 |
 |
 |
| |
Topline Results from a 12-Week Phase 2a Trial
(DUET) Evaluating TERN-501, a Highly Selective Thyroid Hormone Receptor (THR) β Agonist, Either as Monotherapy or in Combination with TERN-101, a Nonsteroidal Farnesoid X Receptor (FXR) Agonist, Demonstrated Significant Reductions in MR-Based Liver Fat Content and Fibroinflammation in Patients with Presumed MASH
|
| |
| |
AASLD 2023 nov 10-14
listen to live session
https://www.aasld.org/the-liver-meeting


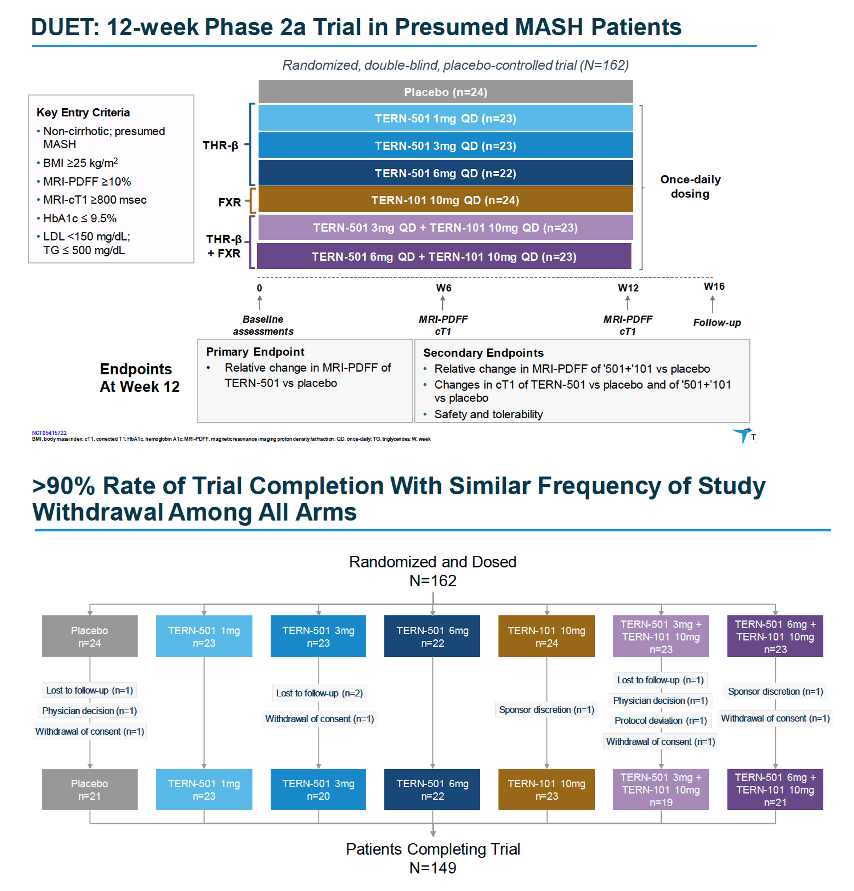
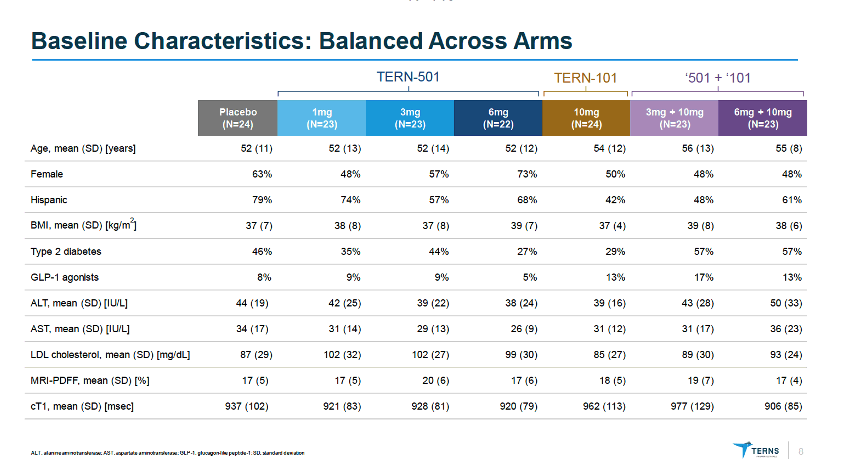
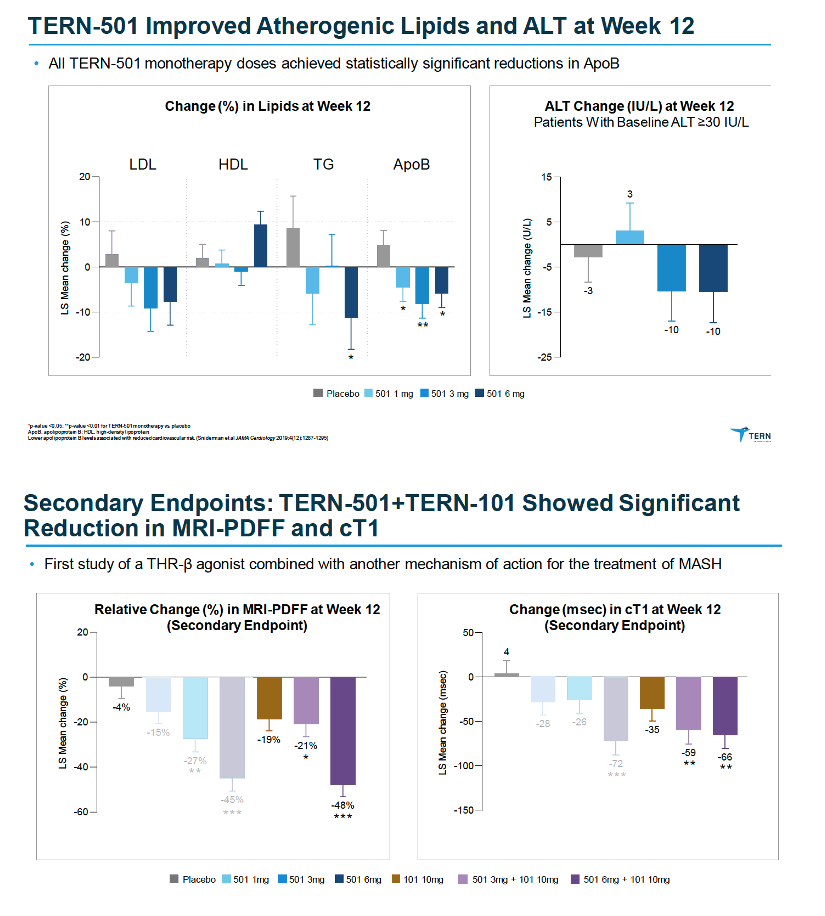
MRI-PDFF is a quantitative imaging biomarker that enables accurate, repeatable and reproducible quantitative assessment of liver fat over the entire liver (9, 11-14). Thus, MRI-PDFF is emerging as one of the leading non-invasive quantitative biomarkers suitable as a surrogate to liver biopsy for assessing treatment response in a subset of NASH trials.
