 |
 |
 |
| |
Under 25% With HCV and HCC Got DAAs in Nationwide US Insurance Database
|
| |
| |
AASLD 2023, The Liver Meeting, November 10-14, 2023, Boston
https://www.aasld.org/the-liver-meeting
Mark Mascolini
Fewer than 1 in 4 people with HCV-related hepatocellular carcinoma (HCC) got treated with potentially life-saving direct-acting antivirals (DAAs) in a 2015-2021 nationwide US insurance claims analysis by Stanford University's Leslie Kam and colleagues [1]. A significantly higher proportion of DAA-treated than untreated people-all of them with private health insurance-were alive after 5 years. This 3922-person study published online just before AASLD 2023 [2] found that getting care from a gastroenterologist or infectious diseases (GI/ID) clinician tripled chances of taking DAA therapy.
The arrival of DAAs in the clinic late in 2013 transformed HCV infection from a progressive disease that sometimes responded to toxic therapy into a defanged malady that could be cured in 8 weeks. Left unchecked, HCV infection can lead to HCC, among the few cancers with ever-rising incidence and a 5-year survival of only 18%, the Stanford team noted. DAA combinations not only clear more than 90% of HCV infections, they can do so in people with advanced liver disease, including HCC.
Kam and coworkers undertook this study to gauge use of DAAs in people with HCV-related HCC, to nail down predictors Using of that use, and to estimate the survival impact of DAA therapy in this population. ICD-9 and -10 codes, they identified people with HCV-linked HCC in the Clinformatics Data Mart Database, an adjudicated insurance claims warehouse that collects clinical and prescription information on 61 million people in the United States. Participants in this study had to be at least 18 years old, have both an HCV and HCC diagnosis, and have at least 6 months of insurance coverage before their HCC diagnosis. Participants could not have had a liver transplant or coinfection with hepatitis B or D or with HIV.
The researchers gathered baseline demographics, clinical features, and lab values within 6 months of HCC diagnosis. They defined DAA therapy as at least 1 DAA prescription around or after HCC diagnosis. Follow-up ran from the date of HCC diagnosis until death, date of last follow-up, or end of study enrollment, whichever came first. Multivariable Cox proportional hazards regression estimated hazard ratios relating baseline traits to DAA treatment and DAA treatment to survival outcome.
This analysis involved 3922 people averaging 66.1 years in age, of whom 922 (23.5%) got DAA therapy and 3000 (76.5%) did not. Three quarters of these people, 73.5%, were male, 50.7% were white, 16.2% black, 17.3% Hispanic, and 3.7% Asian. Almost half, 46.3%, received care from a GI/ID clinician plus an oncologist, 35.8% got care from a GI/ID clinician but no oncologist, and 7.6% had an oncologist but no GI/ID clinician.
While 53.7% of these people had a history of decompensated cirrhosis (56% with DAA treatment, 53% without), 20.5% got a liver transplant during the study period. Average Charlson Comorbidity Index was virtually identical in the DAA group and the no-DAA group (8.7 and 8.6). The DAA group differed from the no-DAA group in proportion with diabetes (45.2% vs 50.2%, P = 0.01), history of alcohol use (37.2% vs 32.4%, P = 0.01), and history of smoking (44.7% vs 35.9%, P < 0.001), but not history of drug use (18.2% and 16.1%, P = 0.13). Median follow-up stood at 2.3 years in the DAA group and 2.0 years in the no-DAA group (P < 0.001).
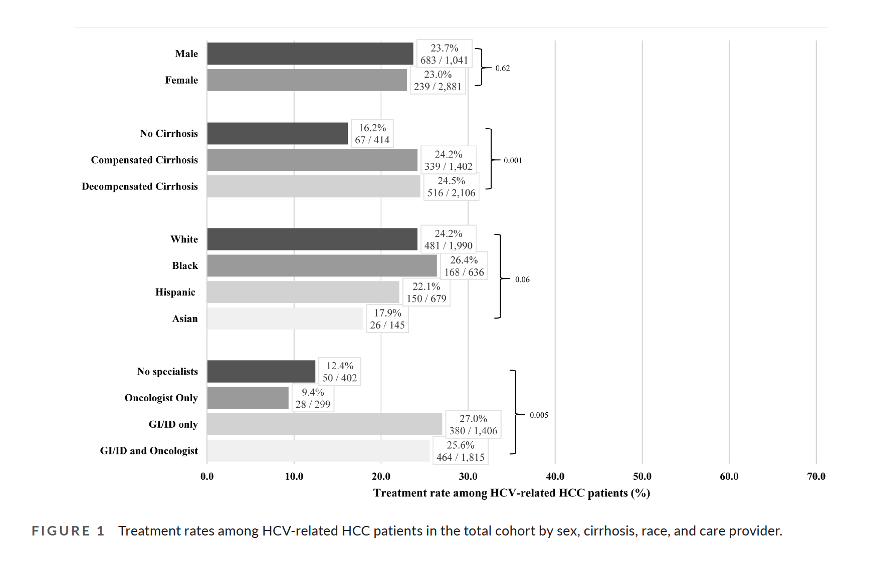
The low DAA use rate of 23.5% did not differ significantly by sex but was lower in Asians (17.9%) and Hispanics (22.1%) than in whites (24.2%) and blacks (26.4%) (P = 0.06). DAA therapy proved more frequent in people with either compensated or decompensated cirrhosis (24.2% to 24.5%) than in people without cirrhosis (16.2%) (P = 0.001). Treatment was more likely in people seeing a GI/ID with or without an oncologist (27.0% and 25.6%) than in people seen only by an oncologist or no specialist (9.4% and 12.4%).
