 |
 |
 |
| |
Long-term effect of dolutegravir use on more depressive symptoms among older people with HIV
|
| |
| |
DTG Linked to Depression-But Not in People Under 60, Black, or on Antidepressants
International Workshop on HIV & Aging 2023, October 26-27, Washington, DC
Reported by Mark Mascolini for NATAP and Academic Medical Education
Echoing earlier reports, analysis of the US CHARTER cohort tied dolutegravir (DTG) therapy to more depressive symptoms [1]. But unlike earlier studies, the CHARTER analysis factored in the potential impact of age, race, and antidepressant use and found that DTG did not promote depressive symptoms in people younger than 60, black, or taking an antidepressant.
Four earlier studies of various design and size linked treatment with the integrase inhibitor DTG to depressive symptoms [2-5]. But these studies did not use a standardized depression assessment or try to discern interactions between DTG, antidepressants, and demographics like age or race.
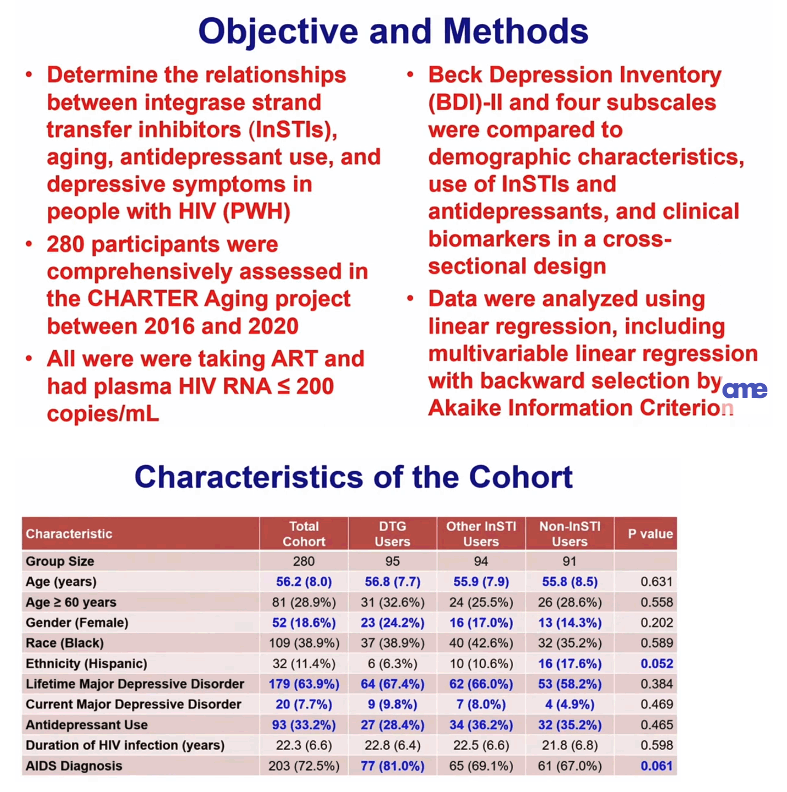
The CHARTER analysis involved 280 participants comprehensively studied in the CHARTER Aging project between 2016 and 2020, all of them taking antiretroviral therapy and with a viral load at or below 200 copies. This cross-sectional study compared results of the Beck Depression Inventory (BDI)-II and four subscales according to demographics, integrase inhibitor use, and antidepressant use in study participants.
