 |
 |
 |
| |
CAB + RPV LA in Europe: HIV Control in 87% With Standard or Enhanced Support
|
| |
| |
IDWeek 2022, October 19-23, 2022, Washington, DC
Mark Mascolini
An implementation study of long-acting injected cabotegravir plus rilpivirine (CAB + RPV LA) found that 87% of participants kept their viral load below 50 copies for 12 months regardless of whether they and their providers received standard or enhanced support [1]. In the CARISEL trial, 6% of participants getting this every-2-month regimen stopped taking it for injection-related reasons.
European and US antiretroviral treatment guidelines endorse every-2-month CAB + RPV LA to maintain an undetectable viral load in already virologically suppressed people. The CAB And RPV Implementation Study in European Locations (CARISEL) study, a phase 3b open-label trial, is weighing different strategies to support CAB + RPV LA in 5 European countries: Belgium, France, Germany, the Netherlands, and Spain.
CARISEL randomized 18 clinics to a standard implementation strategy (Arm S) or an enhanced strategy (Arm E). Thirteen of the 18 clinics had no experience with CAB + RPV. Before injections began, staff in Arm E got face-to-face training in injecting the antiretrovirals. Training in Arm S was done virtually. Staff and HIV-positive participants in both arms got CAB + RPV "toolkits." Only Arm E clinics had (1) meetings to discuss implementation tips before the first injection and (2) support to identify problems and plan solutions.
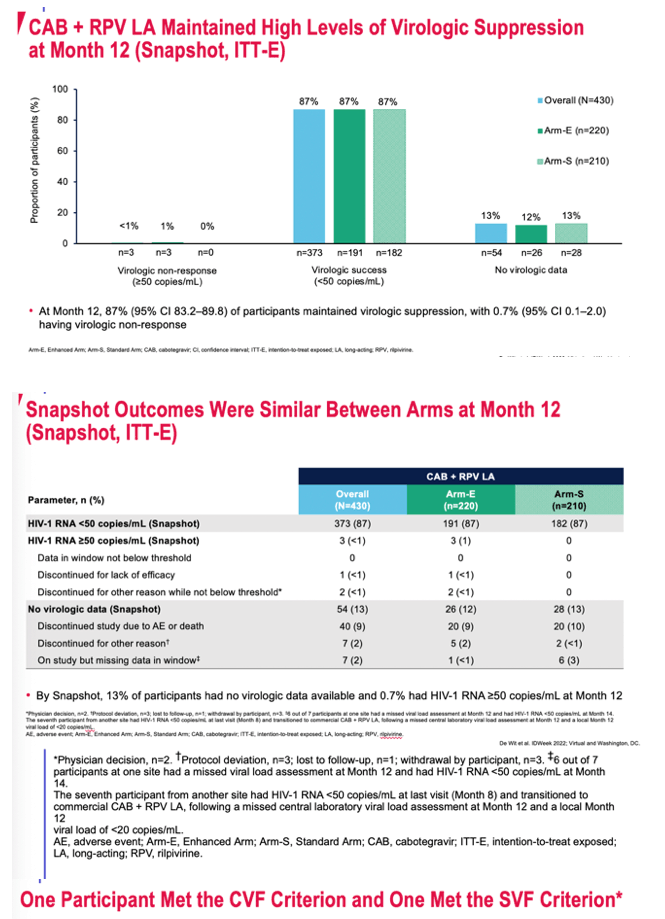
Primary virologic endpoints were (1) proportion of participants with a viral load above and below 50 copies at month 12 by the FDA snapshot algorithm and (2) incidence of confirmed virologic failure (two consecutive viral load at or above 200 copies) or suspected virologic failure (one viral load at or above 200 copies).
Arm E did not differ much from Arm S in median age (44 overall), median body mass index (25 kg/m2), or proportion of females (25%), whites (78%), blacks (18%), or Asians (2%). Arm E had 220 enrollees and Arm S had 210.
After 12 months 87% overall and 87% in each arm had a viral load below 50 copies. There were no virologic failures in arm S and 3 in Arm E (1 confirmed virologic failure and 2 suspected virologic failures). There were no virologic data in 13% (mostly study dropouts due to adverse events or other reasons).