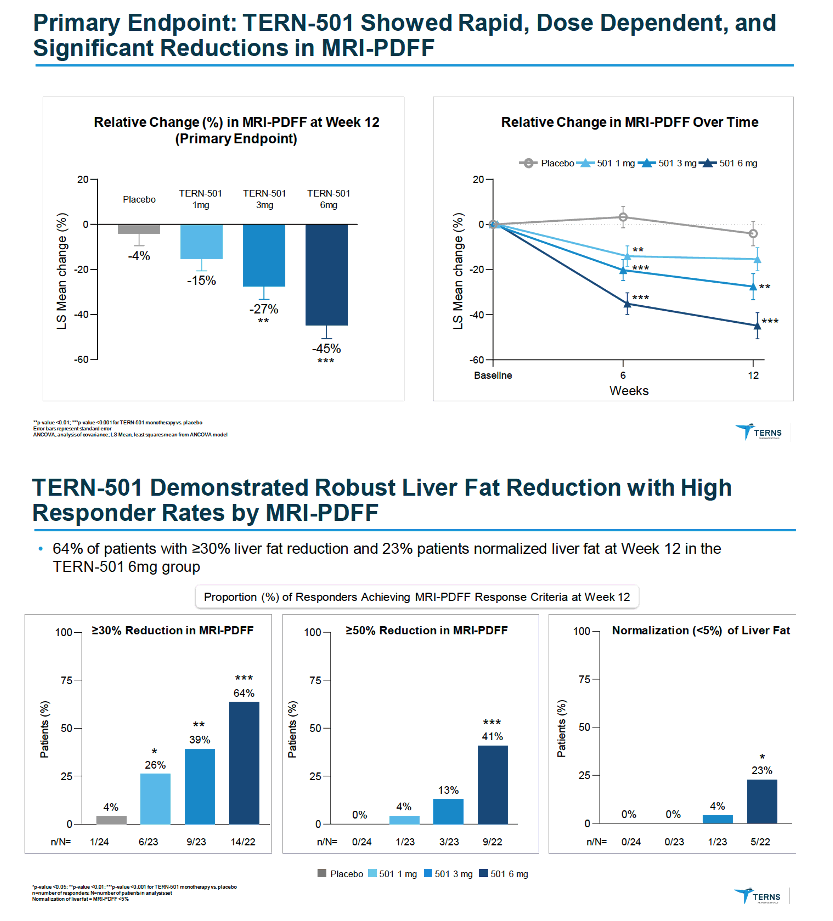
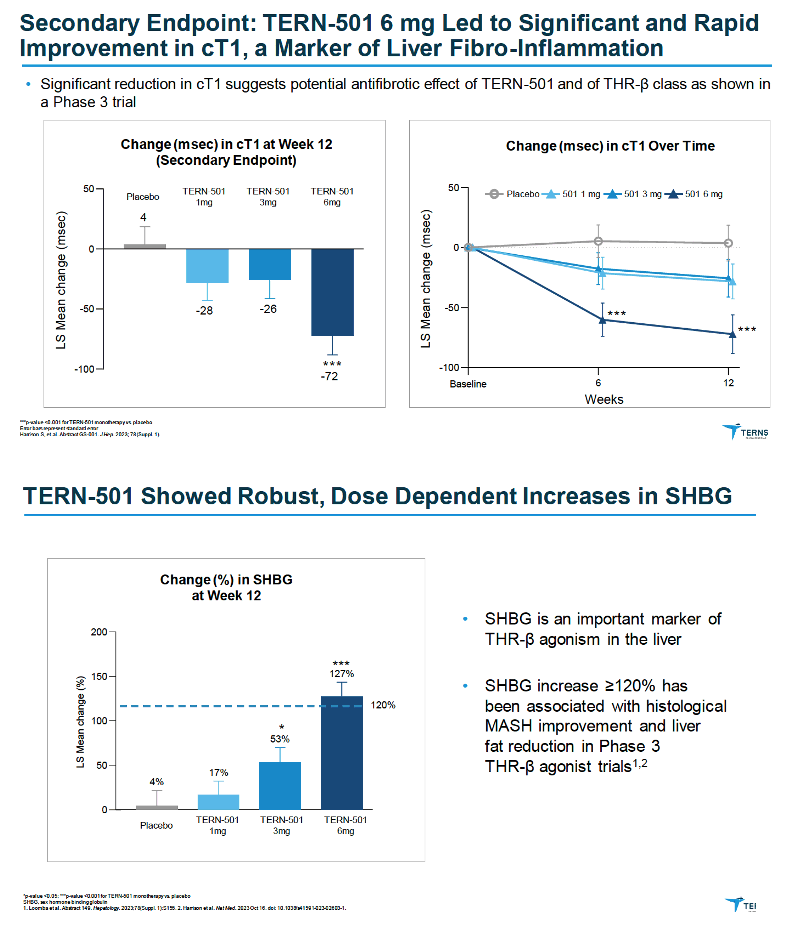
|
| |
|
 |
 |
|
|