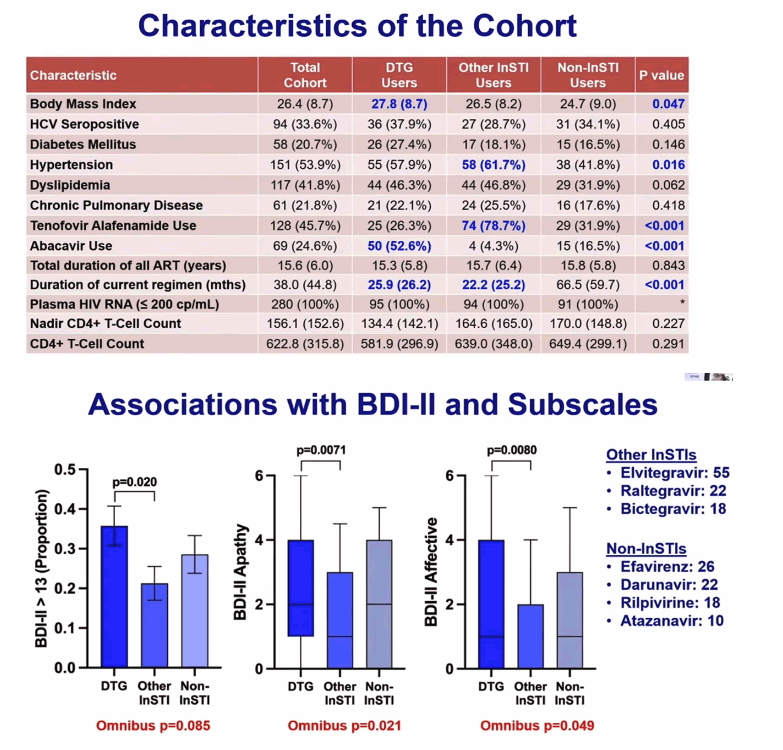
The 280-person cohort included 95 taking DTG, 94 taking some other integrase inhibitor, and 91 not taking an integrase inhibitor. These three groups did not differ significantly by age (overall 56.2), proportion of women (18.6% overall), proportion of blacks (38.9% overall), proportion with lifetime major depressive disorder (63.9% overall), or antidepressant use (33.2% overall). The group not taking an integrase inhibitor had the highest proportion of Hispanics (17.6% vs 11.4% overall, P = 0.052), and people taking DTG tended to have the highest proportion with AIDS (81.0% vs 72.5% overall, P = 0.061). Body mass index was significantly higher in people taking DTG than in the other groups (27.8 kg/m2 vs 26.4 kg/m2 overall, P = 0.047), a higher proportion of people taking DTG also took abacavir (52.6% vs 24.6% overall, P < 0.001), and a higher proportion not taking an integrase inhibitor were taking tenofovir alafenamide (TAF) (78.7% vs 45.7% overall P < 0.001).
Compared with people using other integrase inhibitors (elvitegravir, raltegravir, bictegravir), a significantly higher proportion taking DTG had a BDI-II above 13, that is, out of the normal range (about 35% vs 20%, P = 0.020). DTG users also had significantly higher proportions out of the BDI-II normal range for apathy (P = 0.0071) and affective symptoms (P = 0.0080). Further comparative analyses linked DTG to higher (worse) BDI-II values in people older than 60 (P = 0.021), nonblacks (P = 0.0029), and people not taking an antidepressant (P < 0.001).