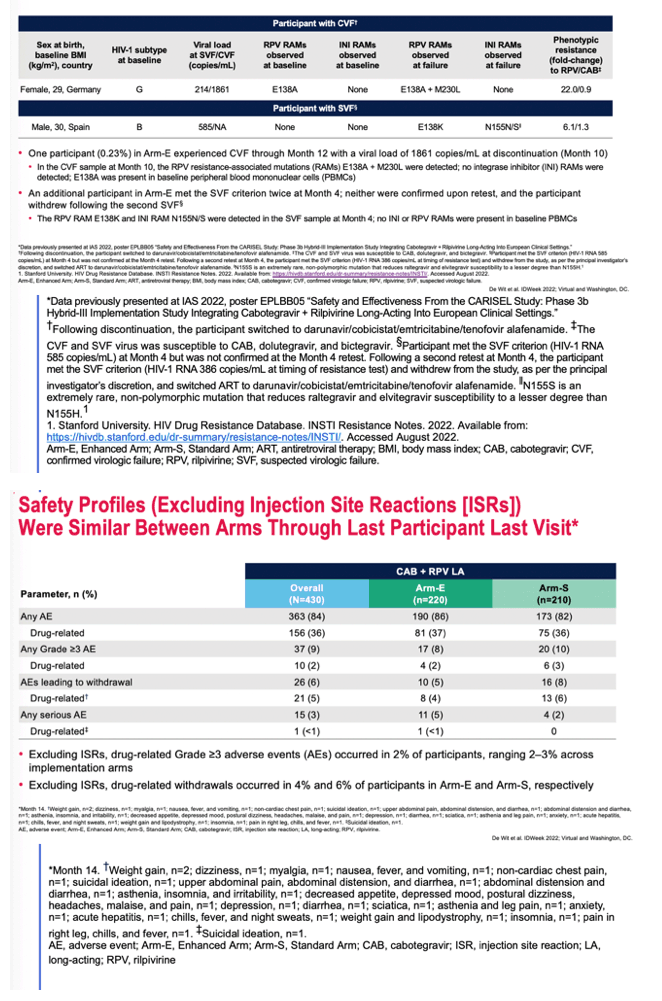
Excluding injection site reactions in results reported in this paragraph, rates of drug-related adverse events were 37% in Arm E and 36% in Arm S; respective proportions of grade 3 or worse drug-related adverse events were 2% and 3%. Drug-related adverse events leading to stopping the drugs affected 8 people (4%) in Arm E and 13 (6%) in Arm S. A drug-related serious adverse event occurred in 1 person in Arm E.
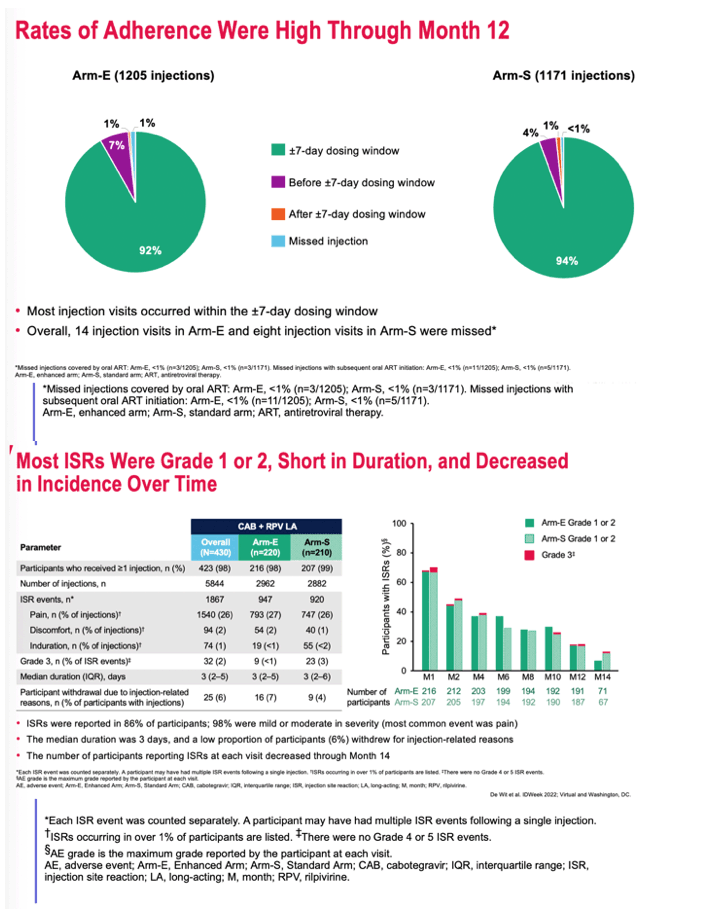
Injection site reactions occurred with 947 of 2962 injections (32%) in Arm E and 920 of 2882 injections (32%) in Arm S. In Arm E there were 9 grade 3 injection site reactions (0.1%) and in Arm S 23 (3%). Injection site reactions lasted a median of 3 days in both study arms. Sixteen people (7%) withdrew from Arm E because of these reactions, and 9 (4%) withdrew from Arm S.
At 12 months, 92% of injections in Arm E took place within a +/- 7-day dosing window. In Arm S that proportion was 94%. In Arm E 14 injections were missed, and in Arm S 8 were missed.
The COVID pandemic did not disrupt CAB + RPV treatment or lead anyone to stop treatment.
Although CARISEL researchers did not state this conclusion in their slides, it appears that enhanced support offered no advantage over standard support in CAB + RPV efficacy or safety.
Reference
1. De Wit S, Rami A, Bonnet F, et al. CARISEL: A hybrid III implementation effectiveness study of implementation of cabotegravir plus rilpivirine long acting (CAB+RPV LA) in EU health care settings. IDWeek 2022, October 19-23, 2022, Washington, DC. Abstract 1584.
|
| |
|
 |
 |
|
|