 |
 |
 |
| |
2 Oral Talks - PLWH: Sarcopenia, Mitochondrial Function
|
| |
| |
https://www.youtube.com/watch?v=Mh0HT047hqA

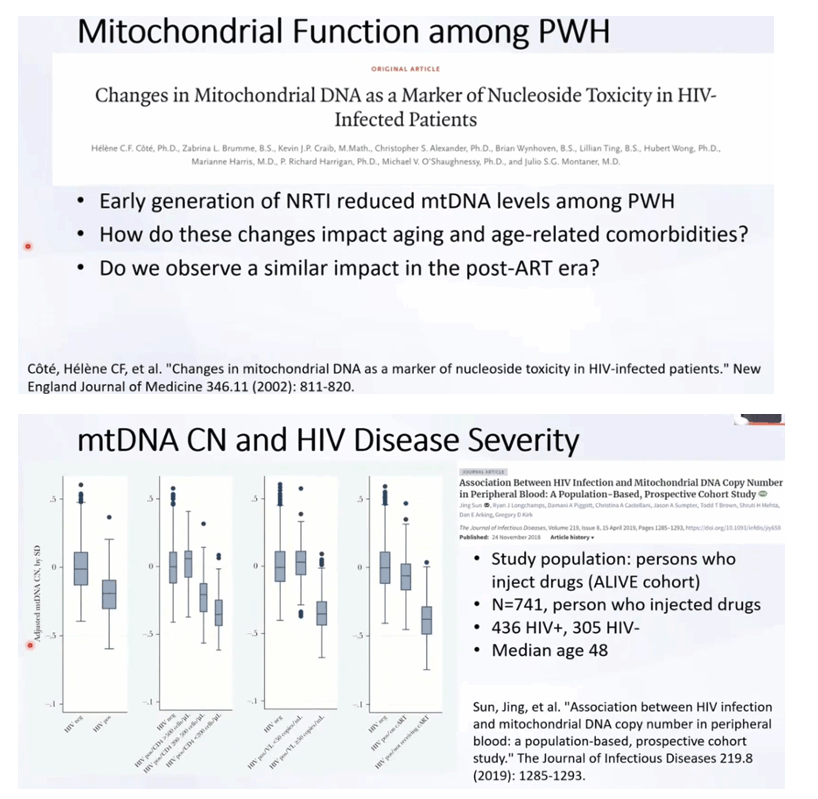
Abstract
Background
Low mitochondrial DNA (mtDNA) copy number (CN) is a predictor of adverse aging outcomes, and its status may be altered in human immunodeficiency virus (HIV)-infected persons. This study evaluated the cross-sectional and longitudinal change of mtDNA CN by HIV markers.
Methods
mtDNA CN was measured in the ALIVE (AIDS Linked to the Intravenous Experience) cohort of persons with a history of injecting drugs. Multivariable linear regression models controlling for demographic characteristics, behavior, and hepatitis C virus (HCV) seropositivity assessed the relationship of mtDNA CN to HIV markers (CD4+ T-cell counts, viral load, antiretroviral therapy [ART] use). Linear mixed models tested the association between HIV markers and age-related mtDNA CN trajectories.
Results
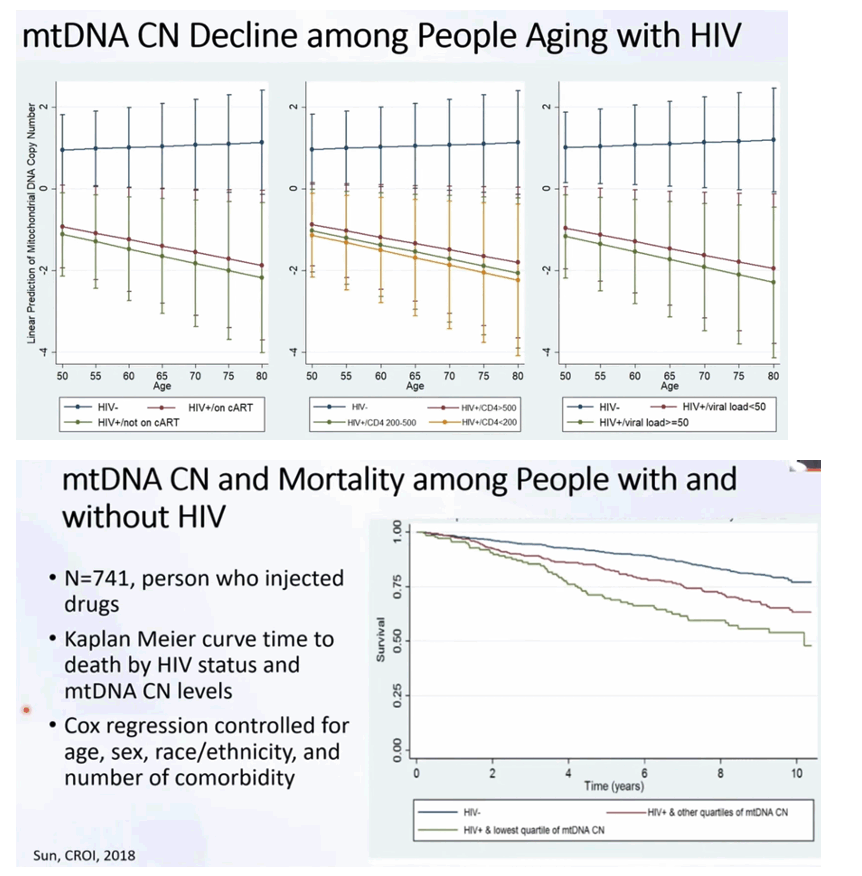
Among 741 individuals at baseline, 436 (59%) were infected with HIV. HIV-infected individuals who had lower CD4+ T-cell counts (P = .01), had higher viral loads (P < .01), and were not receiving ART (P < .01) had significantly lower mtDNA CNs than uninfected persons; there was no difference between participants who were uninfected and HIV-infected individuals who had well-controlled HIV levels. In longitudinal follow-up of 507 participants, from age 50 years onward, mtDNA CN declined significantly faster among HIV-infected individuals than among HIV-uninfected persons (-0.03 units of change/year vs 0.006 units of change/year; P = .04), even among infected individuals with well-controlled HIV.