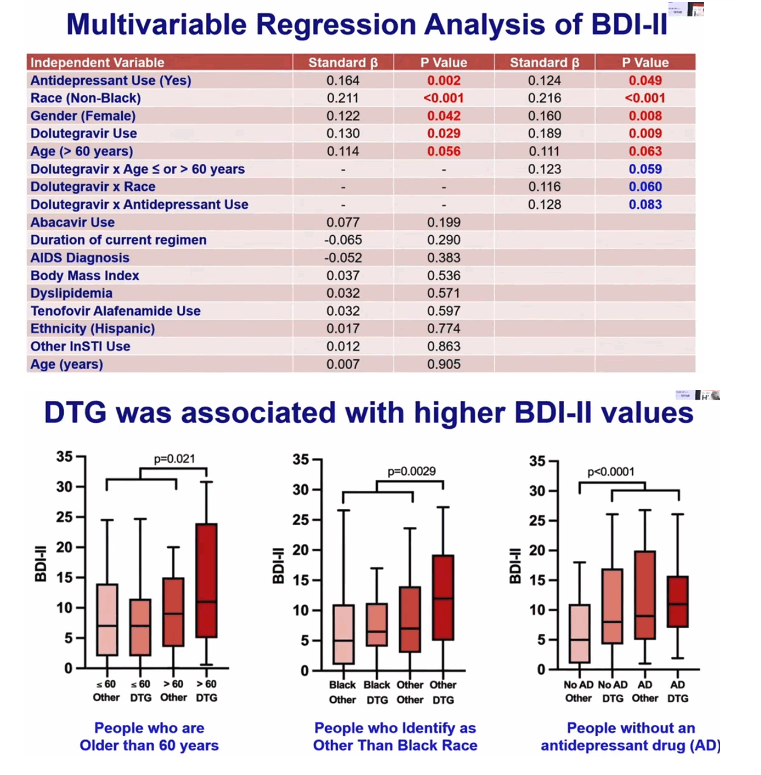
In regression analysis, variables independently associated with higher (worse) BDI-II scores were DTG use (beta 0.130, P = 0.029), antidepressant use (beta 0.164, P = 0.002), age 60 or older (beta 0.114, P = 0.056), nonblack race (beta 0.211, P < 0.001), and female gender (beta 0.122, P = 0.042). Interaction analyses found strong trends toward interactions between DTG and age above or below 60 (beta 0.123, P = 0.059, meaning DTG was associated with worse BDI-II mainly in people older than 60), between DTG and antidepressant use (beta 0.128, P = 0.083, meaning DTG was associated with worse BDI-II mainly in people not using antidepressants), and between DTG and race (beta 0.116, P = 0.060, meaning DTG was associated with worse BDI-II mainly in nonblacks).
CHARTER researchers concluded that DTG “may increase depressive symptoms” in people with HIV who are 60 or older, not black, or not taking an antidepressant. But results indicated no association between DTG and more depressive symptoms in people with HIV who are younger than 60, black, or taking an antidepressant.
Questions were raised about drug levels, timing for taking DTG (early in day or at night) & how these factors might relate to the findings, Dr Letendre said they hsvethis data and he will check.
Link to oral session:
https://www.youtube.com/watch?v=x2tVH2yHKHI
References
1. Roy U, Kamkwalala A, Matthews A, et al. Long-term effect of dolutegravir use on more depressive symptoms among older people with HIV. International Workshop on HIV & Aging 2023, October 26-27, Washington, DC. Abstract 7.
2. de Boer MG, van den Berk GE, van Holten N, et al. Intolerance of dolutegravir-containing combination antiretroviral therapy regimens in real-life clinical practice. AIDS. 2016;30:2831-2834. doi: 10.1097/QAD.0000000000001279. https://journals.lww.com/aidsonline/fulltext/2016/11280/
intolerance_of_dolutegravir_containing_combination.11.aspx
3. Hoffmann C, Welz T, Sabranski M, et al. Higher rates of neuropsychiatric adverse events leading to dolutegravir discontinuation in women and older patients. HIV Med. 2017;18:56-63. doi: 10.1111/hiv.12468. https://onlinelibrary.wiley.com/doi/10.1111/hiv.12468
4. Elzi L, Erb S, Furrer H, Cavassini M, et al. Swiss HIV Cohort Study Group. Adverse events of raltegravir and dolutegravir. AIDS. 2017;31:1853-1858. doi: 0.1097/QAD.0000000000001590.
https://journals.lww.com/aidsonline/fulltext/2017/08240/
adverse_events_of_raltegravir_and_dolutegravir.10.aspx
5. Fettiplace A, Stainsby C, Winston A, et al. Psychiatric symptoms in patients receiving dolutegravir. J Acquir Immune Defic Syndr. 2017;74:423-431. doi: 10.1097/QAI.0000000000001269. https://journals.lww.com/jaids/fulltext/2017/04010/psychiatric_symptoms_in_patients_receiving.11.aspx


|
| |
|
 |
 |
|
|