 |
 |
 |
| |
Phase 3/3b Experience With Long-Acting Cabotegravir and Rilpivirine:
Efficacy and Safety Outcomes Through Week 96 by Race
|
| |
| |
IDWeek 2022 Oct 19-22
Parul Patel1, Emilie R. Elliot2, Ronald D'Amico1, Louise Garside3, Conor Smith4, Jeremy Roberts5,
Sri Byrapuneni6, Joseph W. Polli1, Moti Ramgopal7, Princy Kumar8, Olayemi Osiyemi9,
Bryan Baugh10, Jean van Wyk2
1ViiV Healthcare, Research Triangle Park, NC, United States; 2ViiV Healthcare, Brentford, United Kingdom;
3PHASTAR, Macclesfield, United Kingdom; 4Parexel International, Sheffield, United Kingdom; 5GSK, Mississauga, ON, Canada;
6Parexel International, Research Triangle Park, NC, United States; 7Midway Immunology and Research Center, Fort Pierce, FL, United States; 8Georgetown University Medical Center, Washington, DC, United States; 9Triple O Research Institute PA, West Palm Beach, FL, United States;
10Janssen Pharmaceuticals, Research & Development, Titusville, NJ, United States


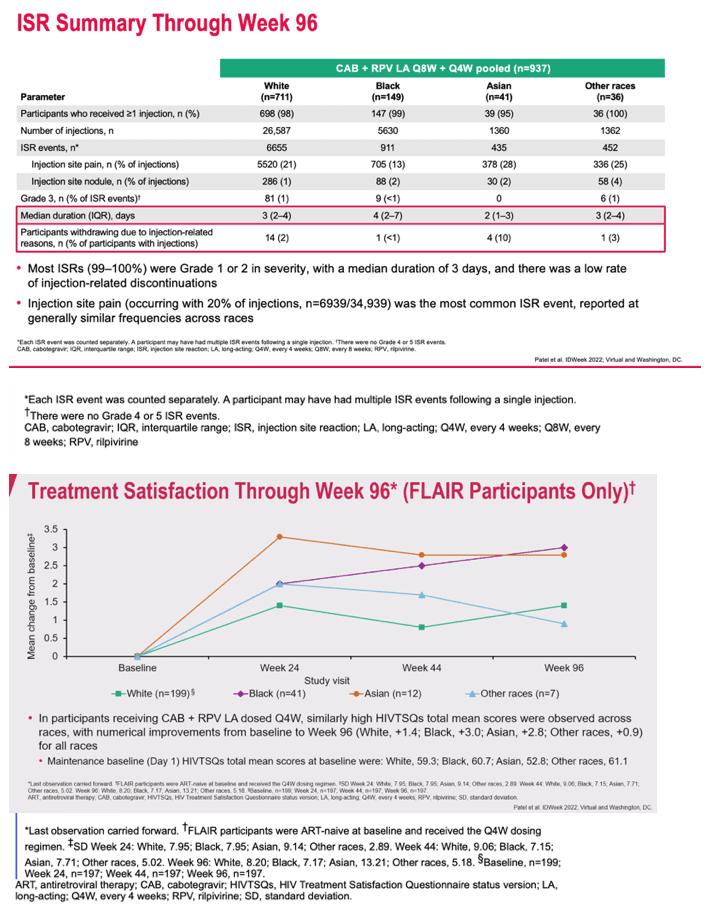
|
| |
|
 |
 |
|
|