Multivariable Cox models identified several predictors of a lower or higher chance of DAA therapy, at the following hazard ratios (HR) (and 95% confidence intervals):
Lower chance of DAA therapy:
- Younger age: HR 0.98 (0.97 to 0.99), P < 0.001
Higher chance of DAA therapy:
- Seen by a GI/ID vs a primary care provider: HR 3.06 (2.13 to 4.51), P < 0.001
- Seen by a GI/ID and an oncologist vs a primary care provider: HR 2.92 (2.03 to 4.30), P < 0.001
- Having compensated cirrhosis vs no cirrhosis: HR 1.60 (1.18 to 2.21), P = 0.003
- Having decompensated cirrhosis vs no cirrhosis: HR 1.45 (1.07 to 1.98), P = 0.02
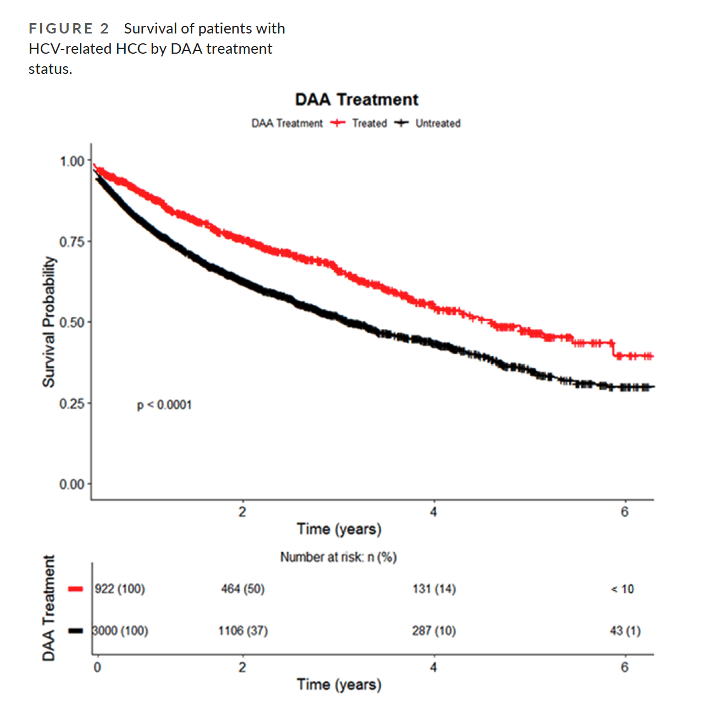
Five-year survival was significantly more likely in people who got DAA therapy (47.2% vs 3.2%, P < 0.001). This survival advantage held true in males and females; whites, blacks, and Hispanics; and people with compensated or decompensated cirrhosis. In people without cirrhosis, 5-year survival was high and similar (about 62%) with and without DAA therapy.
Multivariable analysis adjusted for age, sex, race and ethnicity, comorbidities, presence of cirrhosis, and liver transplant linked DAA therapy significantly and independently to a 39% lower death risk (HR 0.61, 95% CI 0.53 to 0.69, P < 0.001).
Kam and colleagues think their findings underscore persistently low awareness of HCV and DAA therapy in the general public. Both the public and healthcare workers, they said, need "continual education on DAA treatment."
That more than half of the people in this study had decompensated cirrhosis, they observe, "lends further evidence that HCV-related HCC was found late in the course of disease."
References
1. Kam LY, Yeo YH, Ji F, Henry L, Cheun R. Low antiviral treatment rate for patients with hepatitis C (HCV)-related hepatocellular carcinoma (HCC)-a real-world nationwide U.S. study. AASLD 2023, The Liver Meeting, November 10-14, 2023, Boston.
2. Kam LY, Yeo YH, Ji F, Henry L, Cheun R. Treatment rates and factors associated with direct-acting antiviral therapy for insured patients with hepatitis C-related hepatocellular carcinoma - A real-world nationwide study. Alimentary Pharmacol Ther. 2023 Nov 8. DOI: 10.1111/apt.17794. https://doi.org/10.1111/apt.17794 pdf attached above
|
| |
|
 |
 |
|
|