Conclusion
Before 50 years of age, mtDNA CN is similar between HIV-infected individuals with well-controlled HIV and uninfected persons, but from age 50 onward, mtDNA CN declines significantly faster among all infected individuals than among HIV-uninfected persons.
In the current study, we found that PLWH who had well-controlled HIV and were receiving cART had similar mtDNA CNs in their blood at baseline, compared with PWOH, while those with poorly controlled HIV showed a significant decrease in mtDNA CNs from baseline. Although there was no baseline difference in mtDNA CN between PLWH who had well-controlled HIV and PWOH, we observed a significant age-related decline in mtDNA CN among all PLWH (including those with well-controlled HIV) after the age of 50 years, while we observed no decline among PWOH after the age of 50 years. Additionally, the rate of decline in mtDNA CN among PLWH demonstrates a dose response to major HIV biomarkers (CD4+ T-cell count and HIV load) in which the rate of mtDNA CN decline among people with poorly controlled HIV is steeper than that among those with well-controlled HIV. Our results agree with previous studies that have demonstrated lower levels of mtDNA content in PLWH [8, 23-25] as compared to PWOH. They are also consistent with Miura et al’s observation [27] that mtDNA CNs correlate with CD4+ T-cell counts and inversely correlate with viral load. Of even greater significance, our study demonstrates the different trajectories of mtDNA CN among PLWH with different disease severities.
Our observations might suggest that, among PLWH receiving cART, the difference in the mtDNA CN caused by the residual effects of thymidine analogs might be attenuated, while chronic infection and HIV disease severity are the primary contributors that lead to the continued decline of mtDNA CN in PLWH.
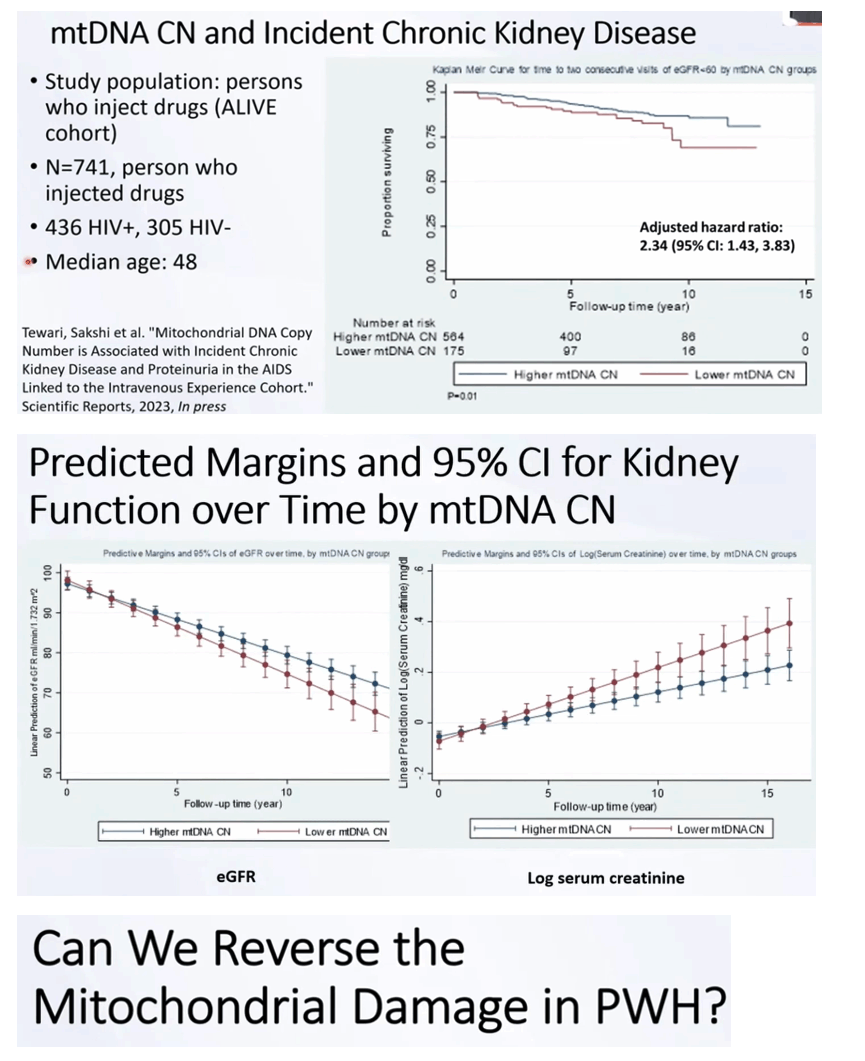
In addition, multiple chronic conditions (eg, chronic kidney disease [30] and cardiovascular disease [31]) are known to be associated with a low mtDNA CN in blood, while PLWH have elevated risk of chronic kidney disease [41] and cardiovascular disease [42] events. These HIV-related comorbidities could also be a major driver of the changes in mtDNA CN during older age. Further exploration of these relationships could provide insights useful to the development of targeted therapies to promote healthy aging among PLWH.

|
| |
|
 |
 |
|
|