35.5% of the time in the deferred ART group. In that period the immediate group's CD4 count averaged 199 cells higher than the deferred group's. After January 1, 2016, the deferred group nearly caught up with the immediate group in time spent taking ART: 99.5% immediate and 95.6% deferred. But the average difference in CD4 count during this period still substantially favored the immediate ART group at 155 cells.
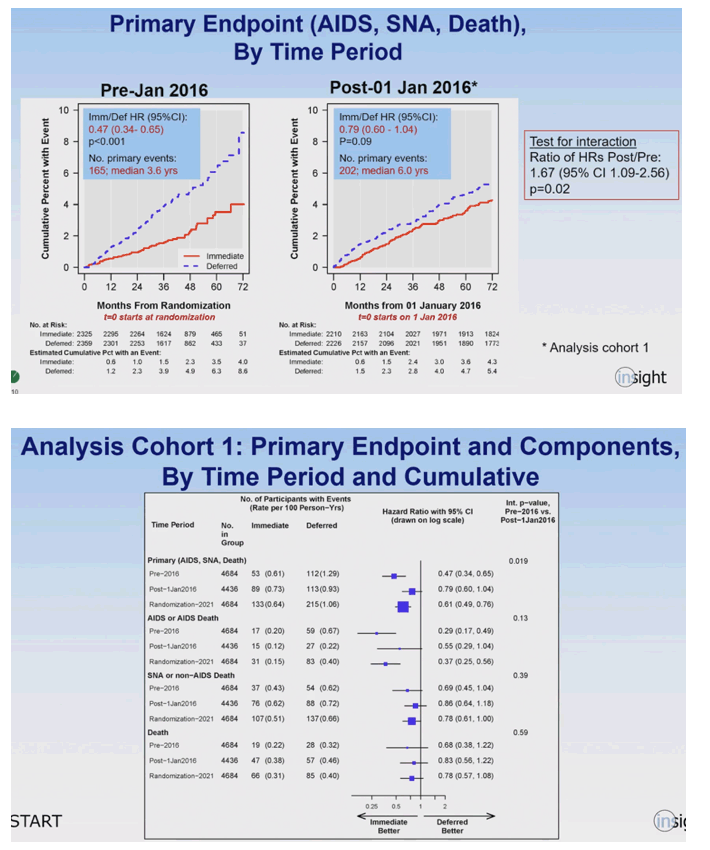
Before January 1, 2016 the immediate ART group had a 53% lower risk of reaching the primary endpoint in the 72 months since randomization (HR 0.47, 95% CI 0.34 to 0.65, P < 0.001). After January 1, 2016 to the end of follow-up 72 months later, the immediate group had a 21% lower risk of reaching the primary endpoint, but that difference from the deferred group had slipped beyond statistical significance (HR 0.79, 95% CI 0.60 to 1.04, P = 0.09). Still, the numerical difference in primary events over this period-89 in the immediate group versus 113 in the deferred group-could be seen as clinically meaningful, certainly to the additional deferred-group people who endured those events.
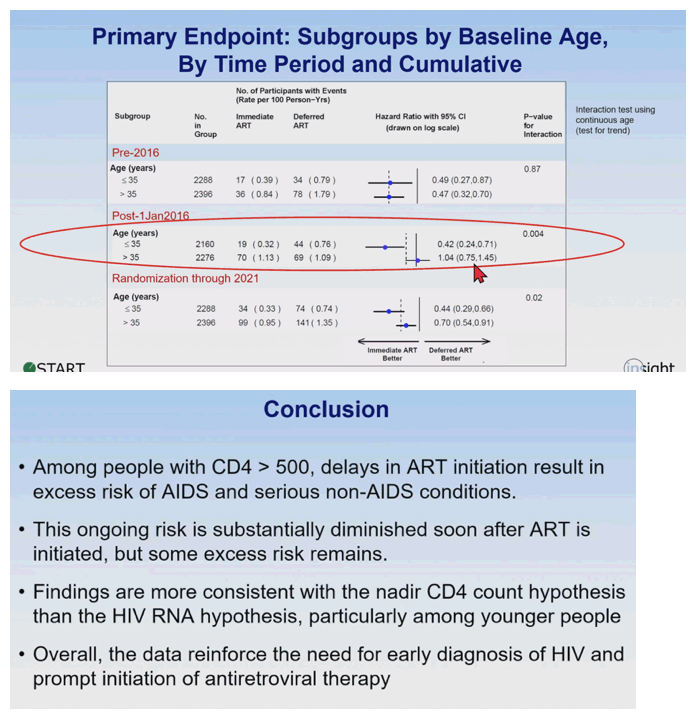
The START team offered the conclusion that their findings "reinforce the need for early diagnosis of HIV and prompt initiation of antiretroviral therapy." Delayed ART in people with a CD4 count above 500, they said, yields an "excess risk of AIDS and serious non-AIDS conditions." Although that excess risk wanes soon after ART started in the deferred treatment group, "some excess risk remains." These findings favor the nadir CD4 count hypothesis explained above. Simple subtraction shows that excess risk amounted to 24 cases of AIDS, other serious conditions, or death: 113 - 89 = 24.
References
1. Babiker AG, Lundgren J, Sharma S, et al. Long term benefits from early antiretroviral therapy initiation in HIV infection: findings from the extended follow-up of the START trial. IDWeek 2022, October 19-23, 2022, Washington, DC. Abstract LB2305.
2. INSIGHT START Study Group, Lundgren JD, Babiker AG, Gordin F, et al. Initiation of Antiretroviral Therapy in Early Asymptomatic HIV Infection. N Engl J Med. 2015;373:795-807. doi: 10.1056/NEJMoa1506816.
https://www.nejm.org/doi/10.1056/NEJMoa1506816?url_ver=Z39.88-2003&rfr_id=ori:rid:crossref.org&rfr_dat=cr_pub%20%200www.ncbi.nlm.nih.gov



|
| |
|
 |
 |
|
|